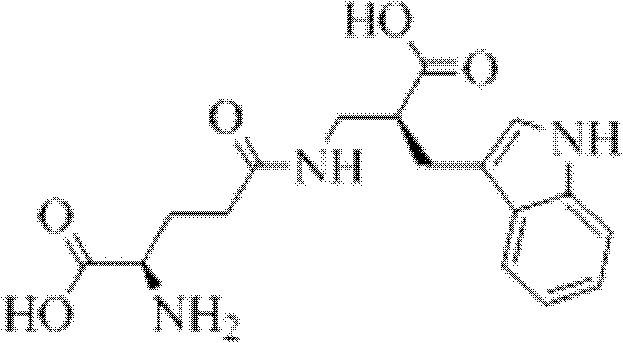
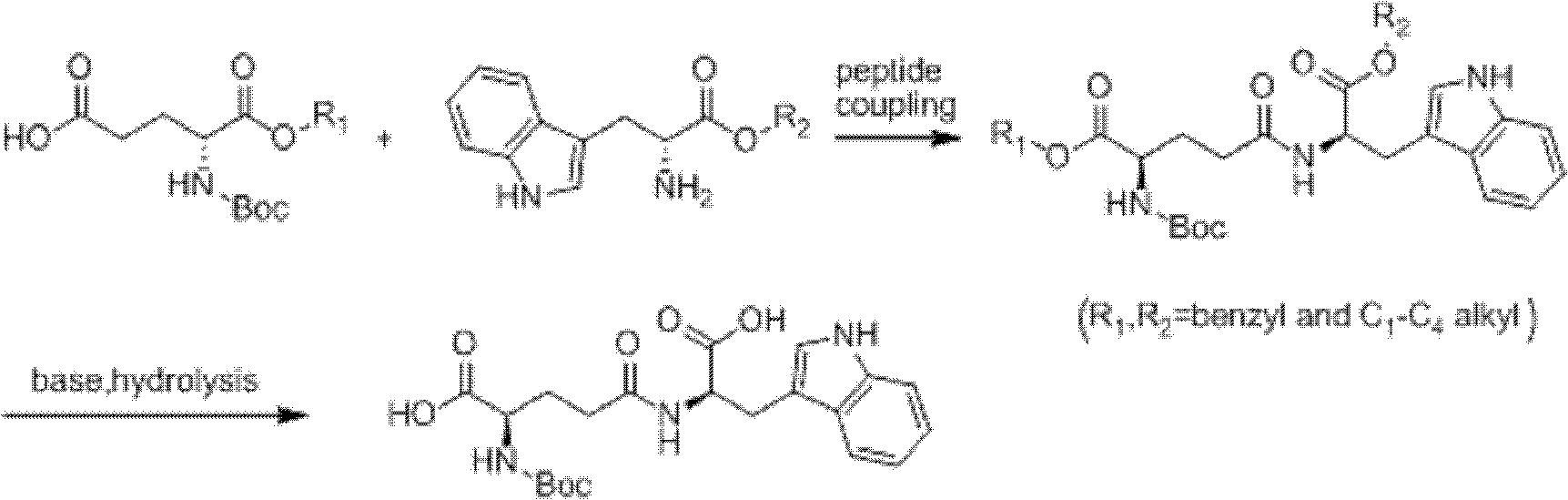
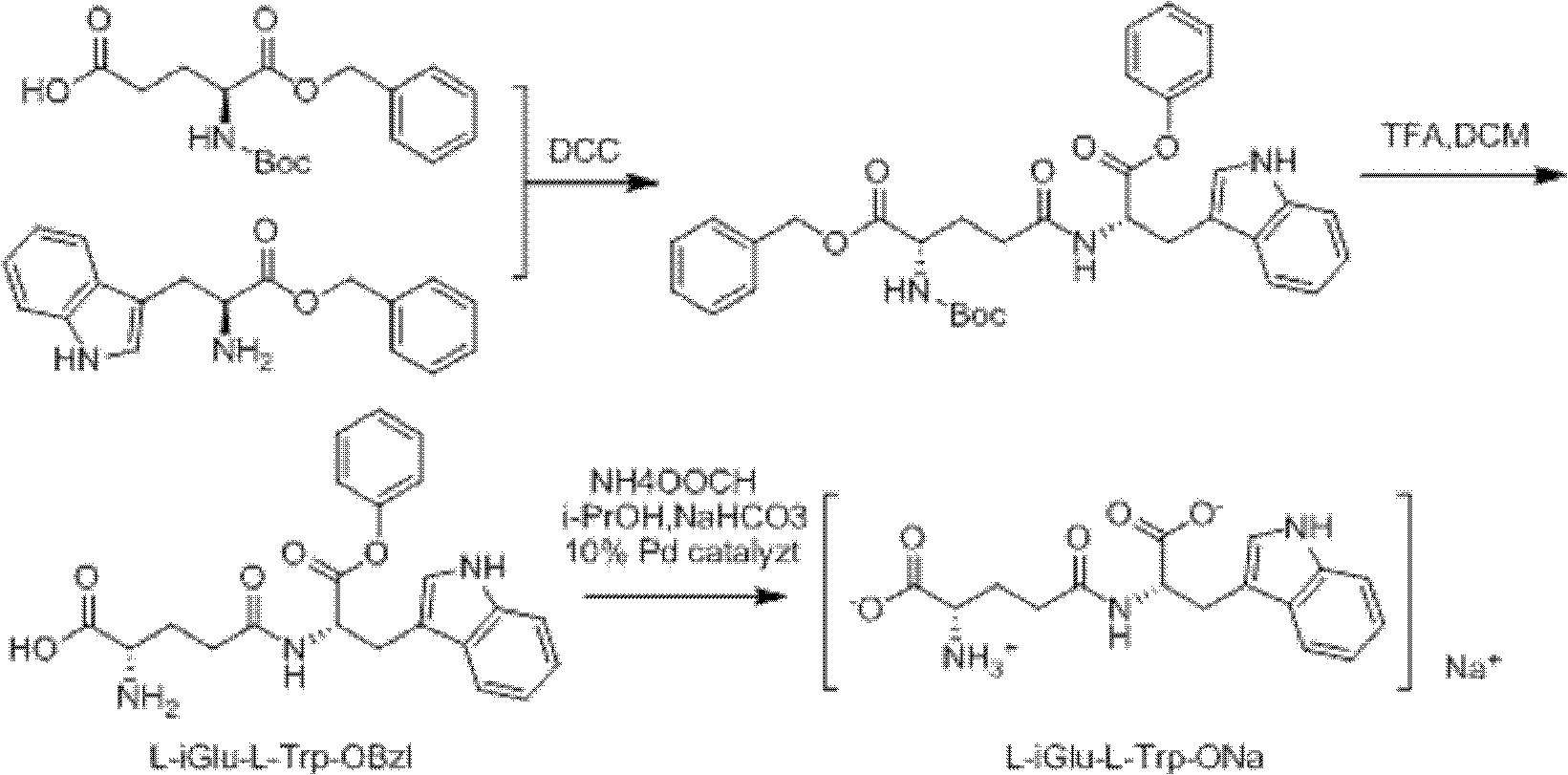
Method for synthesizing D-isoglutamine-D-tryptophan
A technology of isoglutamine and tryptophan, applied in the field of peptide synthesis, can solve the problems of difficult purification and inability to meet the needs of large-scale industrial production.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0061] Synthesis of Fmoc-D-Glu(ONb)-OBzl:
[0062] Fmoc-D-Glu-OBzl (9.18g, 20mmol) and HONb (3.9g, 22mmol) were added into a 100ml two-neck round bottom flask, and stirred with 50mL THF to dissolve. DCC (4.5g, 2.2mmol) was dissolved in 10mL THF and added dropwise in an ice-water bath, and stirred in a water bath for 3h. The reaction solution was filtered, and the filtrate was concentrated to dryness with Et 2 O / DCM (V:V=5:1) dissolves the residual oily matter, and after standing for 1 h, the insoluble matter is removed by filtration. The filtrate is concentrated to dryness by rotary evaporation and then dissolved in 10 mL THF for later use.
[0063] Synthesis of Fmoc-D-Glu(D-Trp-OBzl)-OBzl:
[0064] Add D-Trp-OBzl.HCl (6.6g, 20mmol) and Na in a 250ml two-necked round bottom flask 2 CO 3 (10.6g, 100mmol), dissolved in 40mL deionized water, added 30mL THF and stirred for 10min, slowly added the activated ester THF solution prepared in Example 1, and stirred at room temperatu...
Embodiment 2
[0072] Synthesis of Fmoc-D-Glu(ONb)-OBzl:
[0073] Fmoc-D-Glu-OBzl (9.18g, 20mmol) and HONb (3.9g, 22mmol) were added into a 100ml two-neck round bottom flask, and stirred with 50mL THF to dissolve. DCC (4.5g, 2.2mmol) was dissolved in 10mL THF and added dropwise in an ice-water bath, and stirred in a