Drug information management device and drug information management method
A drug information, drug technology, applied in the direction of drugs or prescriptions, drug reference, patient-specific data, etc., can solve problems such as drug administration limit
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
no. 1 Embodiment approach )
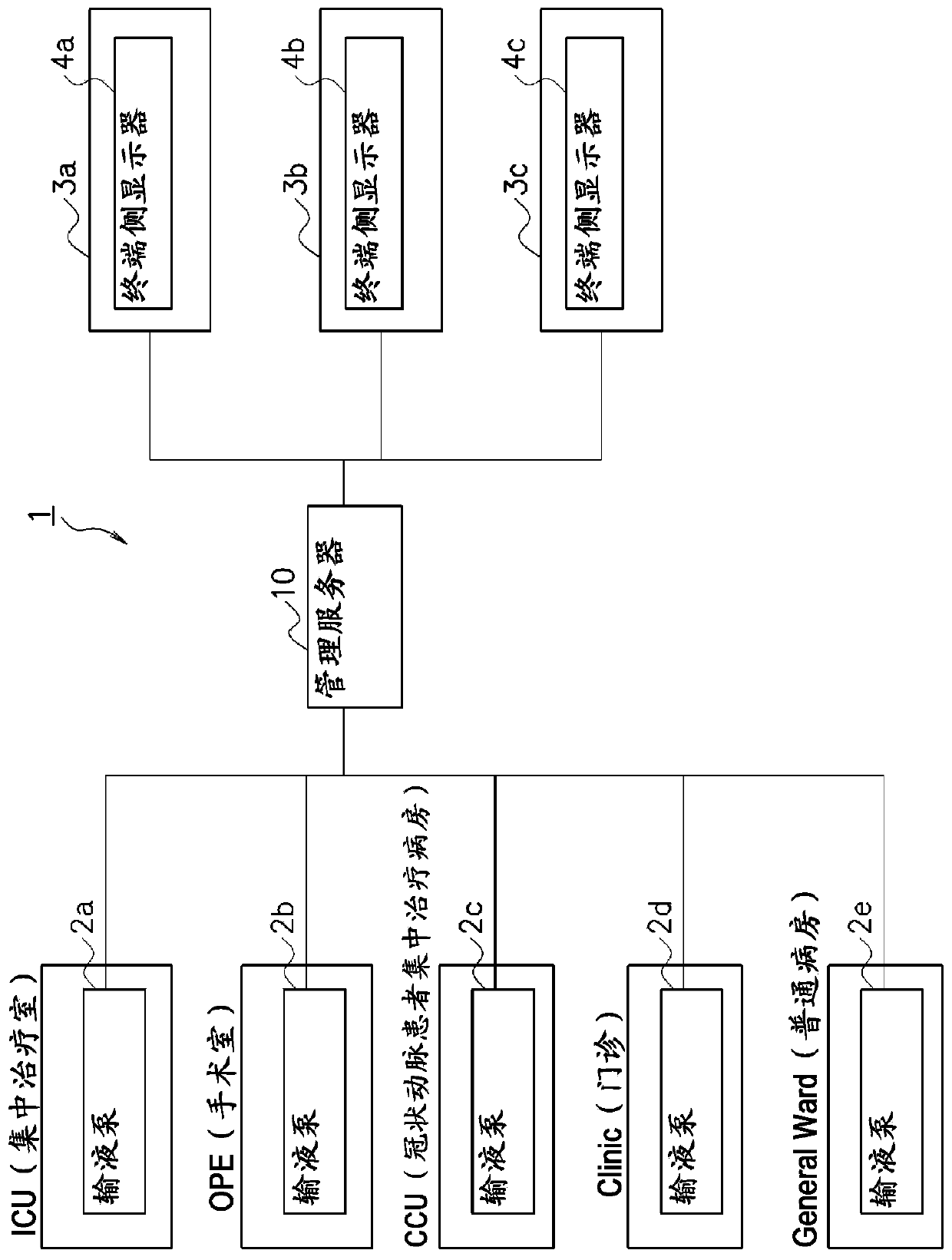
[0047] figure 1 It is a schematic diagram showing a pump CQI (Continuous Quality Improvement) (Continuous Quality Improvement) system 1 of the first embodiment including, for example, a management server 10 as the medical information management device of the present invention.
[0048] like figure 1 As shown, the pump CQI system 1 is equipped with infusion fluids configured in the ICU (intensive treatment room), OPE (operating room), CCU (coronary artery patient intensive treatment ward), Clinic (outpatient clinic), General Ward (general ward) and the like in the hospital. Pumps 2a to 2e etc.
[0049] The infusion pump 2 a and the like are medical devices used together with devices such as infusion to accurately administer medicines and the like to patients.
