A pharmaceutical composition for preventing and treating angina pectoris of coronary heart disease and its application
A technology of coronary heart disease angina pectoris and composition, which is applied in the field of medicine to achieve the effect of increasing patient compliance, increasing safety, and increasing NO content
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
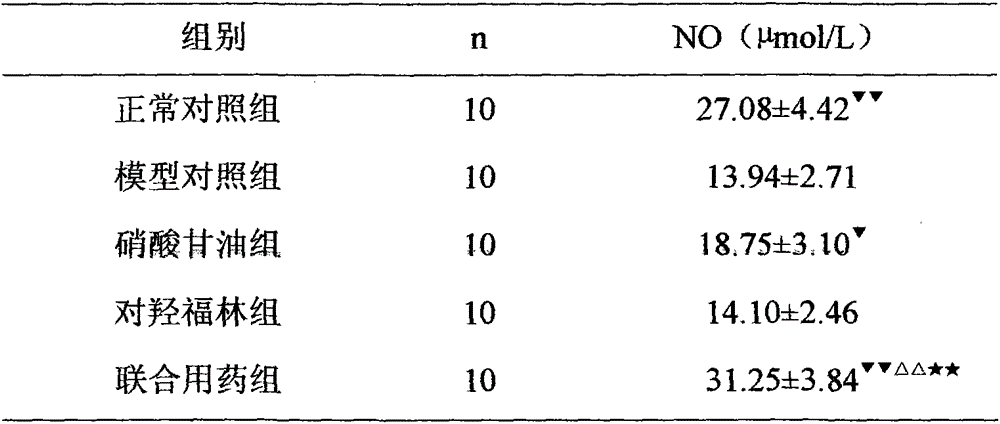
[0017] Example 1 Effect of Nitroglycerin Combined with Paraxyephrine on Serum NO Content in Rats with Acute Myocardial Ischemia
[0018] 50 healthy Wistar rats, weighing 220-250g, male, were randomly divided into the following five groups: normal control group, model control group, nitroglycerin group, p-hydroxyephrine group and combined drug group, 10 in each group. Taking the rat body weight as the benchmark, the rats in the nitroglycerin group were intravenously injected with nitroglycerin 0.2 mg / kg, the rats in the p-hydroxyephrine group were injected with 20 mg / kg p-hydroxyephrine intravenously, and the rats in the combined drug group were injected with nitroglycerin 0.1 mg / kg intravenously. kg and 10 mg / kg of p-hydroxyephrine, the model control group and the normal control group were intravenously injected with equal volume of normal saline. Each group was injected once a day between 9:00 and 10:00 am for 7 consecutive days. Ten minutes after the injection on the 5th, 6...
Embodiment 2
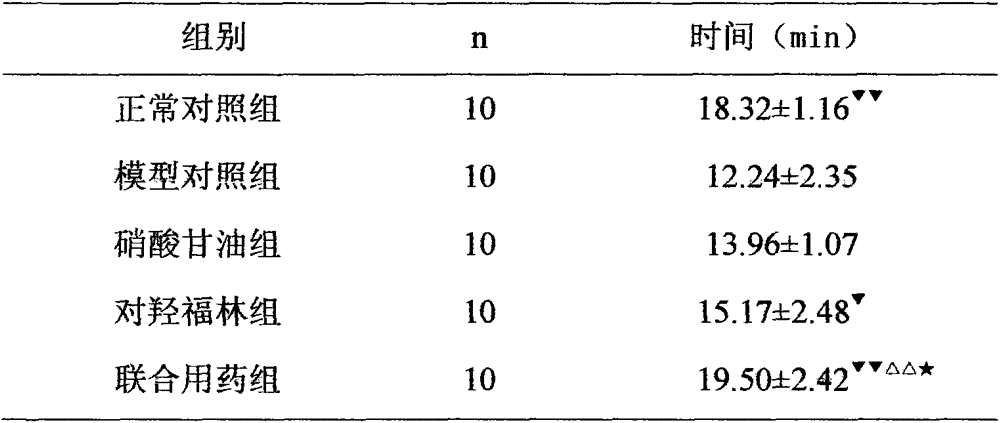
[0024] Example 2 Effect of nitroglycerin combined with oxyephrine on hypoxic tolerance time in acute myocardial ischemia mice
[0025] 50 healthy Kunming mice, weighing 18-22g, half male and half male, were randomly divided into the following five groups: normal control group, model control group, nitroglycerin group, p-hydroxyephrine group and combined drug group, 10 mice in each group . Based on the body weight of the mice, the mice in the nitroglycerin group were intravenously injected with 0.3 mg / kg of nitroglycerin, the mice in the p-hydroxyephrine group were injected with 30 mg / kg of p-hydroxyephrine intravenously, and the mice in the combined drug group were injected with 0.15 mg / kg of nitroglycerin intravenously. kg and 15 mg / kg of p-hydroxyephrine, the model control group and the normal control group were intravenously injected with equal volume of normal saline. Each group was injected once a day between 9:00 and 10:00 a.m. for 8 consecutive days. Ten minutes after...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap