Quality control system for rapidly screening SARS-CoV-2 virus nucleic acid
A virus nucleic acid, sars-cov-2 technology, applied in the field of quality control system for rapid screening of SARS-CoV-2 virus nucleic acid, can solve the problems of low positive rate of virus nucleic acid detection, reliability doubts, etc., to ensure repeatability The effect of stability, accuracy, and high sensitivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0037] A quality control system for rapid screening of SARS-CoV-2 virus nucleic acid: including the collection of samples to be tested, the selection of detection reagents and detection instruments, the construction of detection methods, the interpretation of detection results and the evaluation indicators of effectiveness, statistics Processing; specifically include the following steps:
[0038] Step 1: Collection of samples to be tested:
[0039] 50 cases of throat swab samples with known SARS-CoV-2 virus nucleic acid test results were provided by the Municipal Center for Disease Control and Prevention, including 7 positive samples and 43 negative samples;
[0040] Collect 1,286 nucleic acid preparation / test failure samples out of 37,250 samples tested with reagent A from February 4, 2020 to March 2, 2020;
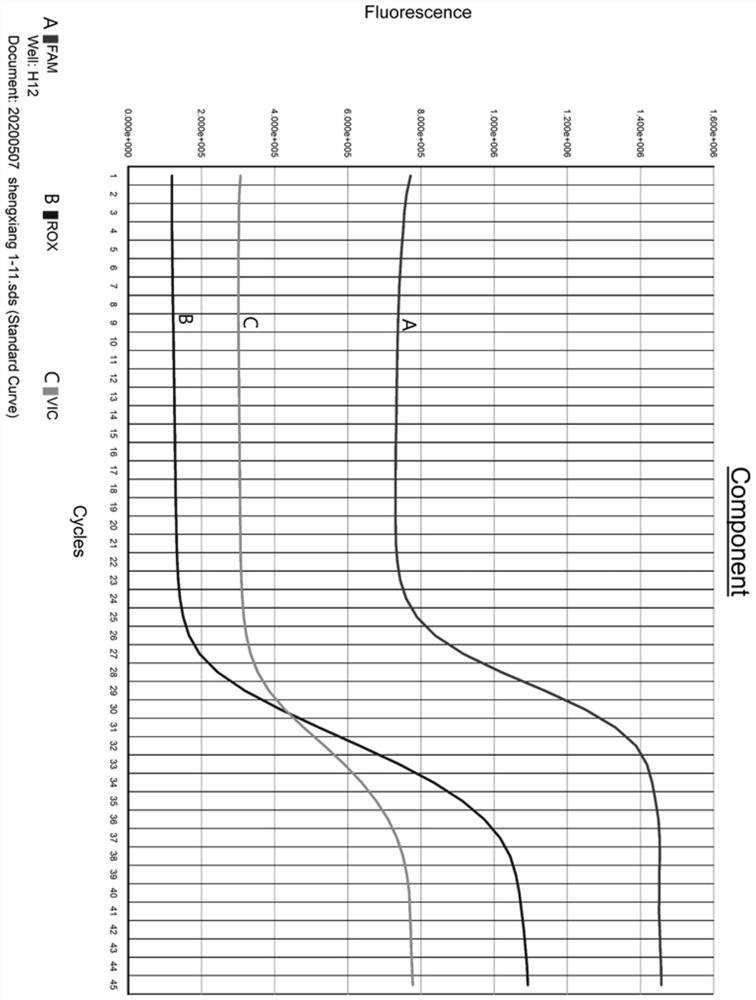
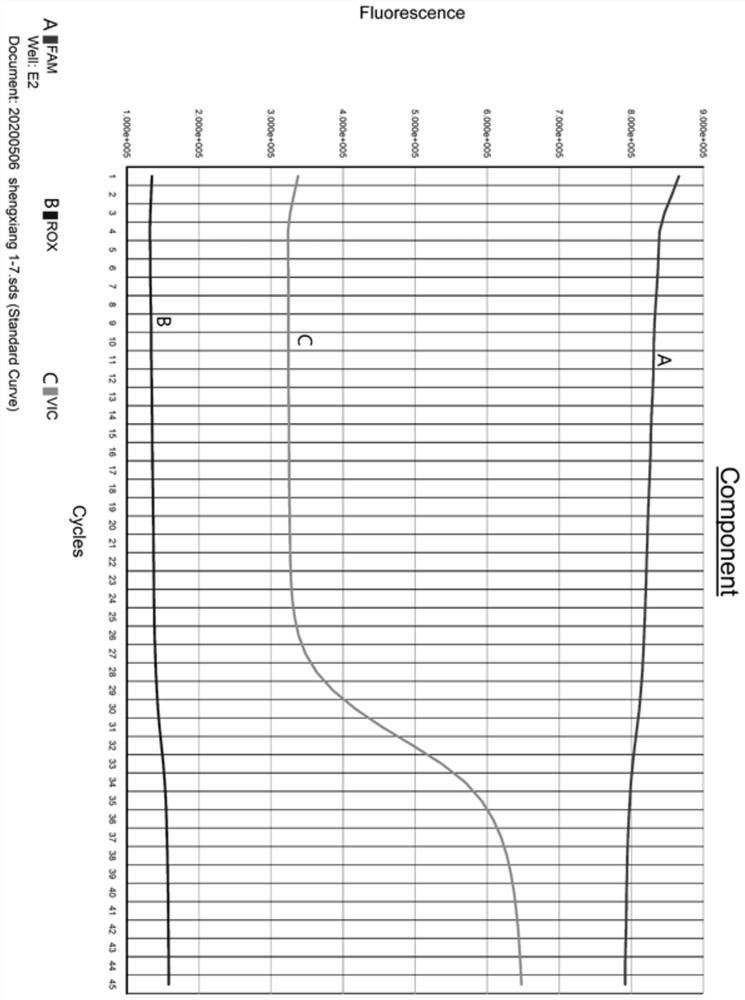
[0041] Collect 68 suspected samples of the 2019 novel coronavirus SARS-CoV-2 ORF 1ab gene, nucleocapsid protein N gene and double-gene signal elevation;
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap