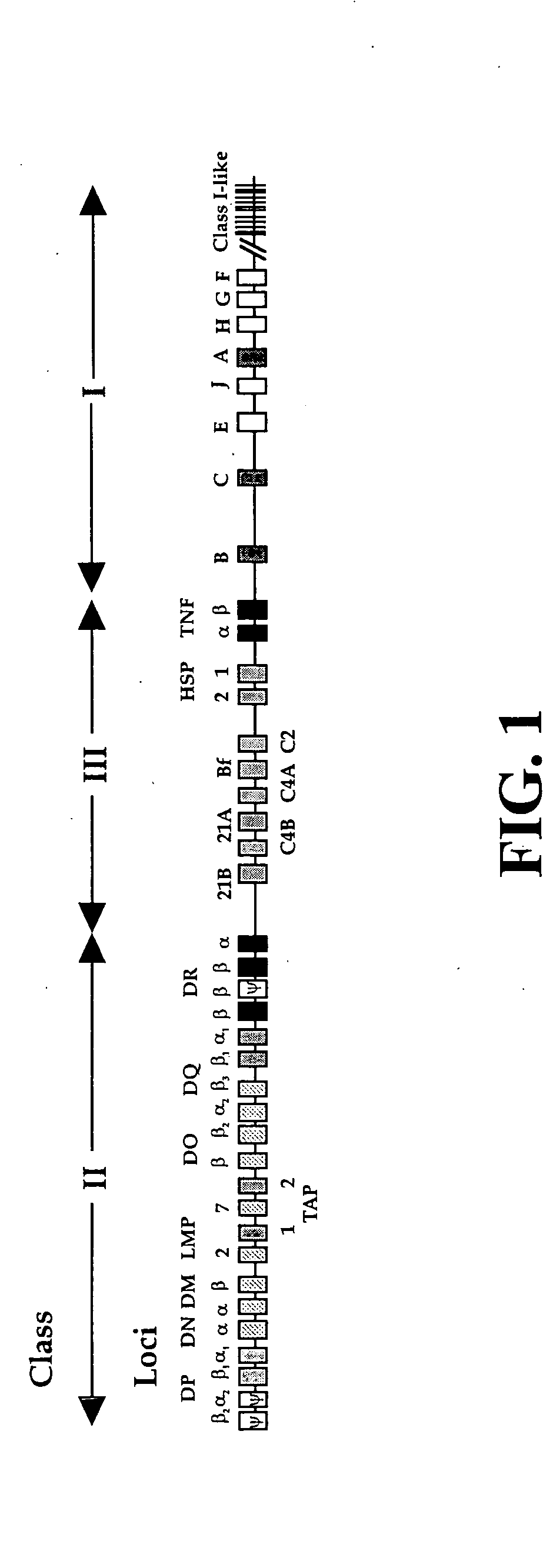
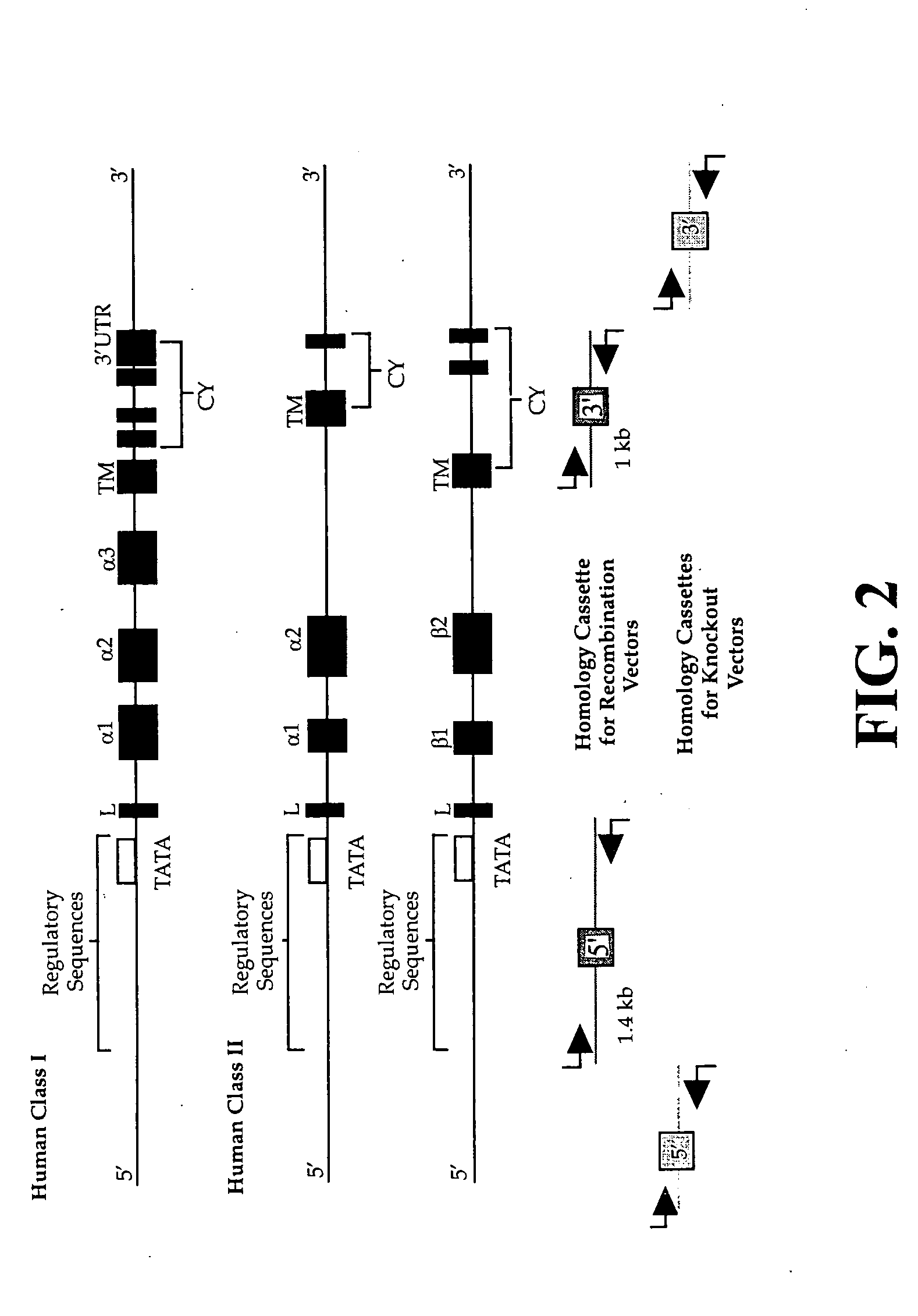
An additional level of complexity is created by the possibility of the expression of haplotype- and isotype-mismatched αβ dimers, i.e., the assembly of an a chain encoded on one haplotype and the b chain from the second haplotype, or the assembly of two chains encoded by the genes of two different class II loci, e.g., DRa Dqβ.
Further complexity is added by the fact that T cells can effectively engage either autologous or allogeneic MHC molecules.
Although the effect of mismatched MHC can be diminished by immunosuppressive drugs, rejection due to these disparities is still a major barrier to successful organ transplantation.
However, numerous transplantation studies have shown that the absence of these antigens do not make these grafts universally accepted.
Furthermore, these MHC-deficient cells cannot perform immune functions to protect the new host from pathogenic invasion.
However, for transplantation to succeed, two major barriers need to be overcome; first, the availability of suitable donor organs and second, immune rejection.
At present, the replacement of failing organs and the treatment of the rejection sequelae is restricted by the limited number of acceptable donors and the need for co-administration of toxic immuno-suppressive drugs in conjunction with long term immuno-suppressive protocols.
Presently, organ transplantation technology is only appropriate in life threatening situations.
Two major obstacles have prevented the broad application of transplantation biology.
First, the demand for transplantable organs outstrips organ availability, and second, the induction of a vigorous immunologic response results in the rejection of the donor organ (Faustman, 1995).
Practically speaking, only the HLA can be matched but even then it would be impossible to match all known HLA in an allogeneic transplant situation.
The major drawbacks to this approach are: the probability of collecting contaminated stem cells; transference of immune cells along with the graft; the expense of collecting cells, educating prospective donors, and maintaining the infrastructure; ethical issues, and availability donors, as exemplified by the problems associated with obtaining bone marrow cells, grafts, and tissues from minority groups.
This is usually done by administering cyclosporin A, azathioprine, or high doses of steroids, however, these drugs are not without side-effects and act in a non-specific manner.
The nonspecific nature of this type of approach could conceivably disrupt the fine balance between preventing rejection and the innate ability of the immune system to combat disease.
Despite the 15% annual increase in demand for transplantable organs, the world supply remains static and in some countries is on the decline.
The major problem with animal organs for transplantation goes beyond the compatibility of MHC antigens and introduces another level that deals with the recognition of species-specific antigens.
This approach to tolerize recipients is not feasible for use in human transplantation, primarily due to the high risk associated with myeloablative conditioning regimens required to achieve re-engraftment of allogeneic bone marrow (Sykes et al., 1991).
The need for autologous tissue will limit the number of centers able to perform these types of services and may make wide-scale use of these therapies impractical.
