Tumor tissue implant liquid as well as preparation method and application thereof
A tumor tissue and tissue technology, applied in biochemical equipment and methods, tissue culture, tumor/cancer cells, etc., can solve the problem that the molecular composition cannot accurately simulate the human TME, affect the research on the invasion process of human cancer, and affect the success rate of pharmaceutical research and development and other problems, to achieve the effect of increasing the success rate of tumor formation, low cost and easy operation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0018] Example 1: Preparation of Human Ewing Sarcoma Cell Line RD-ES Cell Homogenate
[0019] Human Ewing sarcoma RD-ES cells were incubated at 37°C in 5% CO 2 Cultured in a humidified environment, trypsin (EDTA) was routinely passaged, and the medium (85% RPMI-1640+15% FBS) was replaced every 2-3 days. Inject 2×10 subcutaneously in nude mice at 4-6 weeks 6 RD-ES cells, a personal Ewing sarcoma cell line, when the sarcoma volume in mice reaches 250 mm 3 At the same time, the mice were sacrificed, the sarcoma tissue was taken out, and frozen with liquid nitrogen; the frozen sarcoma tissue was ground into a tissue powder with a freeze grinder, and 10 g of the tissue powder was resuspended in 20 ml of pre-cooled 3.4 M NaCl buffer (pH 7.4 ), after homogenization and centrifugation, resuspend in 20 ml of the above-mentioned NaCl buffer, use the DC protein assay (Bio-Rad) to measure the protein concentration, and dilute different batches of tissue fluid with cell culture medium to...
Embodiment 2
[0020] Example 2: Configuration of Tumor Tissue Implantation Solution
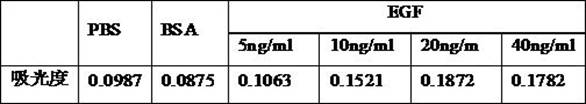
[0021] Take the human Ewing sarcoma cell line RD-ES cell homogenate prepared in Example 1, add different concentrations (5, 10, 20, 40 ng / ml) of EGF factors to prepare tumor implants containing different concentrations of EGF factors liquid. The cell adhesion test was performed on the obtained tumor implant fluid. In a 96-well plate, 100 μl PBS, 10 μg / ml BSA (Sigma-Aldrich), and the above-mentioned tumor implantation solutions containing different concentrations of EGF factors were added to each well, and each concentration was added to six wells, and coated for 24 hours. The implant solution was diluted to 1:10 in PBS, and the excess liquid was removed the next day, and the culture plate was incubated with 100 μl / well 0.1% BSA for 2 hours, and washed with PBS. Add 6000 A549 cells in 100 μl serum-free medium to each well, and culture them in an incubator for 2 h. Non-adherent cells were washed away with...
Embodiment 3
[0025] Example 3: Configuration of Tumor Tissue Implantation Solution
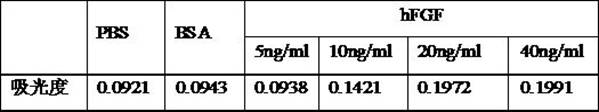
[0026] Take the human Ewing sarcoma cell line RD-ES cell homogenate prepared in Example 1, add different concentrations (5, 10, 20, 40 ng / ml) of hFGF to prepare tumor implantation solutions containing different concentrations of EGF factors . The same method as in Example 2 was used to conduct a cell adhesion test on the obtained tumor implantation fluid. Table 2 is a comparison table of the effects of tumor implant fluids containing different concentrations of hFGF factors on cell adhesion.
[0027] Table 2. Comparison table of the effect of tumor implantation fluid containing different concentrations of hFGF on cell adhesion
[0028]
[0029] It can be seen from Table 2 that the tumor implantation solution prepared after adding hFGF factor at a concentration of 10-40 ng / ml all had high absorbance, and the absorbance measured when the concentration of hFGF factor was 20 ng / ml was the highest.
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap