Auxiliary diagnosis and treatment artificial intelligence application evaluation framework and evaluation method
A technology of artificial intelligence and evaluation indicators, applied in the field of health technology evaluation, can solve the problems of lack of medical artificial intelligence evaluation models and methods
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0040] The following will clearly and completely describe the technical solutions in the embodiments of the present invention with reference to the accompanying drawings in the embodiments of the present invention. Obviously, the described embodiments are only some, not all, embodiments of the present invention. Based on the embodiments of the present invention, all other embodiments obtained by persons of ordinary skill in the art without making creative efforts belong to the protection scope of the present invention.
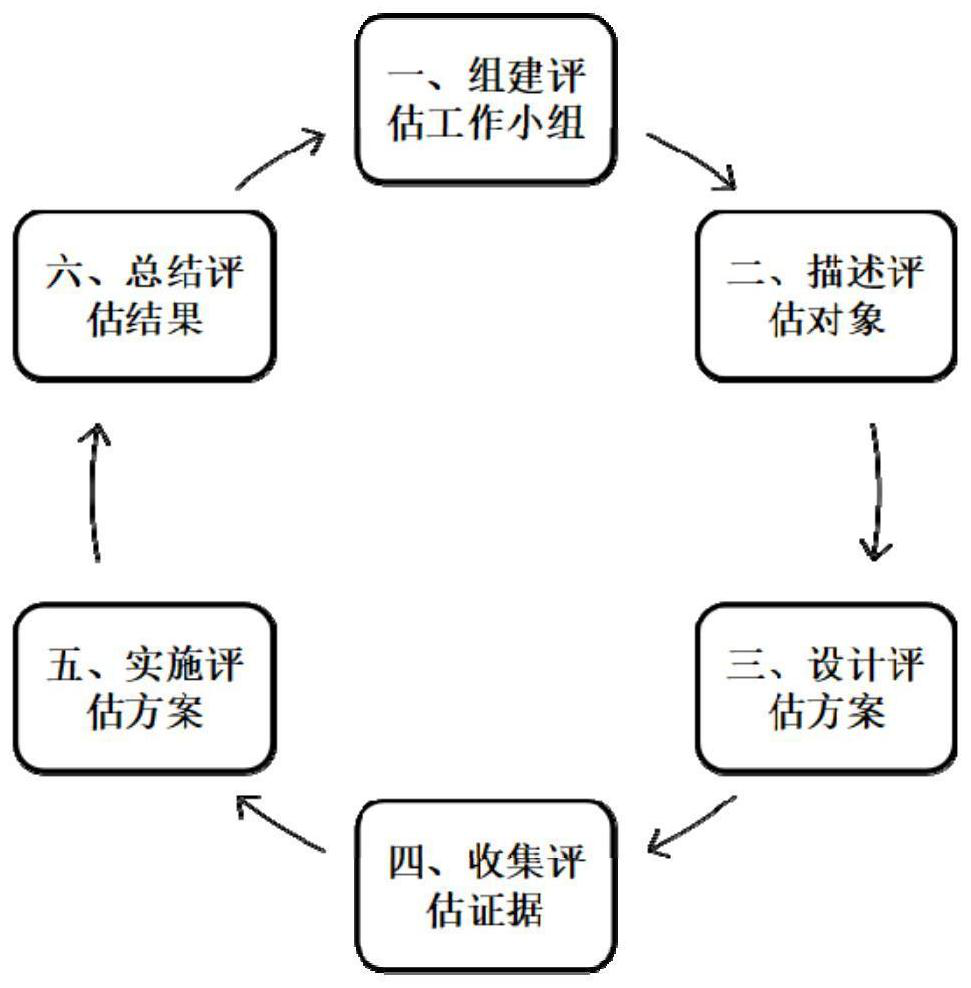
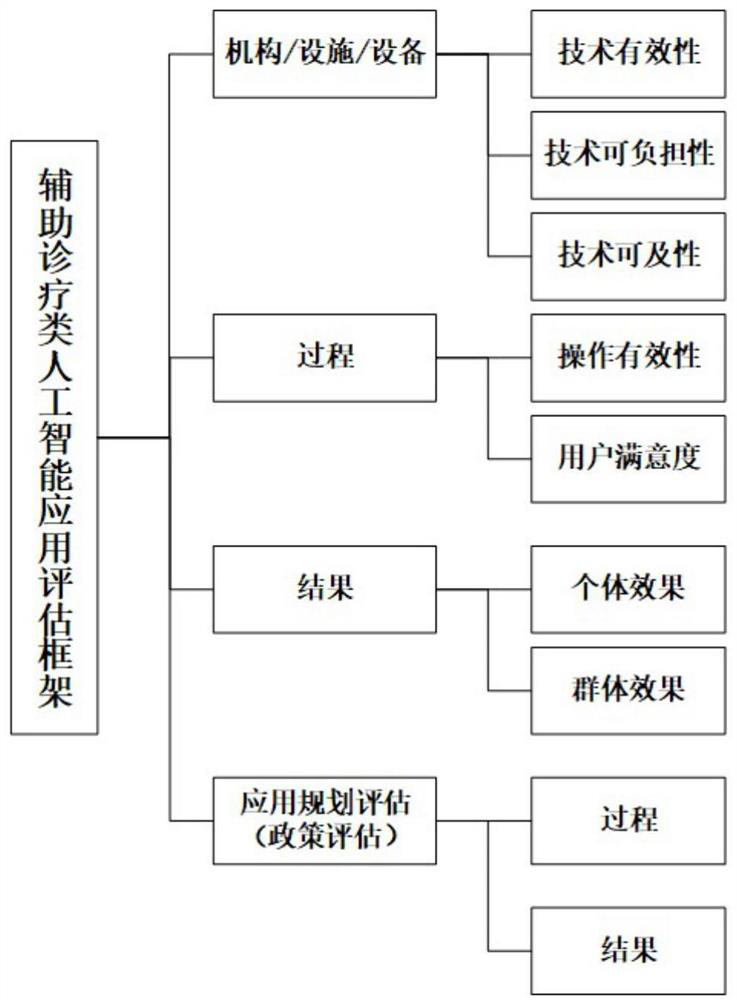
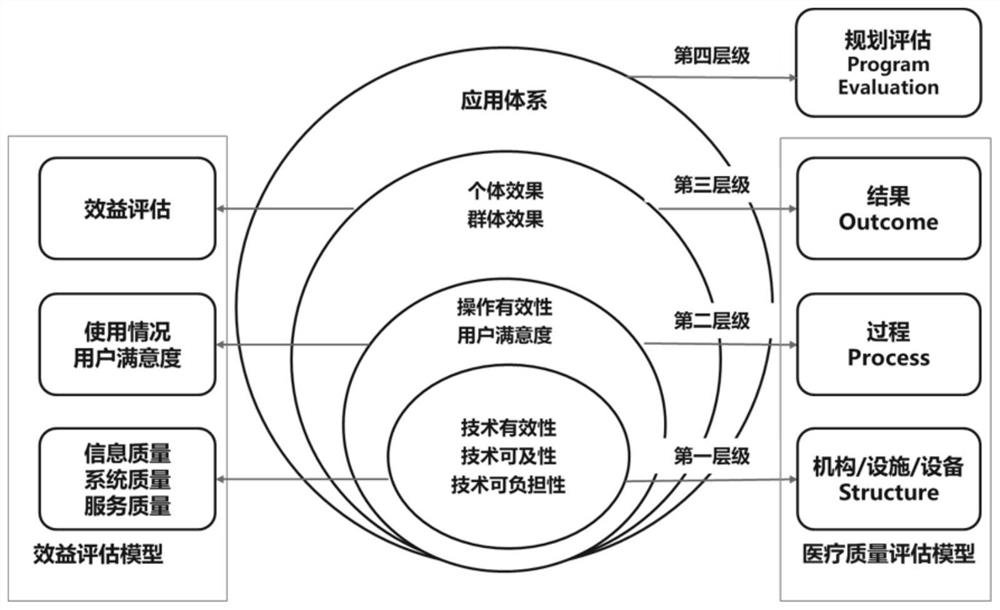
[0041] Such asfigure 1 As shown, this specific implementation mode adopts the following technical solution. The artificial intelligence application evaluation framework for auxiliary diagnosis and treatment described in the present invention includes the organization / facility / equipment layer, the process layer, the result layer and the application planning evaluation layer; where the organization / facility The / equipment layer mainly considers the three dimension...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com