Patents
Literature
8216 results about "Sonification" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
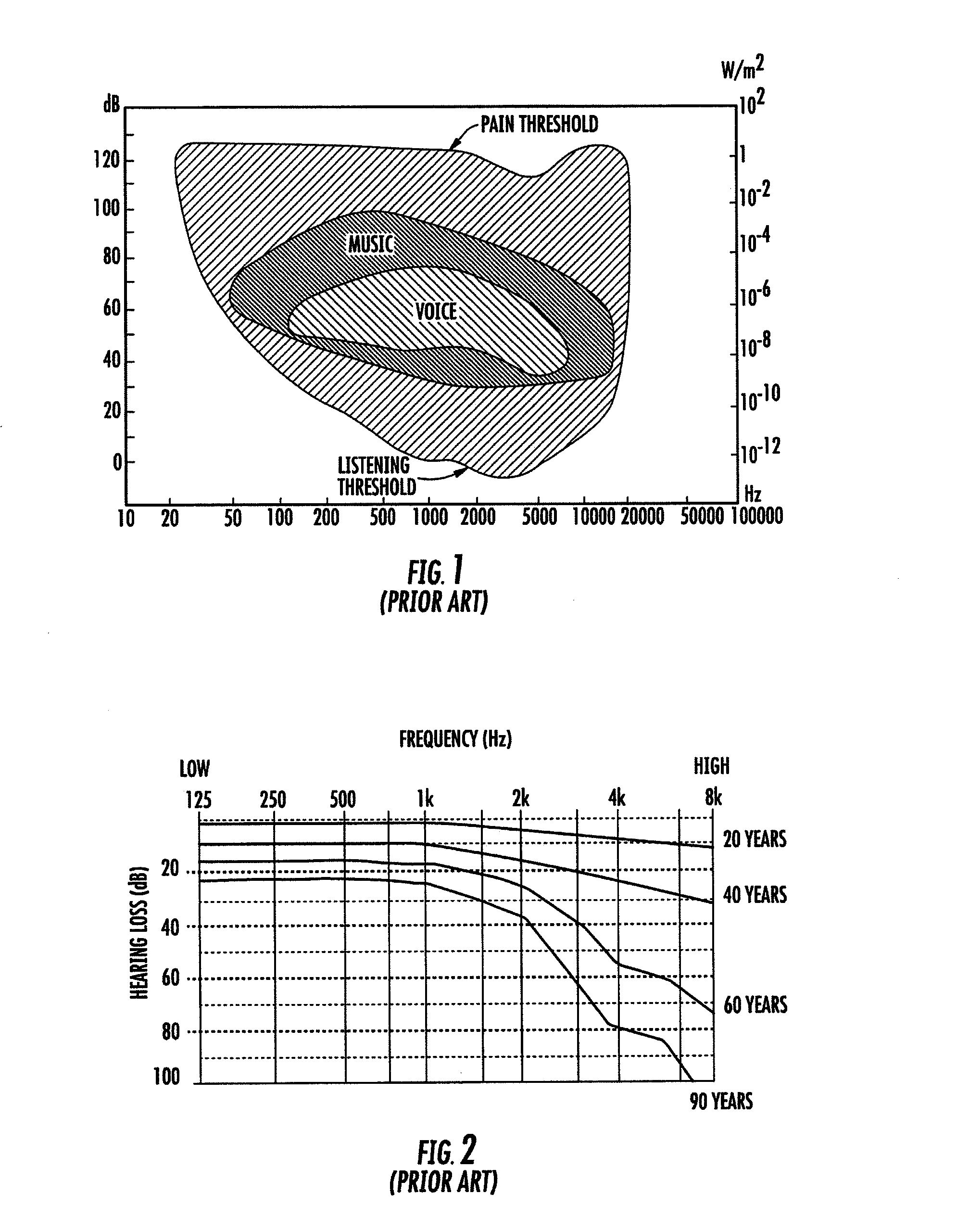
Sonification is the use of non-speech audio to convey information or perceptualize data. Auditory perception has advantages in temporal, spatial, amplitude, and frequency resolution that open possibilities as an alternative or complement to visualization techniques.
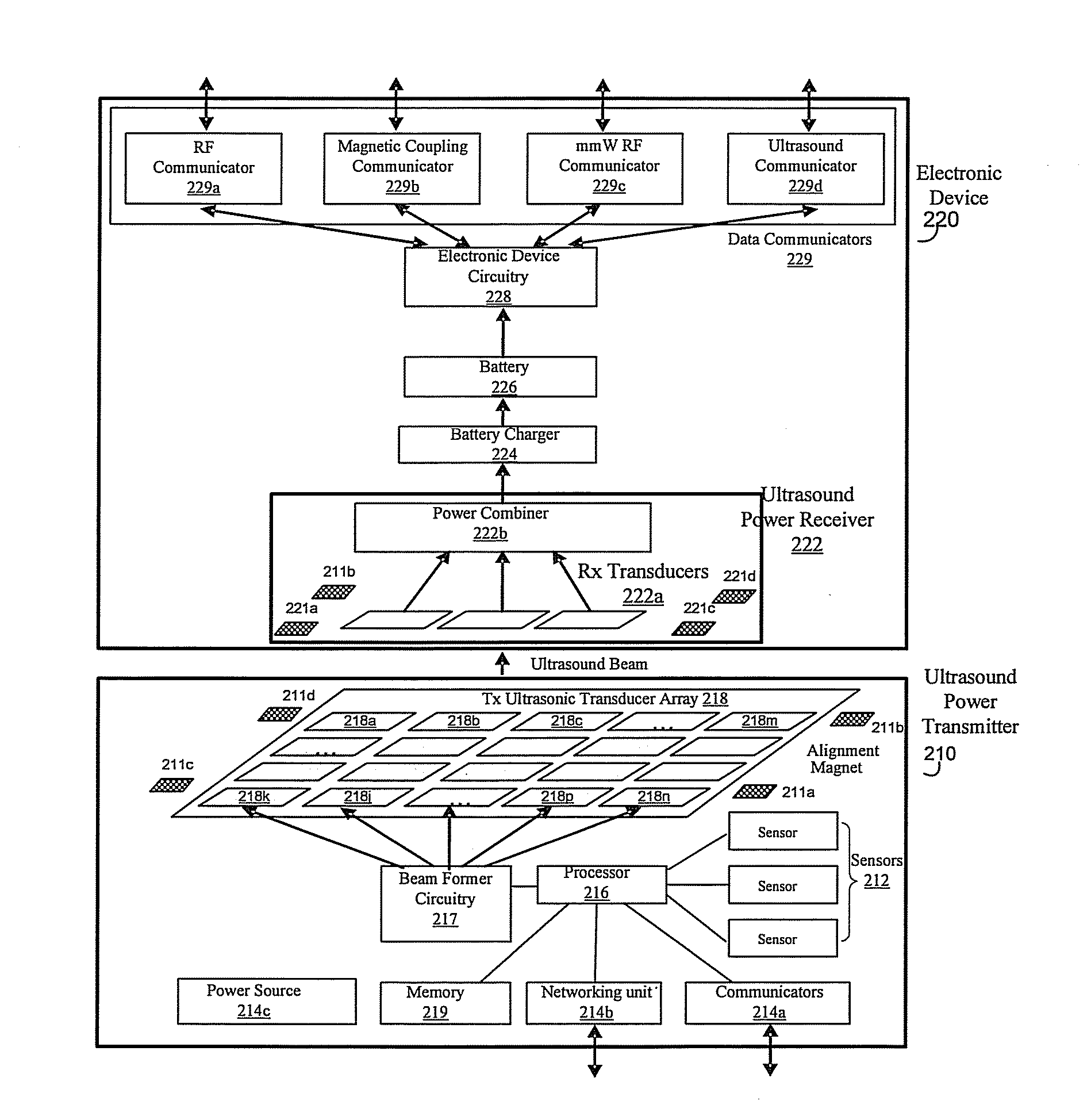
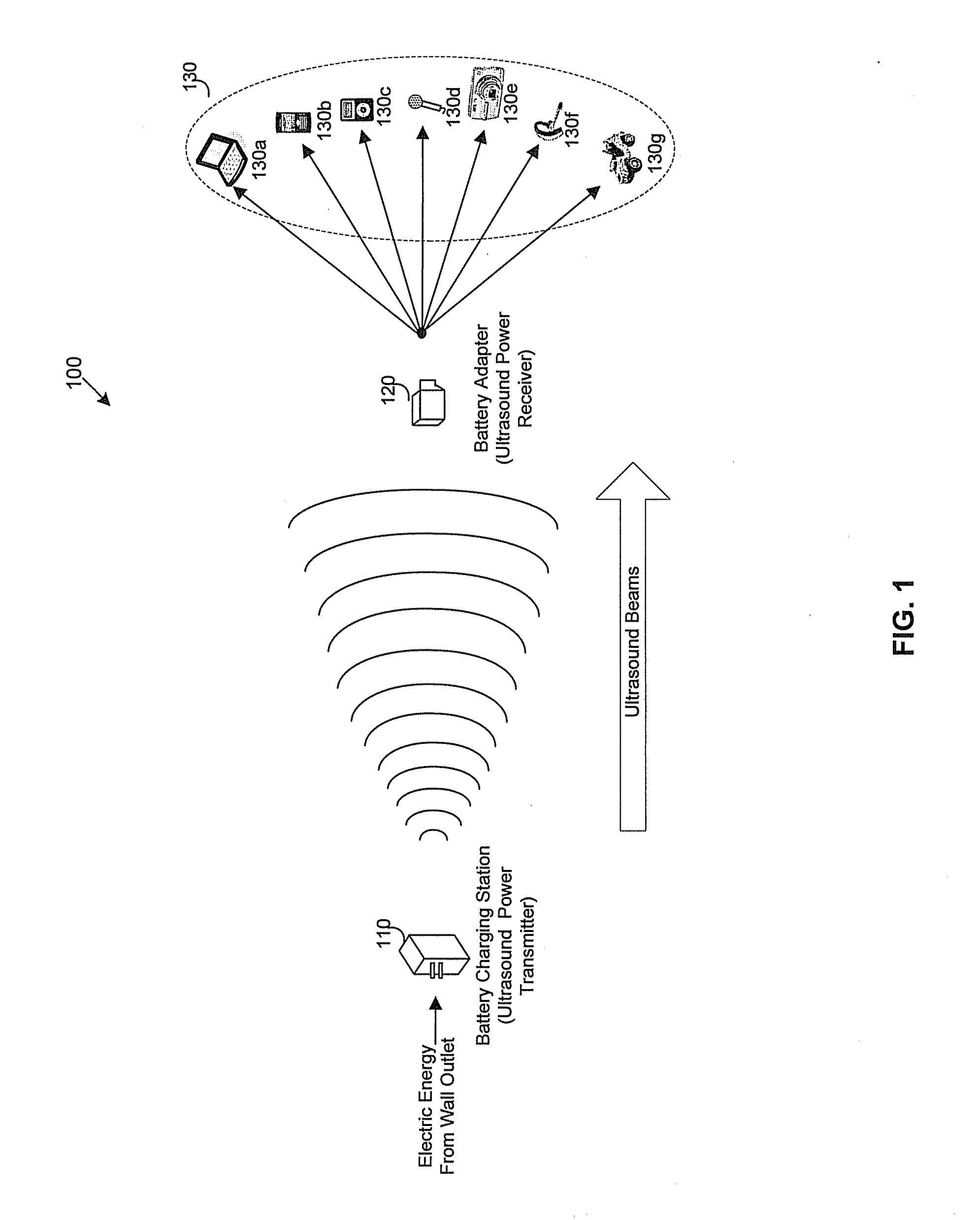
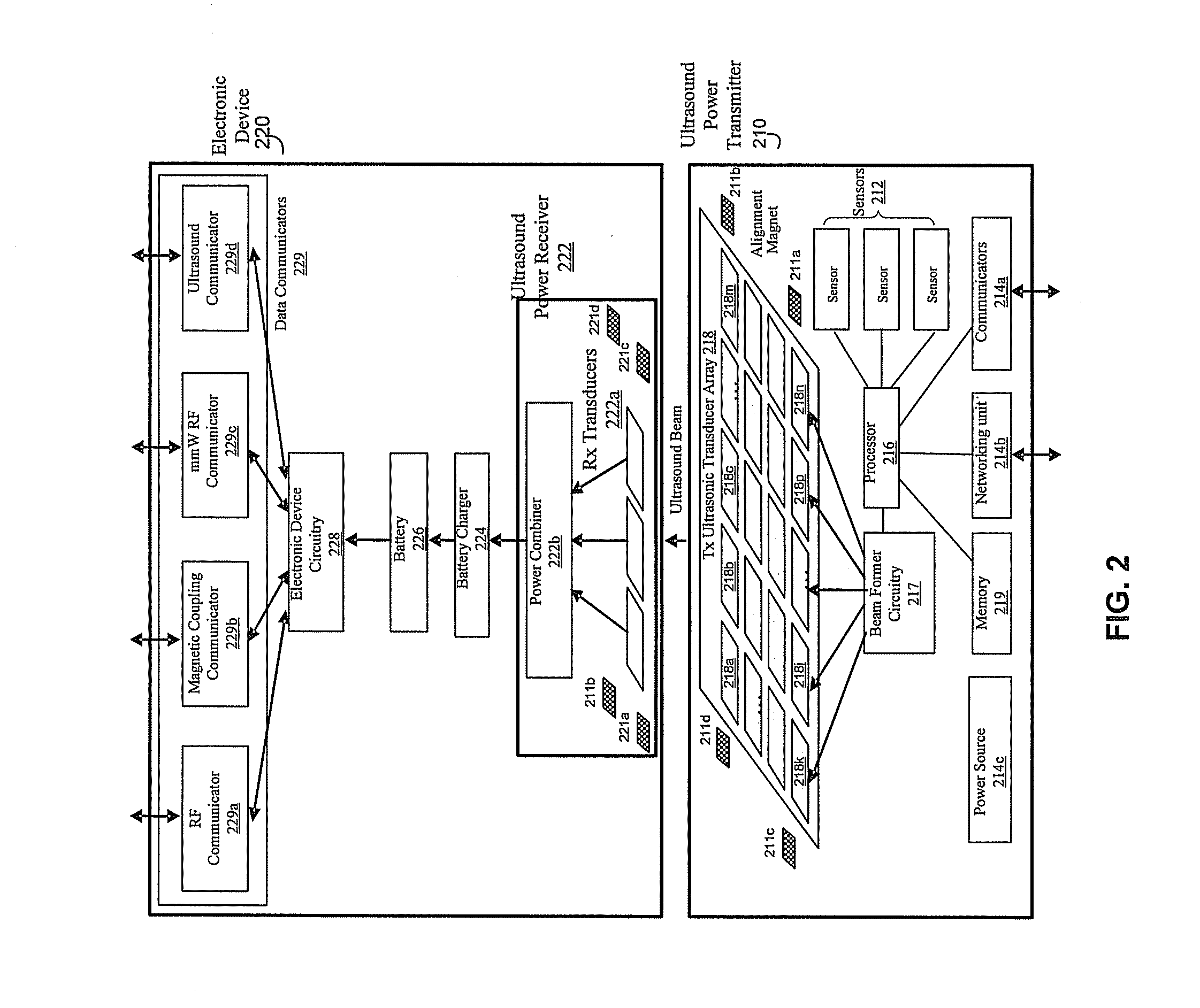
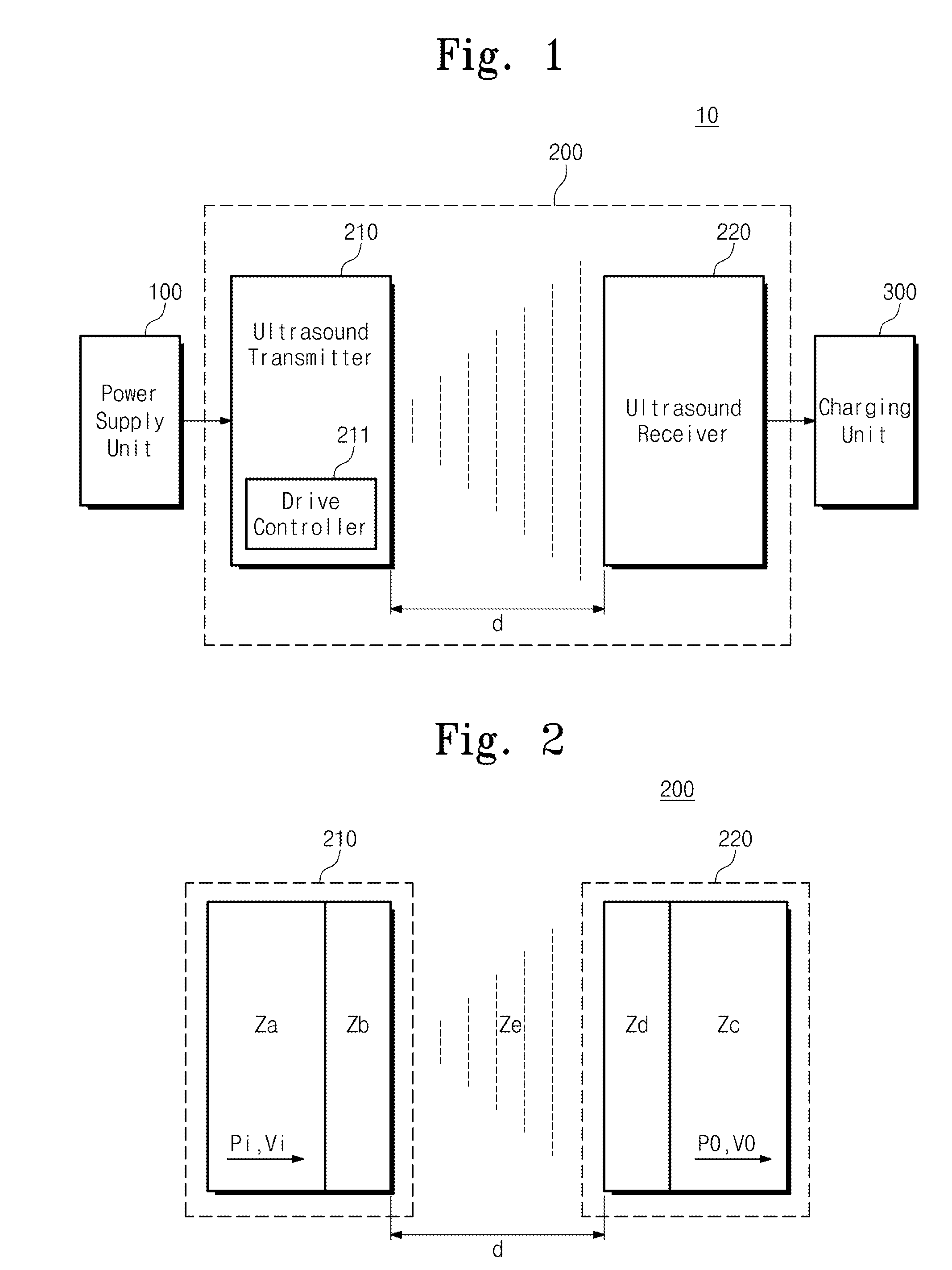
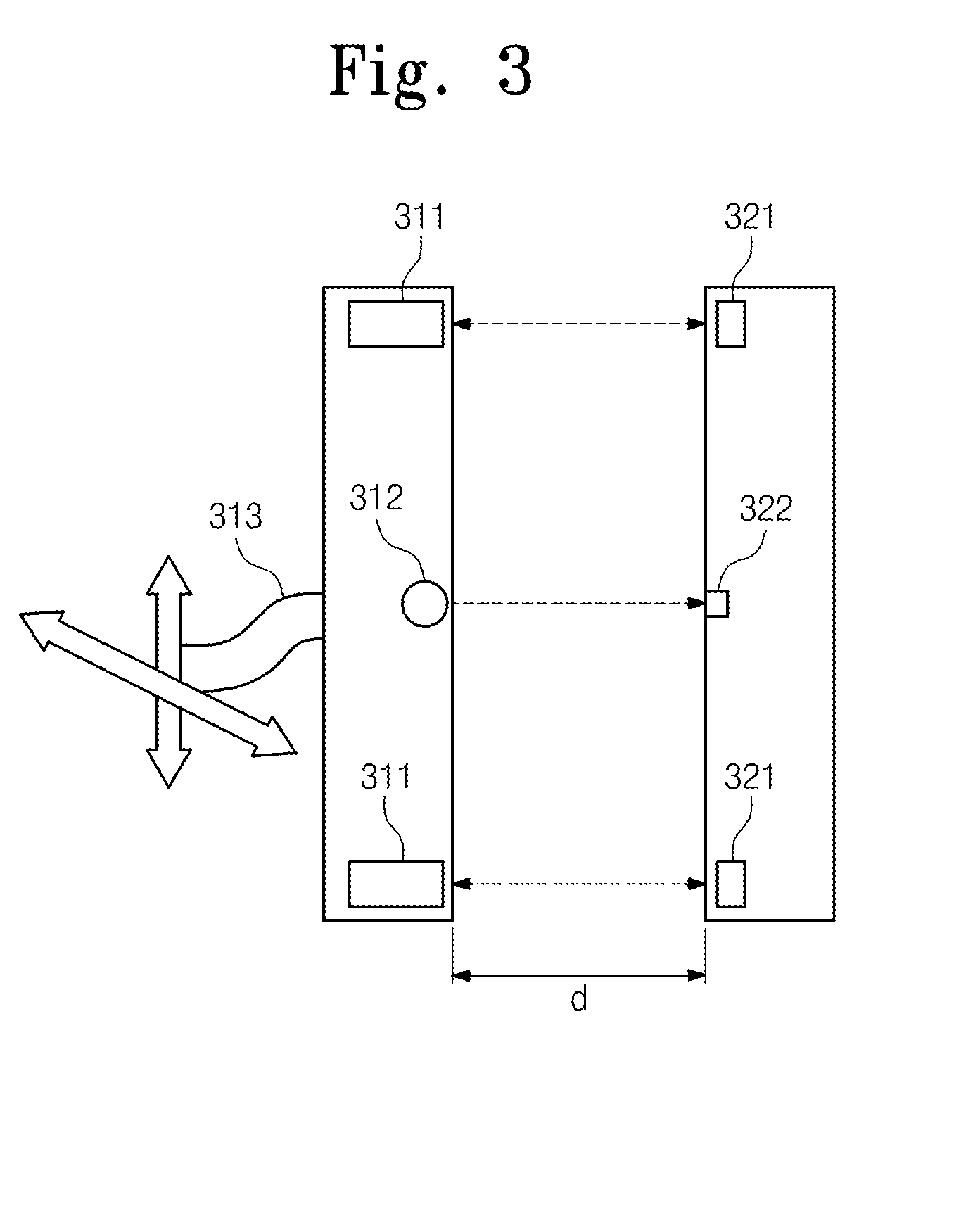
Method and system for wireless battery charging utilizing ultrasonic transducer array based beamforming
InactiveUS20130241468A1Near-field transmissionSonic/ultrasonic/infrasonic transmissionSonificationBattery charge
An ultrasound power transmitter comprising a transmit ultrasonic transducer array has a plurality of transmit ultrasonic transducers. The ultrasound power transmitter activates a set of transmit ultrasonic transducers in close proximity of an electronic device to be arranged to beam ultrasound energy to the electronic device. Alignment magnets of the ultrasound power transmitter are aligned with corresponding alignment magnets of the electronic device to manage the ultrasound beaming. The ultrasound energy may be converted into electric power to charge the battery of the electronic device. Feedbacks may be provided by the electronic device to the ultrasound power transmitter to increase power transmission efficiency. The ultrasound power transmitter may pair the electronic device with other different electronic devices utilizing ultrasonic signals. A spacer with good ultrasound power transmission properties may be located between the ultrasound power transmitter and an ultrasound power receiver of an intended electronic device to enhance power transmission.
