Preparing technology of erlotinib hydrochloride
A technology for the preparation of erlotinib hydrochloride, which is applied in the field of preparation of pharmaceutical compounds, can solve problems such as unfavorable post-processing and large environmental pollution, and achieve the effects of short reaction process route, simple and easy operation, and mild reaction conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
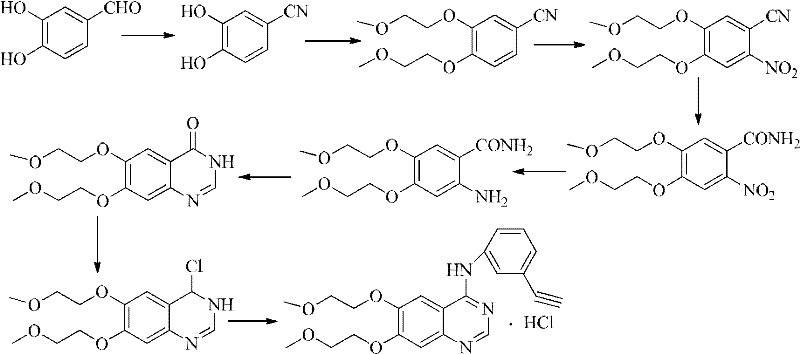
[0029] The preparation process of erlotinib hydrochloride uses 2-nitro-4,5-bis(2-methoxyethoxy)benzonitrile as the starting material to obtain 2-amino-4 through nitro reduction, 5-two (2-methoxyethoxy) benzonitriles, then prepare according to the operational route (1), specifically comprising the following steps:
[0030] 1, the preparation of 2-amino-4,5-bis(2-methoxyethoxy)benzonitrile
[0031] Mix and stir 2-nitro-4,5-bis(2-methoxyethoxy)benzonitrile (100g, 0.336mol), sodium dithionite (204g, 1.176mol) and water (1.7L), and heat to 50-55°C, continue to stir for 2 hours, quickly raise the temperature to 65-70°C, slowly add concentrated hydrochloric acid (450ml) dropwise, the temperature of the reaction solution drops to room temperature after the dropwise addition, adjust the pH value with 40% sodium hydroxide solution to 10, extracted with dichloromethane (3×75ml), the combined organic phases were washed with purified water (2×50ml) and saturated brine (1×50ml) successivel...
Embodiment 2
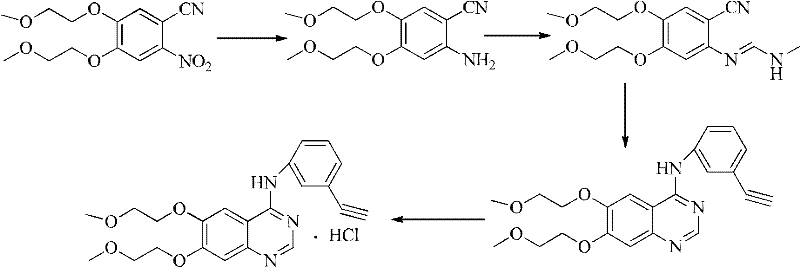
[0037] The preparation process of erlotinib hydrochloride uses 2-nitro-4,5-bis(2-methoxyethoxy)benzonitrile as the starting material to obtain 2-amino-4 through nitro reduction, 5-two (2-methoxyethoxyl) benzonitriles are prepared according to the operational route (2), specifically comprising the following steps:
[0038] 1, the preparation of 2-amino-4,5-bis(2-methoxyethoxy)benzonitrile
[0039] The operation was the same as step 1 in Example 1 to obtain a brown-yellow solid (84.65 g, 94.06%).
[0040] 2, Preparation of Schiff base of 3-ethynylaniline
[0041] Add 3-ethynylaniline (32.6, 0.28mol), toluene (800ml), acetic acid (0.13ml) and DMF-DMA (60g, 0.505mol) into the reaction flask equipped with an oil-water separator, mix and stir, and heat to reflux for 3 After the reaction was completed, the solvent was distilled off to obtain an oily liquid (37.1 g, 0.257 mol). The next reaction was carried out directly without purification.
[0042] 3. Preparation of [6,7-bis(2-m...
Embodiment 3
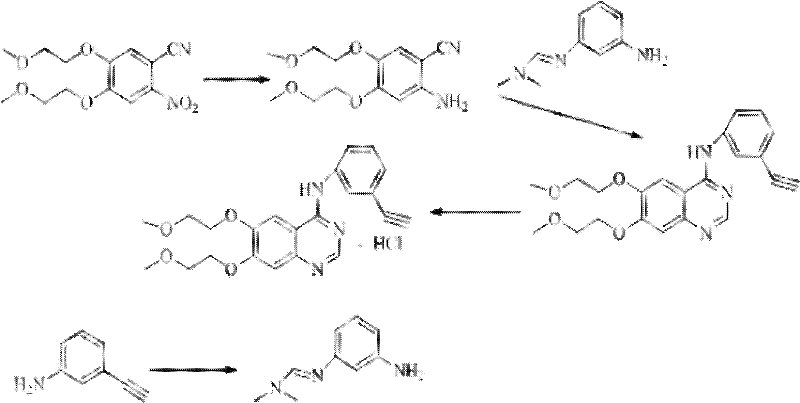
[0048] The preparation process of erlotinib hydrochloride uses 2-nitro-4,5-bis(2-methoxyethoxy)benzonitrile as the starting material to obtain 2-amino-4 through nitro reduction, 5-two (2-methoxyethoxy) benzonitriles, then prepare according to the operational route (1), specifically comprising the following steps:
[0049] 1, the preparation of 2-amino-4,5-bis(2-methoxyethoxy)benzonitrile
[0050] 2-nitro-4,5-di(2-methoxyethoxy)benzonitrile (100g, 0.336mol), iron powder (65.86g, 1.176mol) and water (1.7L) were mixed and stirred, heated to 50-55°C, continue to stir for 2 hours, quickly raise the temperature to 65-70°C, slowly add concentrated hydrochloric acid (450ml) dropwise, the temperature of the reaction solution drops to room temperature after the dropwise addition, adjust the pH value with 40% sodium hydroxide solution to 10, extracted with dichloromethane (3×75ml), and the combined organic phases were washed with purified water (2×50ml) and saturated brine (1×50ml) succ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More