6-Methoxy-2-naphthylacetic acid prodrugs
a technology of naphthylacetic acid and prodrugs, which is applied in the direction of biocide, amide active ingredients, drug compositions, etc., can solve the problems of severe irritation of the gastronintestinal tract at dosage, stomach irritation and/or ulceration
Inactive Publication Date: 2003-04-10
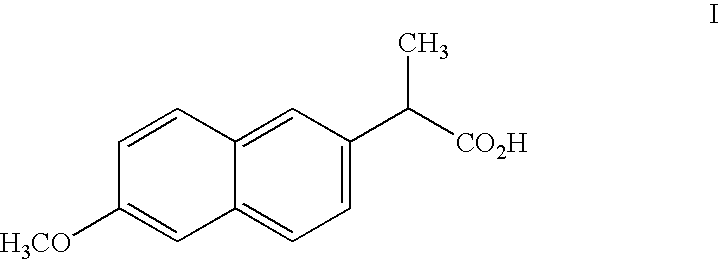
BIOCON LTD
View PDF4 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
The new 6MNA prodrugs demonstrate higher plasma levels and rapid onset of action compared to nabumetone, providing enhanced analgesic and antipyretic effects while minimizing gastrointestinal irritation.
Problems solved by technology
Compound (I) can, however, cause severe irritation of the gastronintestinal tract at dosages only slightly greater than the excess of the therapeutic dose.
Another concern with 6MNA and its related prodrugs is that the presence of the carboxylic acid moiety can cause stomach irritation and / or ulceration.
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
example 1
[0039] Synthesis of 2-(6-Methoxy-naphthalen-2-yl)-ethanol
[0040] 6-MNA (1g, 0.0046 mol) was suspended in anhydrous THF and was cooled with ice bath suspension BH3(1 M solution in THF, 5 ml) was added. The reaction mixture was stirred for 3 hours then Dl water and sodium carbonate added. THF was removed and aqueous phase extracted with ethyl acetate then washed with water, dried over Na2SO4, filtered concentrated and dried via vacuum.
[0041] Yield 93%. Melting point 110-113.degree. C. product was analyzed by elemental analysis, IR, MS, NMR.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
| Property | Measurement | Unit |
|---|---|---|
| pH | aaaaa | aaaaa |
| solubility | aaaaa | aaaaa |
| in plasma levels | aaaaa | aaaaa |
Login to View More
Abstract
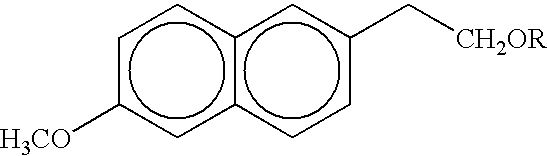
The present invention provides therapeutically effective amounts of 6MNA prodrugs. The pharmaceutical composition comprises wherein R is H or COR', wherein R' is selected from the group consisting of C1 to C6 alkyl, (CH2)mO(CH2)n, (CH2)m(OC2H4)pO(CH2)n, (CH2)m(OC2H4)p, (OCH2H4)pONO2 and (OCH2H4)pO(CH2)n wherein m is an integer from 2 to 4, and n and p are integers from 1 to 4. Alternatively, R is a therapeutic moiety.
Description
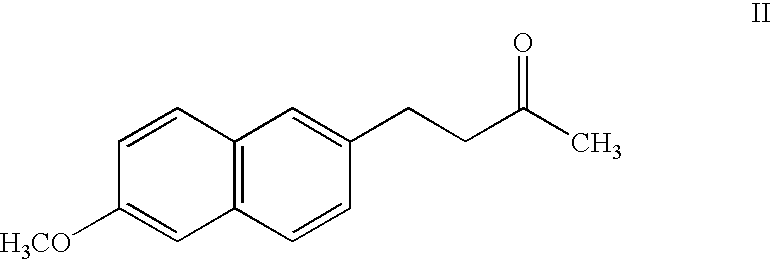
[0001] The present application is a continuation of U.S. application Ser. No. 09 / 697,795, filed Oct. 27, 2000, which claims priority from U.S. Provisional Application No. 60 / 161,864, filed Oct. 27, 1999, the disclosures of which are incorporated by reference herein in their entireties.FIELD AND BACKGROUND OF THE INVENTION[0002] The present invention relates to pharmaceutical compositions useful for treatment of inflammation in humans utilizing compounds that are prodrugs of 6-methoxy-2-naphthylacetic acid (hereinafter "6MNA").[0003] Various naphthalene derivatives are known to be useful for the treatment of inflammation and for various rheumatic and arthritic conditions. For example, Naproxen having the formula (I): 2[0004] as described in U.S. Pat. No. 4,009,197 to Fried et al. Compound (I) can, however, cause severe irritation of the gastronintestinal tract at dosages only slightly greater than the excess of the therapeutic dose.[0005] Another naphthalene derivative is nabumetone ...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(United States)
IPC IPC(8): A61K31/085A61K31/16A61P29/00A61K31/165A61K31/167A61K31/21A61K31/216A61K31/222A61K31/235A61K31/265C07C43/23
CPCA61K31/085A61K31/16A61K31/167A61K31/265A61K31/216A61K31/235A61K31/21A61P29/00A61K31/215
Inventor EKWURIBE, NNOCHIRI NKEMDYAKONOV, TATYANA A.
Owner BIOCON LTD