Owner:ARRAY IP LLC
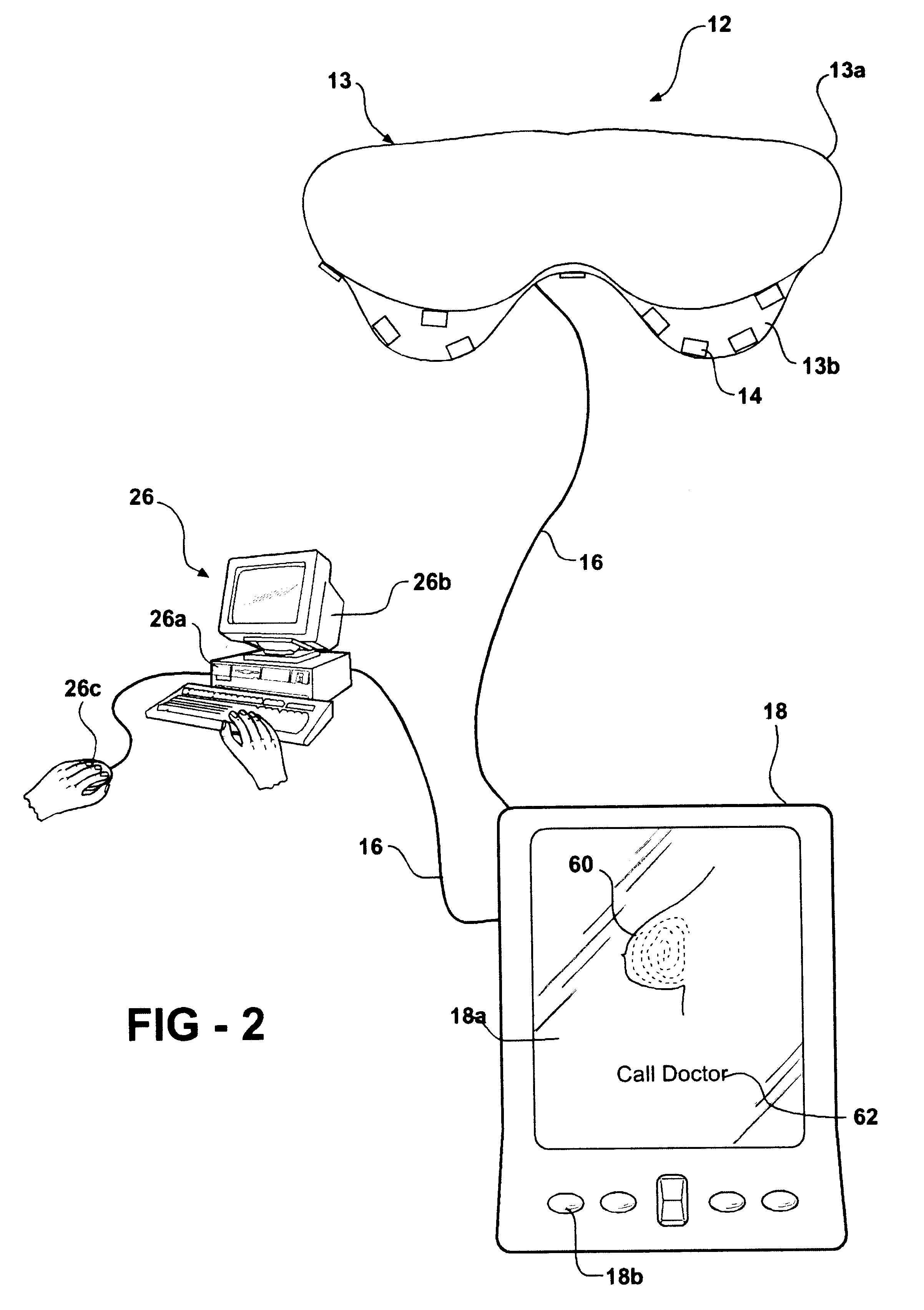
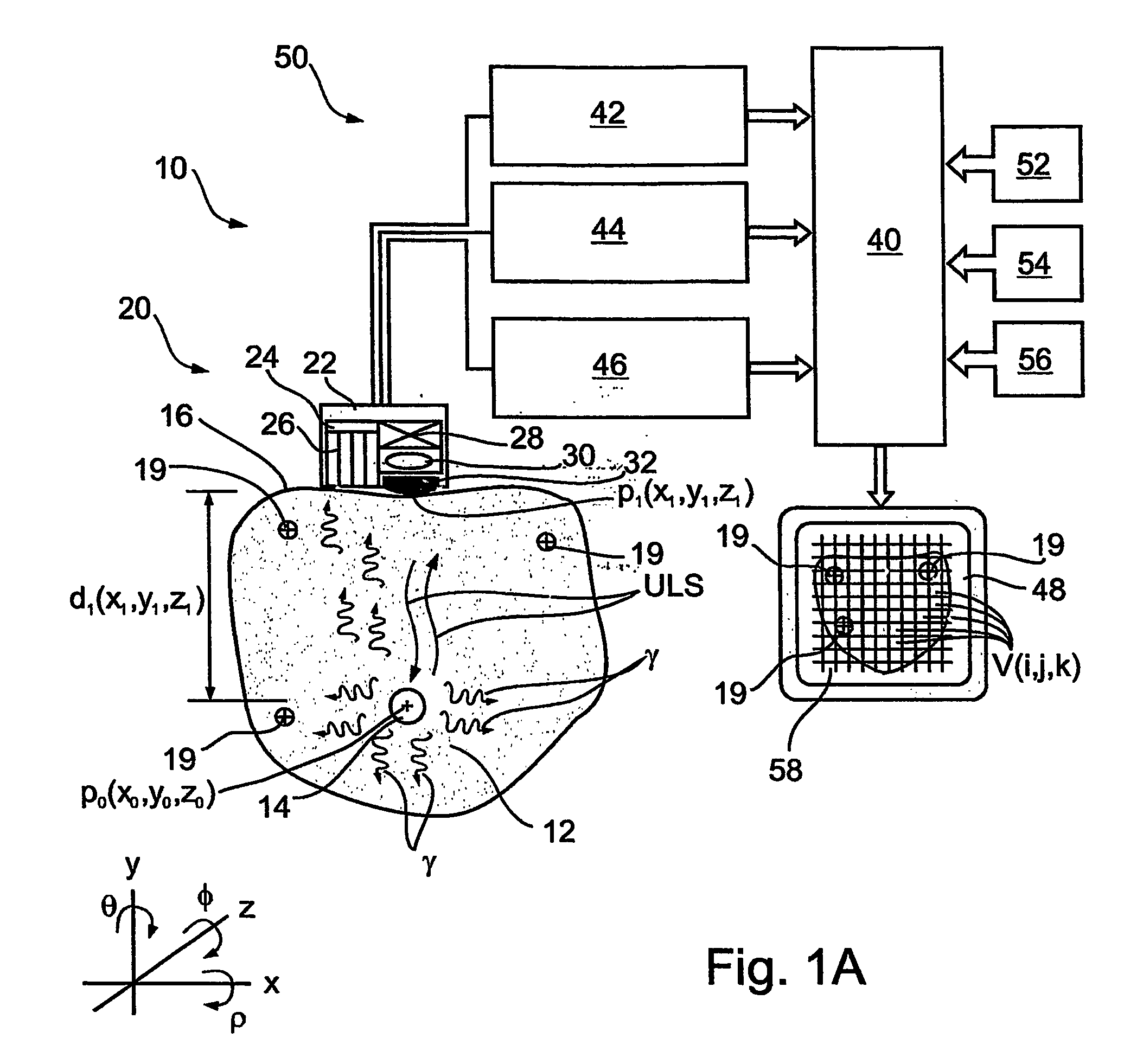
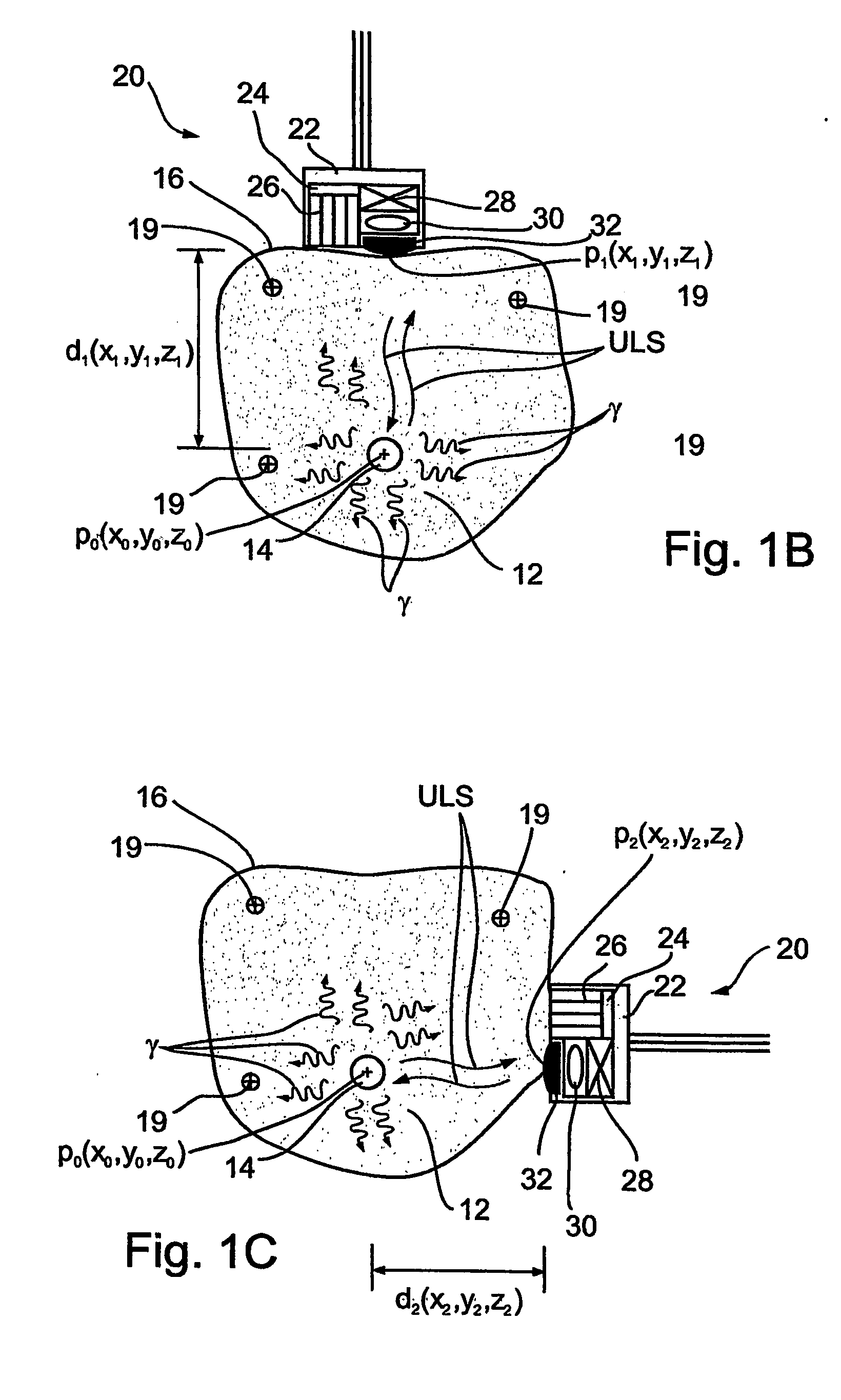
Ultrasonic treatment and imaging of adipose tissue
A system for the destruction of adipose tissue utilizing high intensity focused ultrasound (HIFU) within a patient's body. The system comprises a controller for data storage and the operation and control of a plurality of elements. One elements is a means for mapping a human body to establish three dimensional coordinate position data for existing adipose tissue. The controller is able to identify the plurality of adipose tissue locations on said human body and establish a protocol for the destruction of the adipose tissue. A HIFU transducer assembly having one or more piezoelectric element(s) is used along with at least one sensor wherein the sensor provides feed back information to the controller for the safe operation of the piezoelectric element(s). The sensor is electronically coupled to the controller, and the controller provides essential treatment command information to one or more piezoelectric element(s) based on positioning information obtained from the three dimensional coordinate position data.
Owner:LIPOSONIX