water bath for 3h. The reaction solution was filtered, and the filtrate was concentrated to dryness with Et 2 O / DCM (V:V=5:1) dissolves the residual oily matter, and after standing for 1 h, the insoluble matter is removed by filtration. The filtrate is concentrated to dryness by rotary evaporation and then dissolved in 10 mL THF for later use.
[0074] Synthesis of Fmoc-D-Glu(D-Trp-OBzl)-OBzl:
[0075] Add D-Trp-OBzl.HCl (6.6g, 20mmol) and Na in a 250ml two-necked round bottom flask 2 CO 3 (10.6g, 100mmol), dissolved in 40mL deionized water, added 30mL THF and stirred for 10min, slowly added the activated ester THF solution prepared in Example 1, and stirred at room temperatu...
Embodiment 3
[0083] Synthesis of Fmoc-D-Glu(ONb)-OMe:
[0084] Add Fmoc-D-Glu-OMe (7.66g, 20mmol) and HONb (3.9g, 22mmol) into a 100ml two-necked round-bottomed flask, and stir to dissolve with 50mL THF. DCC (4.5g, 2.2mmol) was dissolved in 10mL THF and added dropwise in an ice-water bath, and stirred in a water bath for 3h. The reaction solution was filtered, and the filtrate was concentrated to dryness with Et 2 O / DCM (V:V=5:1) dissolves the residual oily matter, and after standing for 1 h, the insoluble matter is removed by filtration. The filtrate is concentrated to dryness by rotary evaporation and then dissolved in 10 mL THF for later use.
[0085] Synthesis of Fmoc-D-Glu(D-Trp-OMe)-OMe:
[0086] Add D-Trp-OMe.HCl (6.6g, 20mmol) and Na in a 250ml two-necked round bottom flask 2 CO 3 (10.6g, 100mmol), dissolved in 40mL deionized water, added 30mL THF and stirred for 10min, slowly added the activated ester THF solution prepared in Example 2, and stirred at room temperature for 4h. ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com