It is well accepted that the function of the β-2m is to stabilize the tertiary structure of the heavy chain of MHC class I and that the absence of the β-2m, from the MHC class I complex adversely affects the transport of the molecule (heavy chain) to the cell surface by the endoplasmic reticulum.
This approach to transplantation has not been successful for it appears that cells lacking MHC antigens are targets for natural killer (NK)-mediated cytolysis and are therefore still vulnerable to immune rejection.
Not only are MHC-deficient cells rejected by the immune system, they are unable to present self and non-self antigens to immune surveillance, seriously undermining the ability of the immune system to recognize the presence of pathogenic organisms.
No other technology can accomplish all of these things simultaneously.
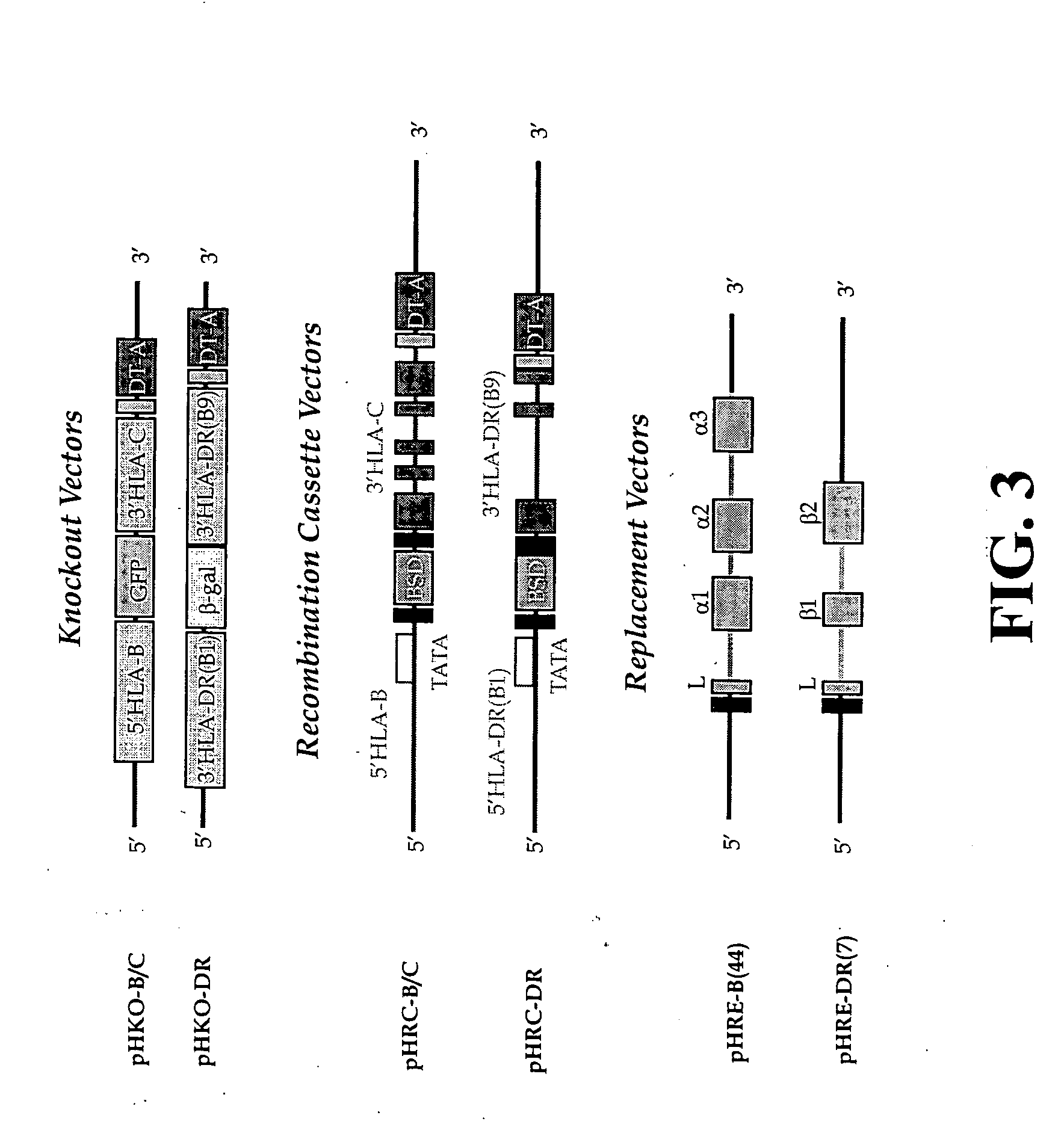
Although mammalian cells can mediate recombination between homologous DNA sequences very efficiently, they have an even greater predilection for mediating nonhomologous recombination.
The problem is to identify homologous recombination events among the vast pool of nonhomologous recombination events.
 Login to View More
Login to View More