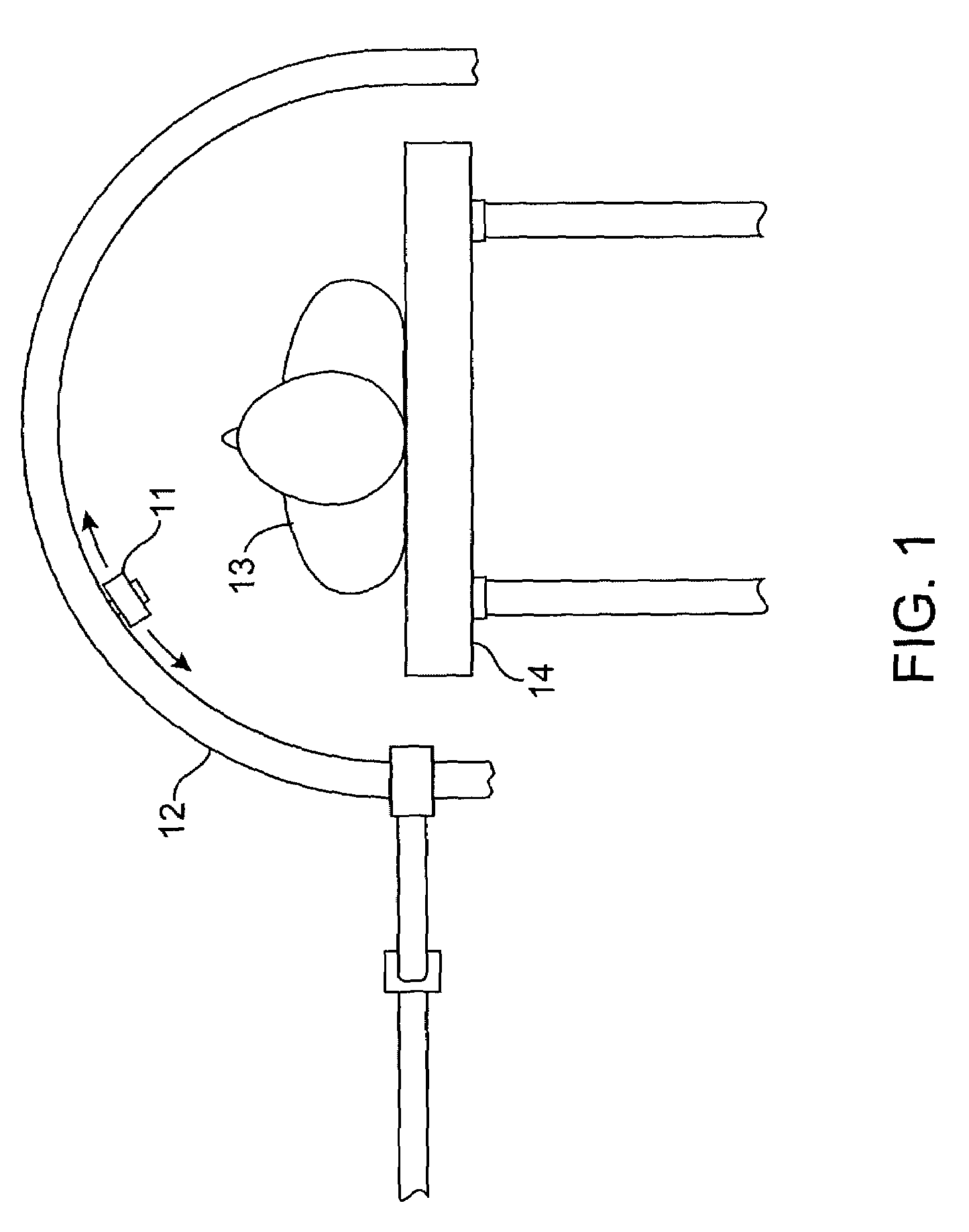
System and method of ultrasonic mammography
Owner:MICROLIFE MEDICAL HOME SOLUTIONS
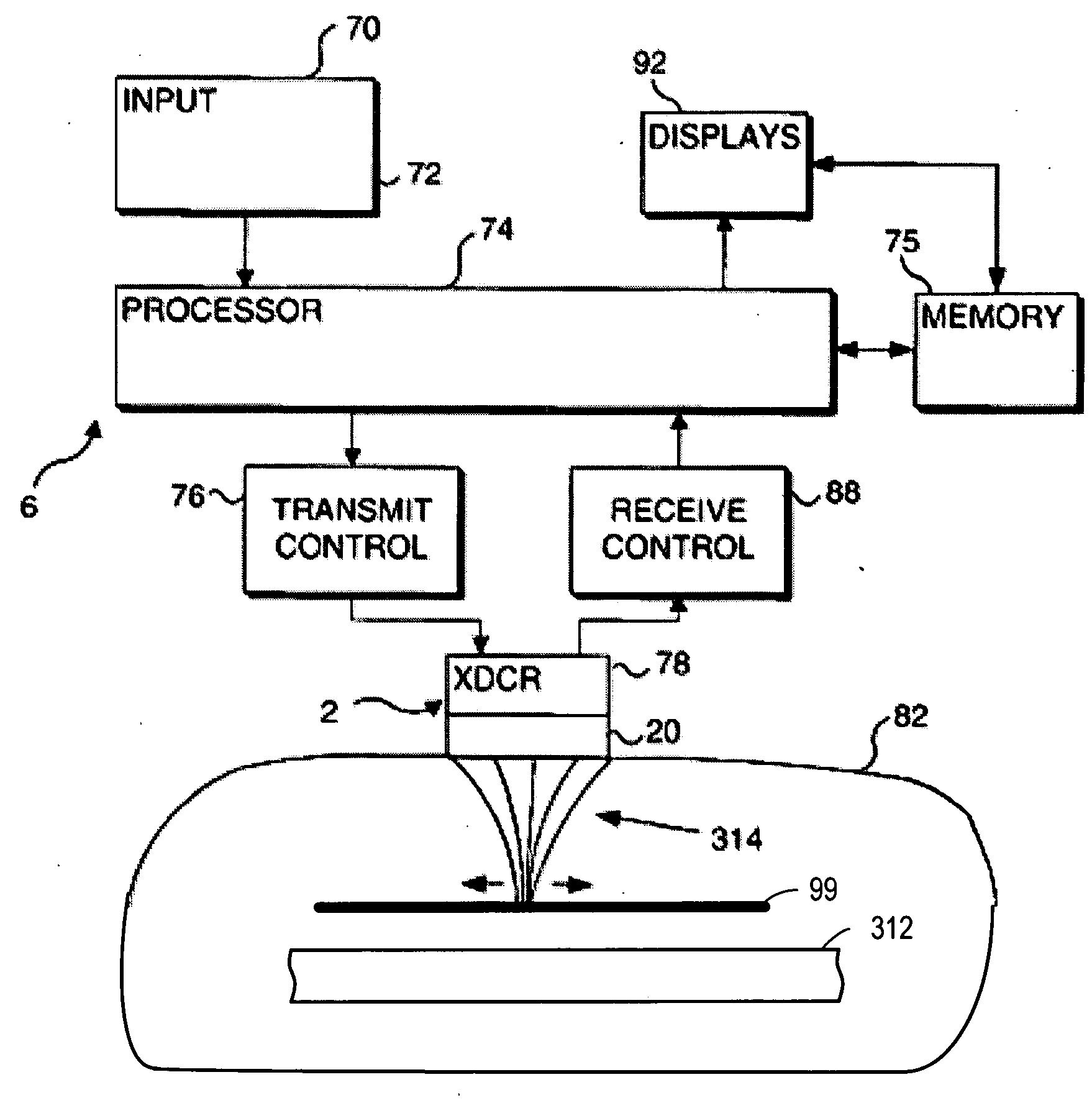
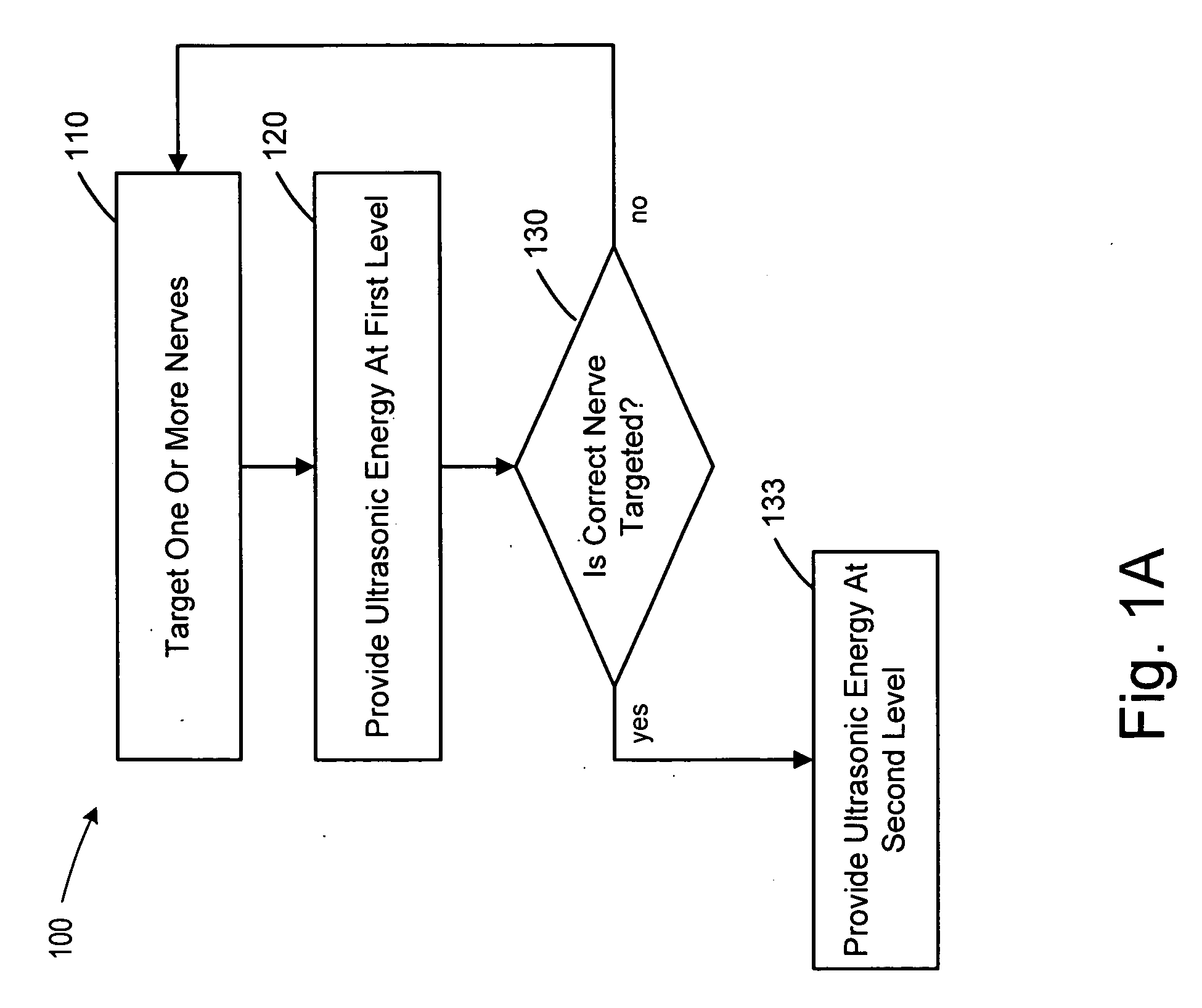
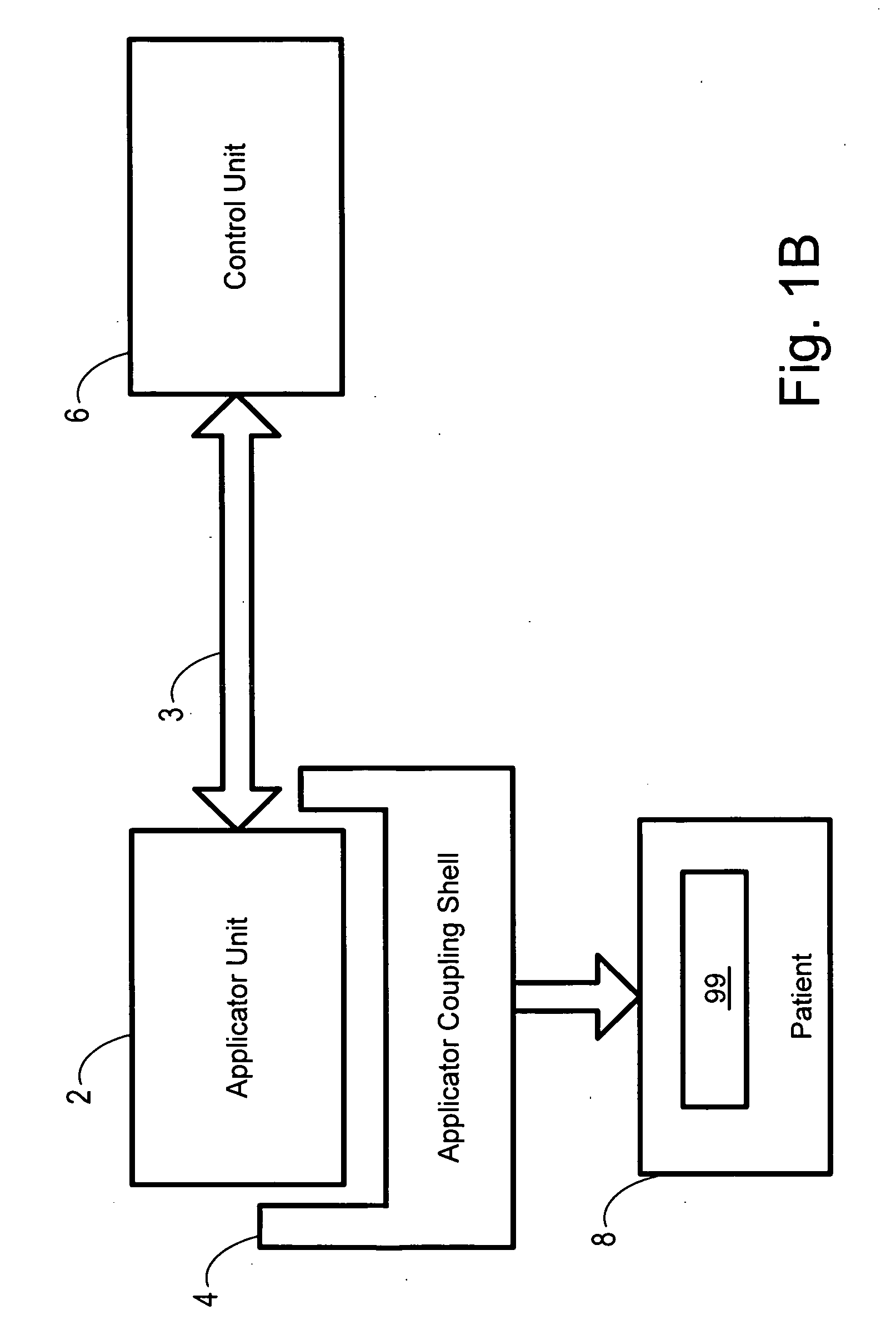
Focused ultrasound for pain reduction
InactiveUS20060184069A1Relieve painUltrasonic/sonic/infrasonic diagnosticsUltrasound therapySonificationTransducer
Methods and devices that provide ultrasonic energy used to cause one or more nerves to become dysfunctional. A nerve to be treated is placed in the focal zone of ultrasonic energy emitted by ultrasound transducer. A first level of ultrasonic energy is provided to the nerve using the ultrasound transducer, the first level sufficient to stimulate the nerve. A verification is made that the desired nerve is being stimulated by the first level of ultrasonic energy. For example, the patient may be asked to confirm that the ultrasonically stimulated nerve corresponds to the pain that is affecting the patient. Subsequent to verifying the stimulated nerve is the nerve desired for the reduction of pain, a second level of ultrasonic energy is delivered to the nerve using the ultrasound transducer, the second level of ultrasonic energy sufficient to cause nerve dysfunction.
Owner:VAITEKUNAS JEFFREY J
Ultrasound guided high intensity focused ultrasound treatment of nerves
InactiveUS7510536B2Relieve painEasy procedureUltrasound therapyBlood flow measurement devicesSonificationHigh doses
A method for using high intensity focused ultrasound (HIFU) to treat neurological structures to achieve a desired therapeutic affect. Depending on the dosage of HIFU applied, it can have a reversible or irreversible effect on neural structures. For example, a relatively high dose of HIFU can be used to permanently block nerve function, to provide a non-invasive alternative to severing a nerve to treat severe spasticity. Relatively lower doses of HIFU can be used to reversible a block nerve function, to alleviate pain, to achieve an anesthetic effect, or to achieve a cosmetic effect. Where sensory nerves are not necessary for voluntary function, but are involved in pain associated with tumors or bone cancer, HIFU can be used to non-invasively destroy such sensory nerves to alleviate pain without drugs. Preferably, ultrasound imaging synchronized to the HIFU therapy is used to provide real-time ultrasound image guided HIFU therapy of neural structures.
