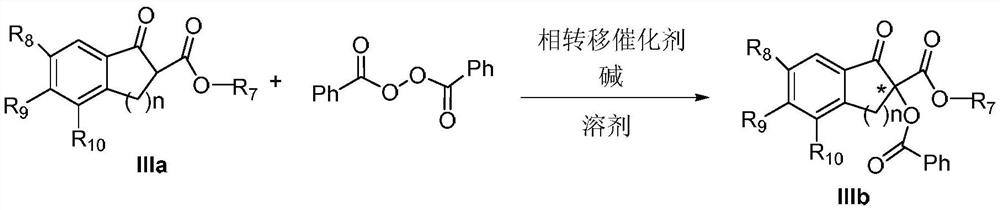
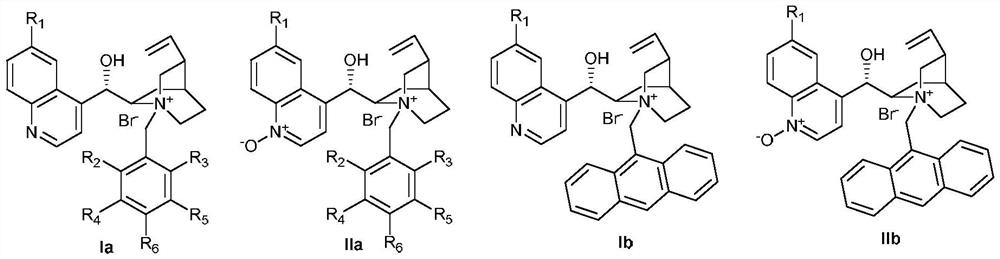
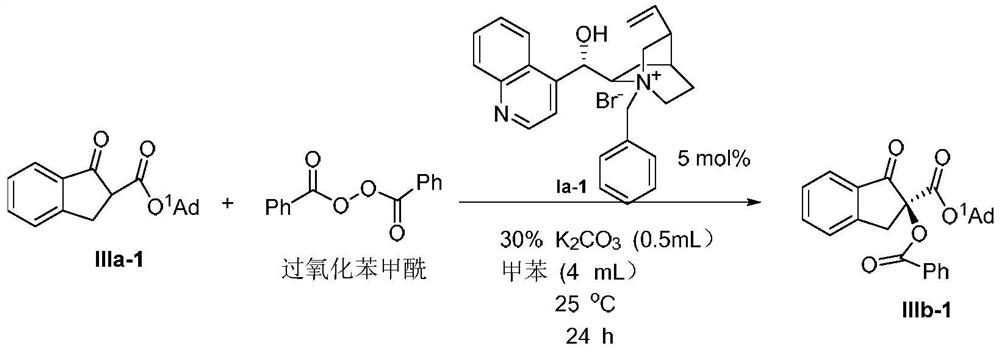
Phase-transfer-catalyzed asymmetric α-benzoylation of β-keto esters
A technology of phase transfer catalysis and phase transfer catalyst, applied in organic chemistry methods, chemical instruments and methods, preparation of carboxylate esters, etc., to achieve good catalytic effect and easy separation.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0024] Preparation of (S)-2-benzoyl-1-indanone-2-carboxylic acid methyl ester IIIb-1
[0025]
[0026] Weigh 0.1mmol 1-indanone-2-carboxylate amantadine IIIa-1, add 5mol% phase transfer catalyst Ia-1, 0.15mol benzoyl peroxide (mass fraction 70%), put it into a 20mL single-port reaction tube, add K with a mass concentration of 30% 2 CO 3 The aqueous solution was 0.5 mL, 4 mL toluene, and the reaction was stirred at 25°C. After reacting for 24 hours, the mixed solution was extracted 3 times with ethyl acetate, washed 3 times with water, dried over anhydrous sodium sulfate, filtered, and spin-dried. The crude product was separated by column chromatography (petroleum ether: ethyl acetate=25:1) to obtain the asymmetric benzoylation product IIIb-1 (31mg, yield 72%, 43%ee); [α] D 25 110.6(c 0.62, CHCl 3 ) 1 H NMR(400MHz, Chloroform-d) δ8.21–8.08(m,2H), 7.88(d,J=7.6Hz,1H), 7.73–7.66(m,1H), 7.60(d,J=7.4Hz, 1H),7.54–7.42(m,4H), 4.14(d,J=17.4Hz,1H), 3.47(d,J=17.4Hz,1H), 2.17–2.09(m,3H),2....
Embodiment 66
[0071] Preparation of (S)-2-benzoyl-1-indanone-2-carboxylic acid methyl ester IIIb-1 (phase transfer catalyst recycling)
[0072]
[0073] Weigh 2.5mmol 1-indanone-2-carboxylate IIIa-1, add 2.5mol% phase transfer catalyst II-b, 3.8mmol benzoyl peroxide (mass fraction 70%), put it into a 500mL reaction flask, add K with a mass concentration of 30% 2 CO 3 25mL of aqueous solution, 250mL of toluene, stirred at 15°C for reaction. After 24 hours of reaction, the mixture was separated into layers. The phase transfer catalyst was insoluble in the organic layer and suspended in the aqueous layer. The organic layer was collected and spin-dried to dry the organic solvent. Petroleum ether: ethyl acetate=25:1) to obtain the asymmetric benzoylation product IIIb-1. Add 2.5mmol 1-indanone-2-adamantyl carboxylate IIIa-1, 3.8mmol benzoyl peroxide (mass fraction 70%), 250mL toluene to the water layer containing the phase transfer catalyst again, continue to stir the reaction, after the reaction is...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap