Synthesis method of tetrahydrofurfuryl ethyl ether
A technology of ethyl tetrahydrofurfuryl ether and tetrahydrofurfuryl ether, which is applied in the field of synthesis of ethyl tetrahydrofurfuryl ether, can solve the problems of endangering the health of operators, high risk, and environmental pollution, and achieve the elimination of Risk factors, less toxic raw materials, and the effect of reducing pollution emissions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0016] Embodiment 1, synthetic ethyl tetrahydrofurfuryl ether
[0017] In a 500ML reactor, add 102g of tetrahydrofurfuryl alcohol, 0.5mol of diethyl carbonate, and 1mol of potassium hydroxide, react under stirring, control the reaction temperature at 5°C, and finish the reaction after reacting for 9 hours. For the product, the fraction at 150°C-160°C was collected to obtain 98g of the product.
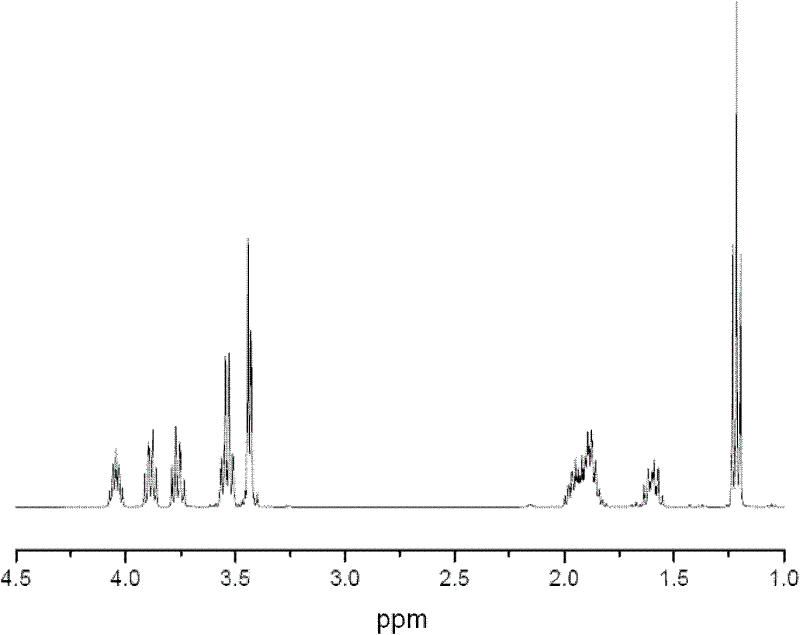
[0018] Adopt Bruker AVANCE DRX 400MHz nuclear magnetic resonance spectrometer to measure product, solvent is deuterated chloroform; Its nuclear magnetic spectrum and resonance peak assignment are as table 1 and figure 1 Shown:
[0019] Table 1. ETE's 1 H-NMR formant assignment
[0020]
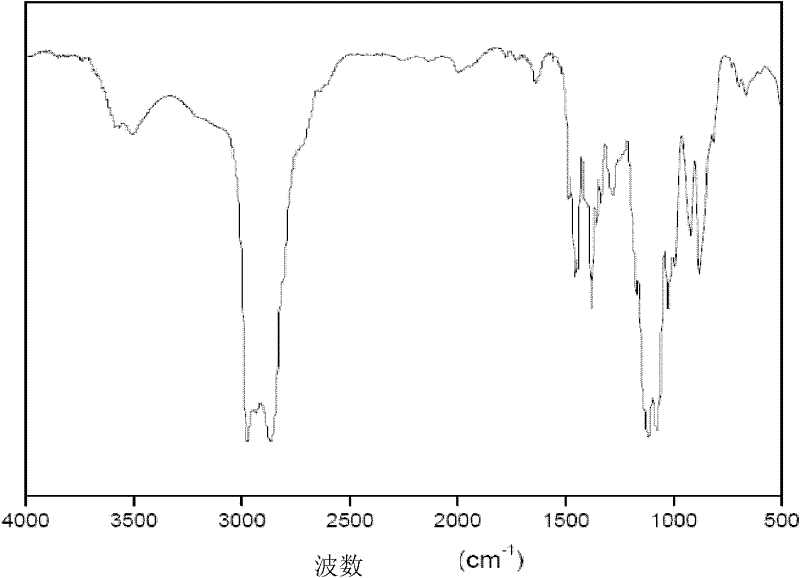
[0021] The structure of the product was analyzed by a Nicolet 20DXB FT-IR infrared chromatograph, and the composition and purity of the reaction product were analyzed by a Shimadzu GC-14A gas chromatograph. Chromatographic column: 30m×0.32mm PEG-20M capillary column; column temperature: 40°C / 3 m...
Embodiment 2
[0026] Embodiment 2, synthetic ethyl tetrahydrofurfuryl ether
[0027] In a 500ML reactor, add 102g of tetrahydrofurfuryl alcohol, 0.6mol of diethyl carbonate, and 1mol of sodium hydroxide, react under stirring, control the reaction temperature at 5°C, and finish the reaction after 9 hours of reaction. For the product, the fraction at 150°C-160°C was collected to obtain 96g of the product.
[0028] Adopt Bruker AVANCE DRX 400MHz nuclear magnetic resonance spectrometer to measure product, solvent is deuterated chloroform; Its nuclear magnetic spectrum resonant peak belongs as in Table 2:
[0029] Table 2. ETE's 1 H-NMR formant assignment
[0030]
[0031] The structure of the product was analyzed by a Nicolet 20DXB FT-IR infrared chromatograph, and the composition and purity of the reaction product were analyzed by a Shimadzu GC-14A gas chromatograph. Chromatographic column: 30m×0.32mm PEG-20M capillary column; column temperature: 40°C / 3 minutes to 100°C / 7 minutes; heatin...
Embodiment 3
[0036] Embodiment 3, synthetic ethyl tetrahydrofurfuryl ether
[0037] In a 500ML reactor, add 102g tetrahydrofurfuryl alcohol, 0.6mol diethyl carbonate, and 1mol sodium carbonate, react under stirring, control the reaction temperature at 5°C, and finish the reaction after reacting for 9 hours, and distill the reaction product at atmospheric pressure , collecting fractions at 150°C-160°C to obtain 103g of the product.
[0038] Adopt Bruker AVANCE DRX 400MHz nuclear magnetic resonance spectrometer to measure product, solvent is deuterated chloroform; Its NMR resonance peak attribution is as shown in table 3:
[0039] Table 3. ETE's 1 H-NMR formant assignment
[0040]
[0041] The structure of the product was analyzed by a Nicolet 20DXB FT-IR infrared chromatograph, and the composition and purity of the reaction product were analyzed by a Shimadzu GC-14A gas chromatograph. Chromatographic column: 30m×0.32mm PEG-20M capillary column; column temperature: 40°C / 3 minutes to 10...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com