Owner:UNIV OF WASHINGTON
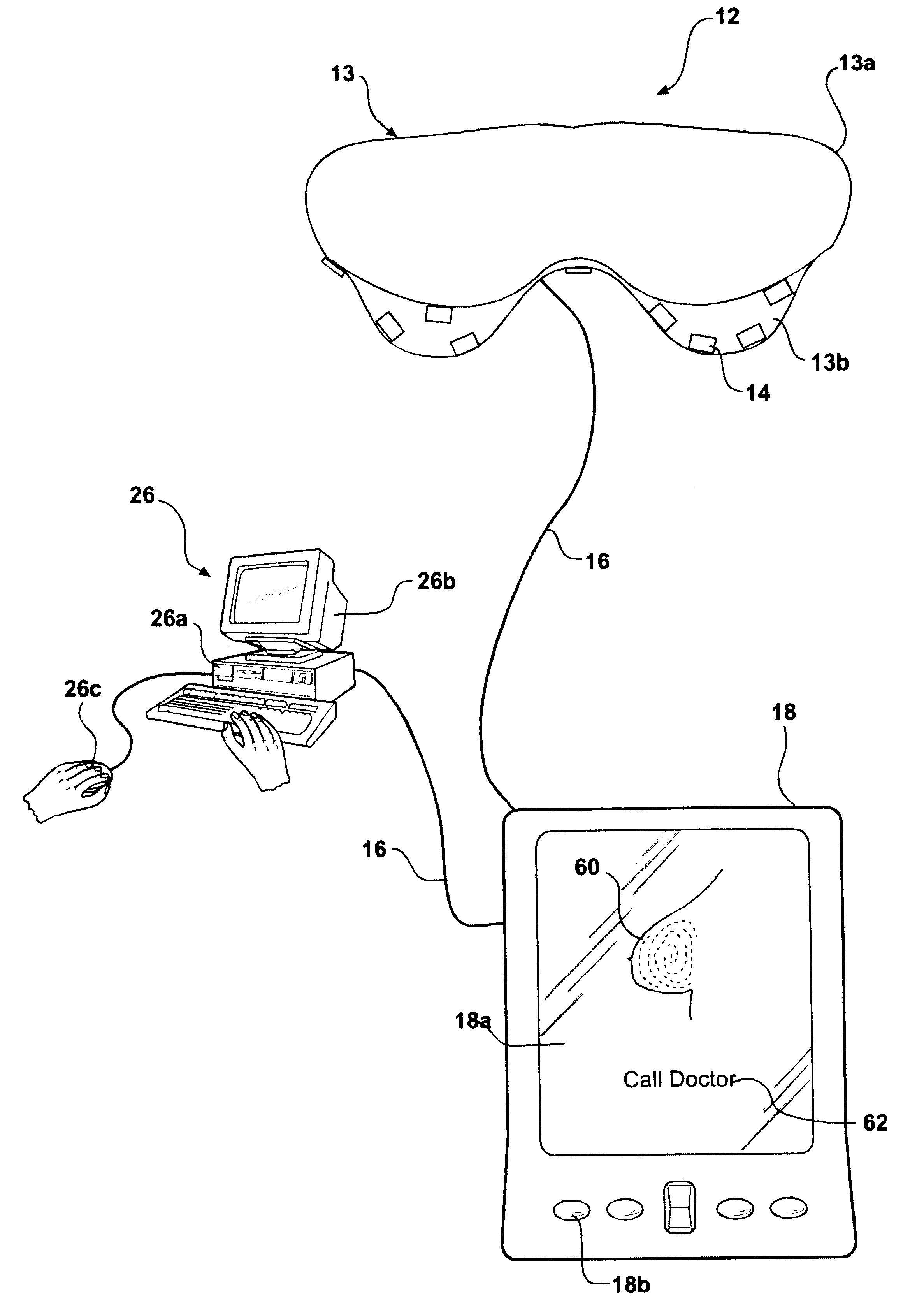
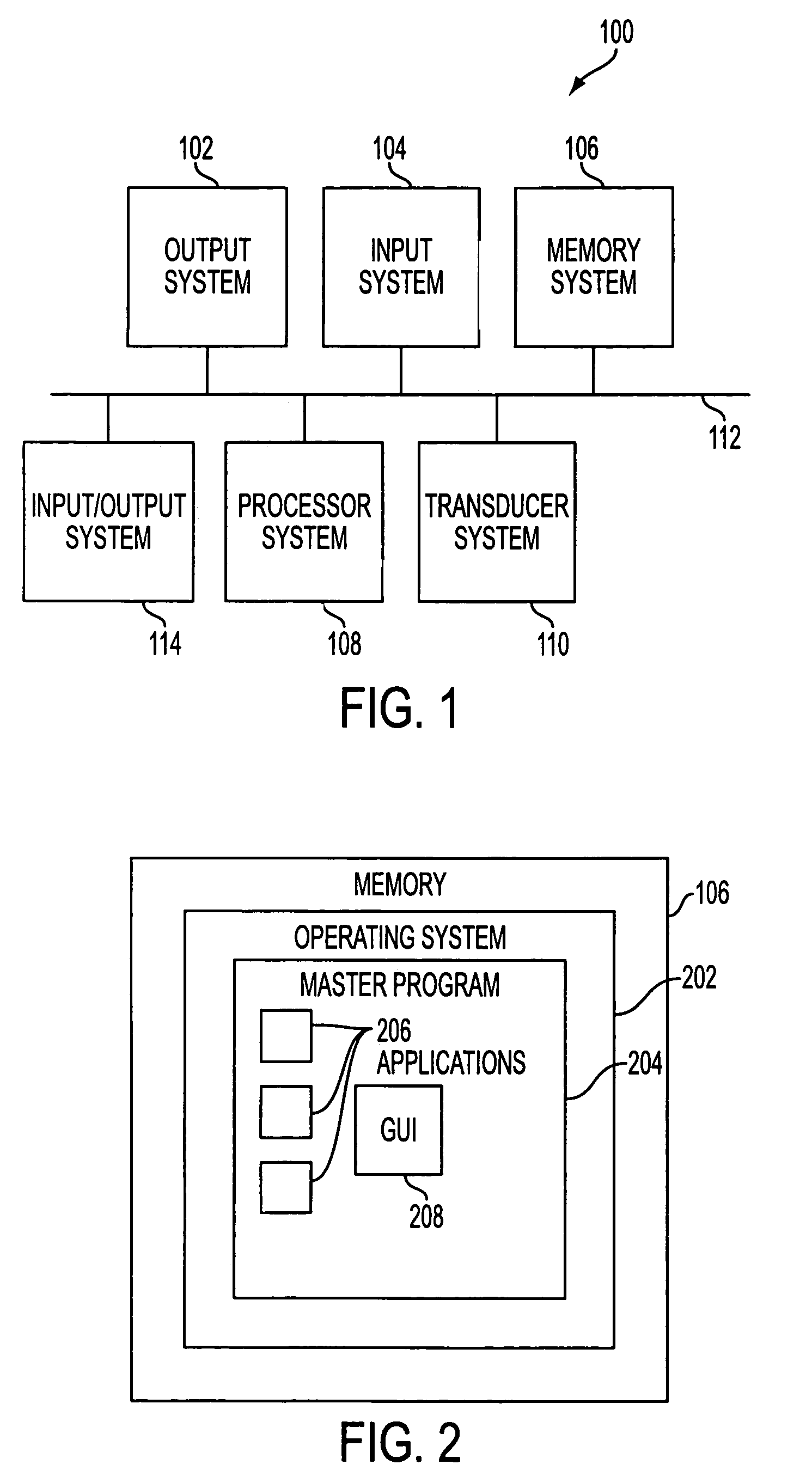
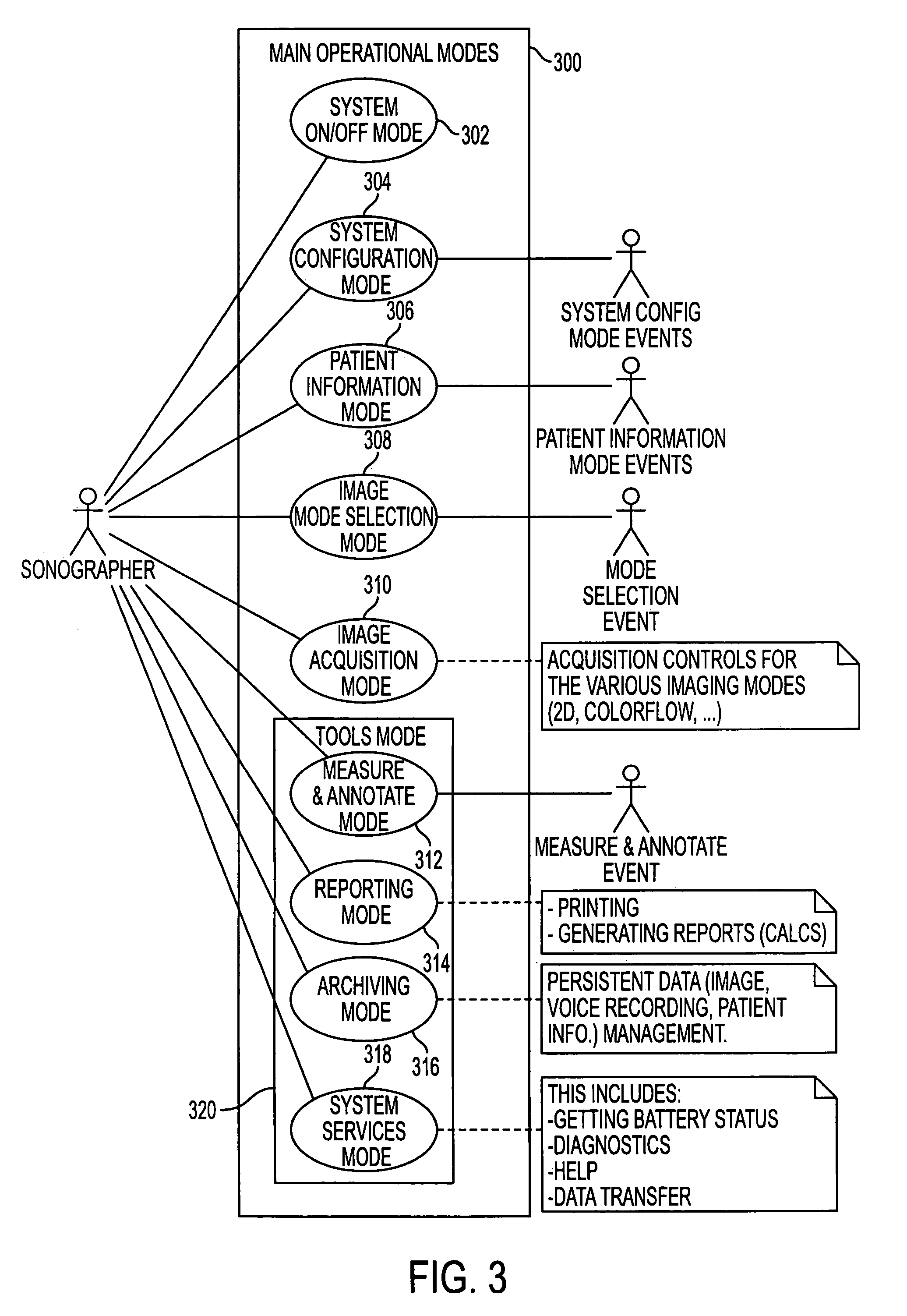
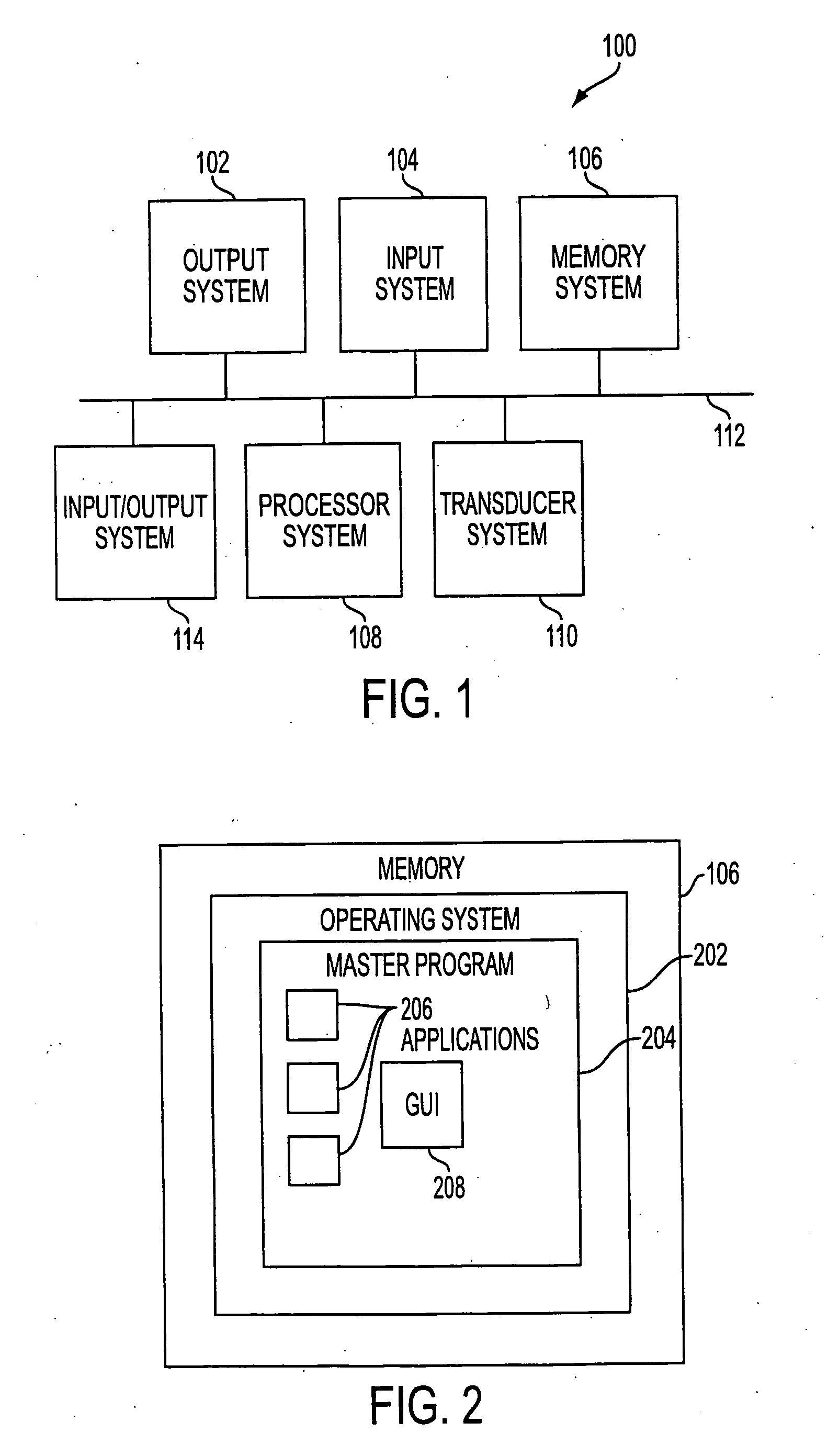
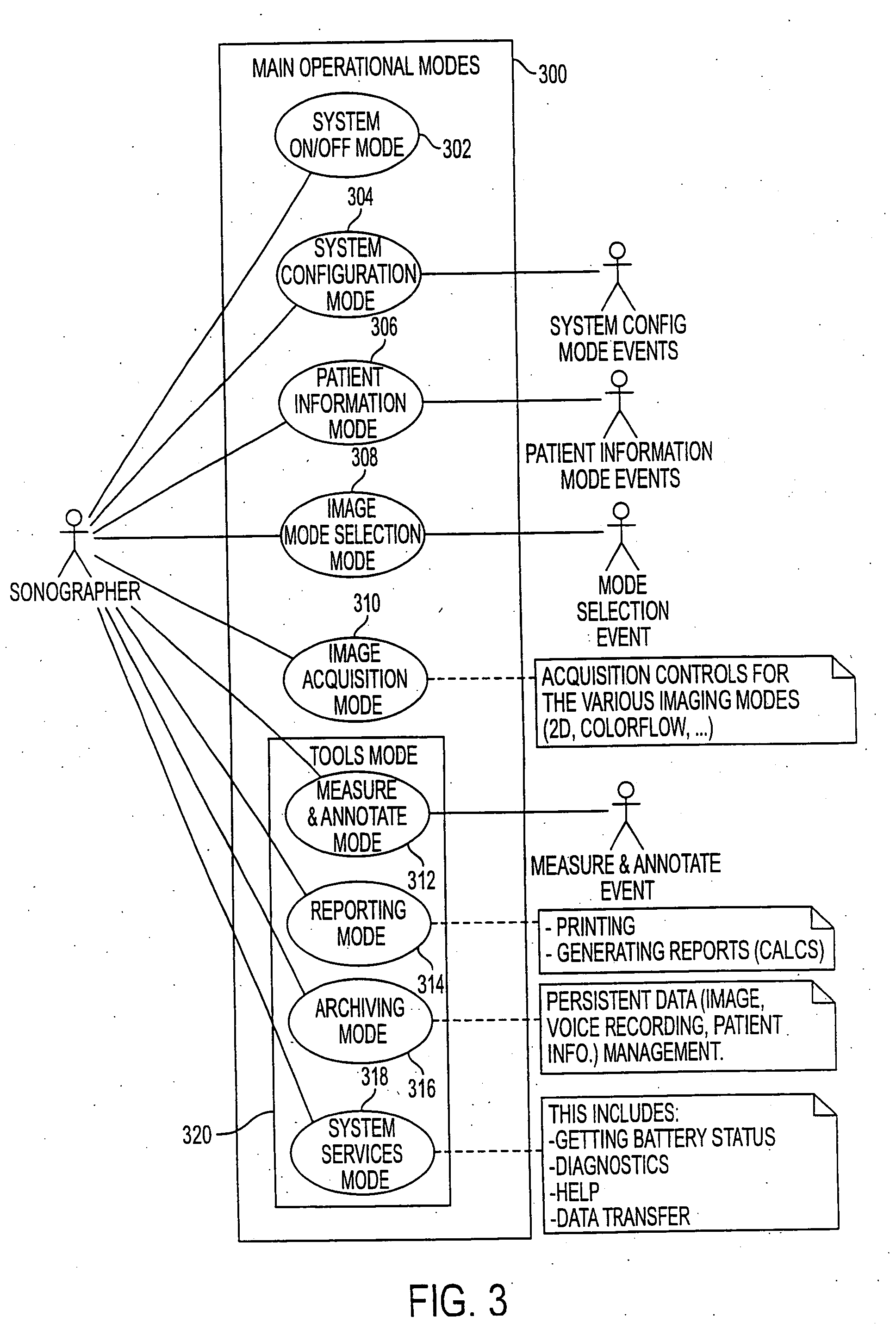
User interface for handheld imaging devices
InactiveUS7022075B2Minimize timeEasy to distinguishLocal control/monitoringBlood flow measurement devicesData displayUltrasonography
A Graphical User Interface (GUI) for an ultrasound system. The ultrasound system has operational modes and the GUI has corresponding icons, tabs, and menu items image and information fields. The User Interface (UI) provides several types of graphical elements with intelligent behavior, such as being context sensitive and adaptive, called active objects, for example, tabs, menus, icons, windows of user interaction and data display and an alphanumeric keyboard. In addition the UI may also be voice activated. The UI further provides for a touchscreen for direct selection of displayed active objects. In an embodiment, the UI is for a medical ultrasound handheld imaging instrument. The UI provides a limited set of hard and soft keys with adaptive functionality that can be used with only one hand and potentially with only one thumb.