[0050] In addition, in this embodiment, the infusion pump 2a and the like are used for description, but as the medical device of the present invention, a syringe pump or the like may be used in addition to the infusion...
no. 2 Embodiment approach )
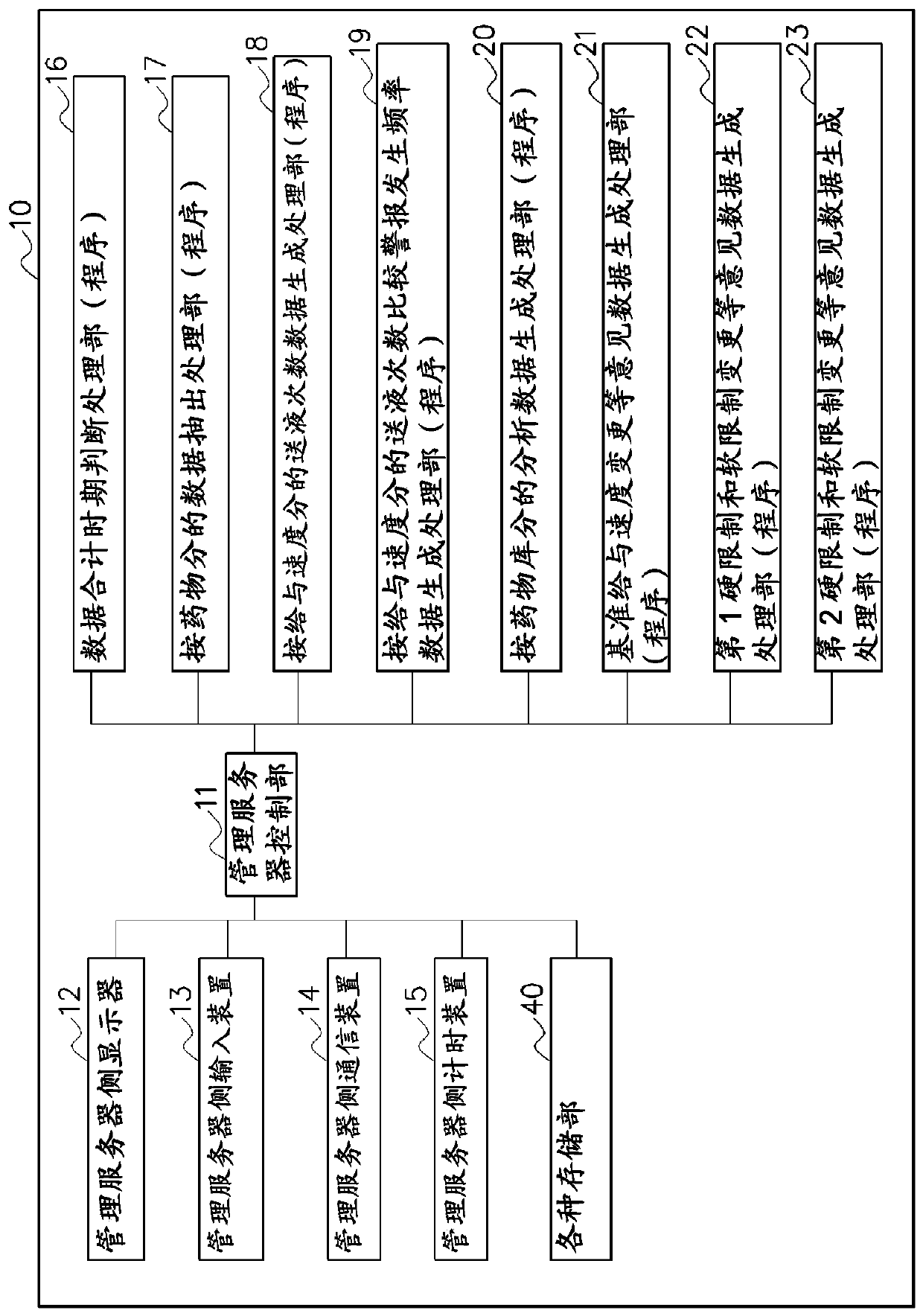
[0122] Figure 10 It is a schematic block diagram showing the main configuration of the management server 100 according to the second embodiment of the present invention.
[0123] Many configurations of the present embodiment are the same as those of the above-mentioned first embodiment, and therefore the same reference numerals and the like are attached to the same configurations and the like, and description thereof will be omitted, and the following description will focus on different points.
[0124] Figure 11 It is a schematic flowchart showing main operations of the management server 100 and the like in the CQI system according to this embodiment.
[0125] First, in the present embodiment, the steps ST1 to ST12 are executed in the same manner as in the first embodiment described above, and "analysis data by drug library" is generated and stored.
[0126] Next, go to ST21. In ST21, Figure 10 The "data generation processing unit (program) 101" for the change of refer...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More