Owner:SHENZHEN MINDRAY BIO MEDICAL ELECTRONICS CO LTD
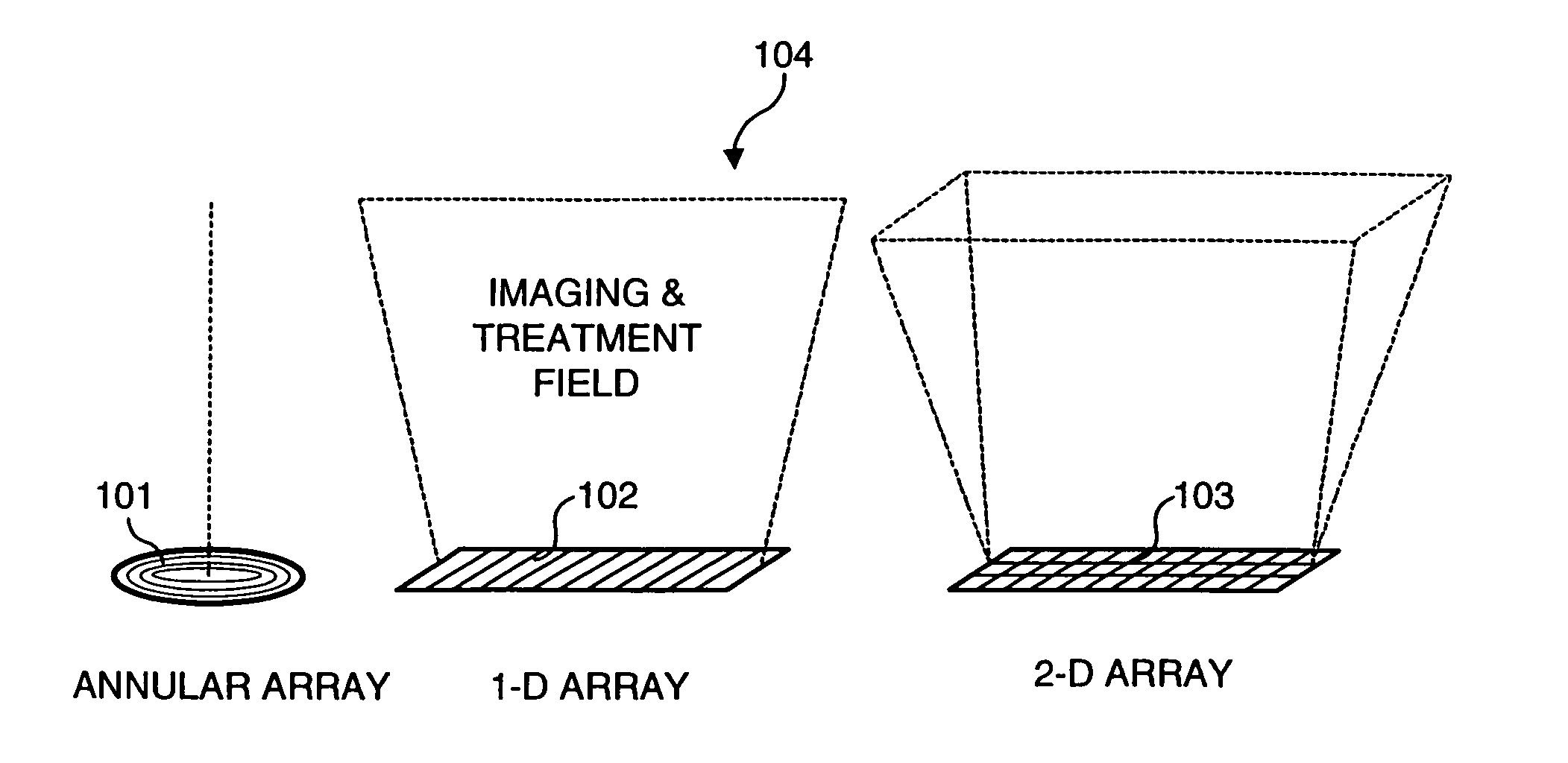
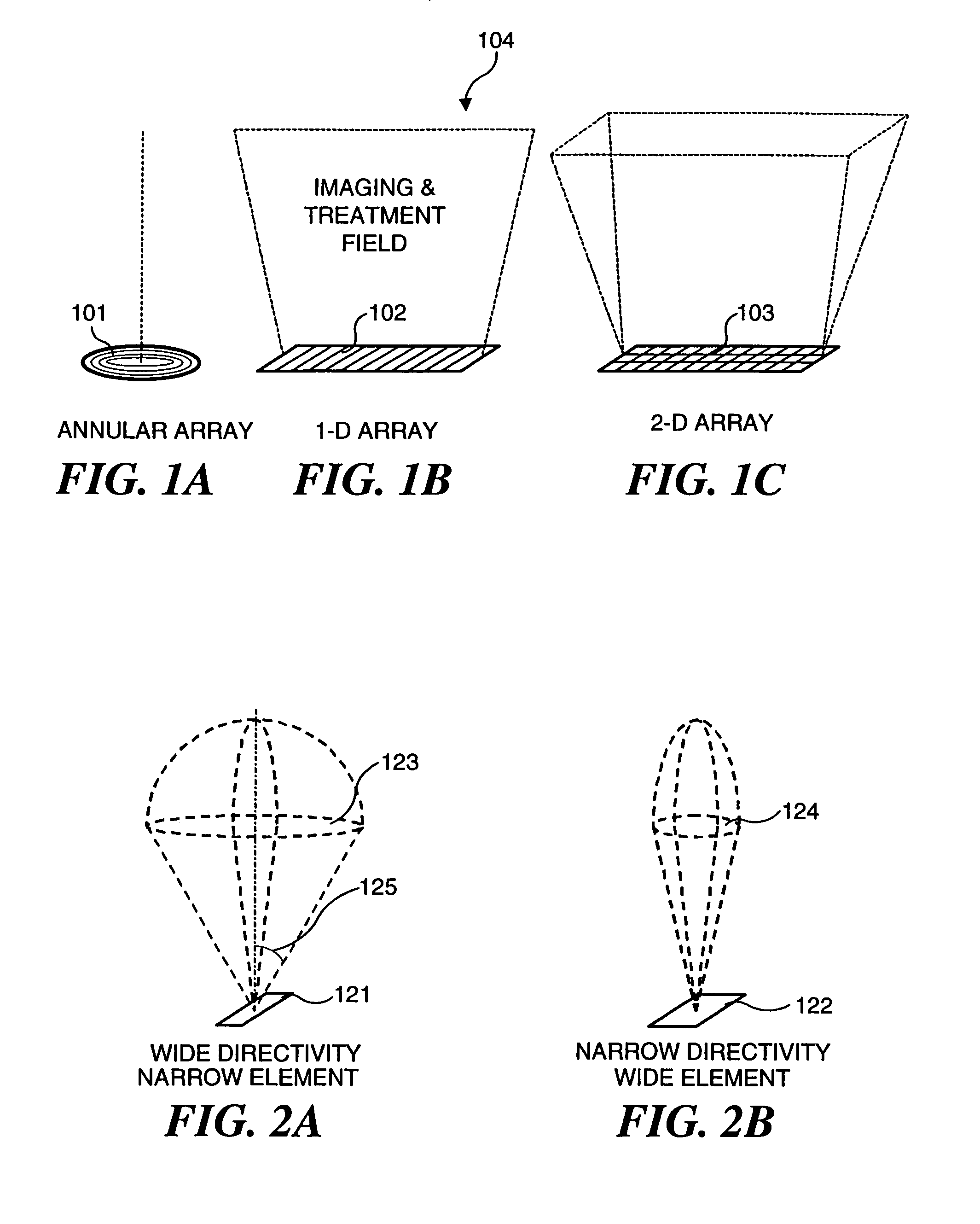
Ultrasound transducers for imaging and therapy
InactiveUS7063666B2Reduce in quantityReducing cross-talk and heatingUltrasonic/sonic/infrasonic diagnosticsUltrasound therapyElectrical resistance and conductanceSonification
Owner:OTSUKA MEDICAL DEVICES
Component ultrasound transducer
InactiveUS20050154314A1Eliminate dangerUltrasonic/sonic/infrasonic diagnosticsUltrasound therapySonificationUltrasonic sensor
An ultrasound transducer having multiple focal zones is described. In one embodiment there is an ultrasound transducer manufactured as a single piece but having two or more focal zones. In a second embodiment there is a transducer assembly combining a high frequency and low frequency transducer. In a third embodiment there is an interchangeable assembly allowing for different ultrasound transducers to be used based on procedural needs. Variations of each embodiment are also disclosed.
Owner:LIPOSONIX
Focused ultrasound system with adaptive anatomical aperture shaping
ActiveUS20060058671A1Reducing ultrasound energyUltrasound therapyBlood flow measurement devicesSonificationTransducer
A method of treating tissue within a body includes directing an ultrasound transducer having a plurality of transducer elements towards target body tissue, and delivering ultrasound energy towards the target tissue from the transducer elements such that an energy intensity at the target tissue is at or above a prescribed treatment level, while an energy intensity at tissue to be protected in the ultrasound energy path of the transducer elements is at or below a prescribed safety level.
Owner:INSIGHTEC
Pulsed cavitational ultrasound therapy
ActiveUS20080319356A1Improve drug deliveryEasy to transportOrgan movement/changes detectionSurgerySonificationMicrobubbles
Owner:RGT UNIV OF MICHIGAN
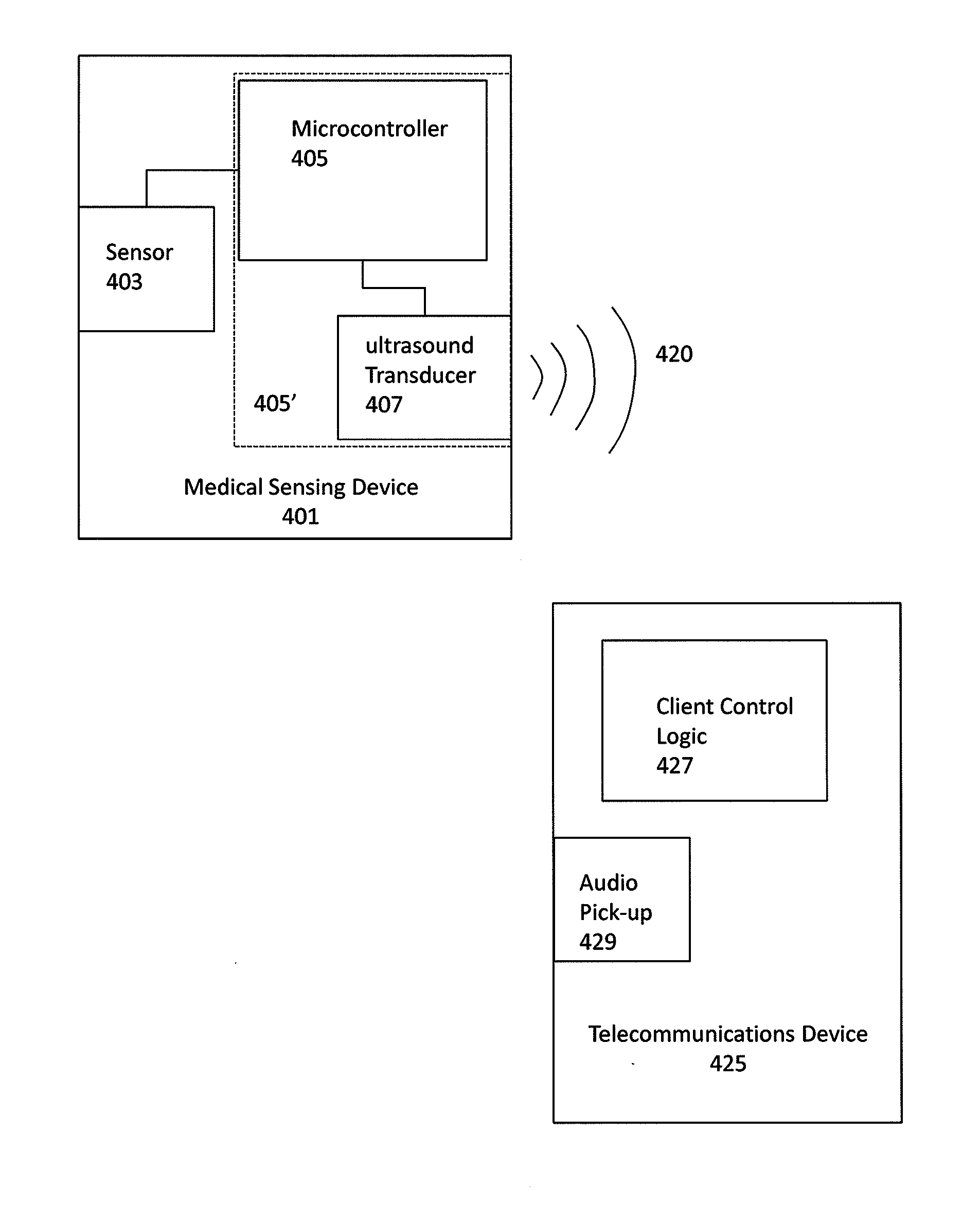
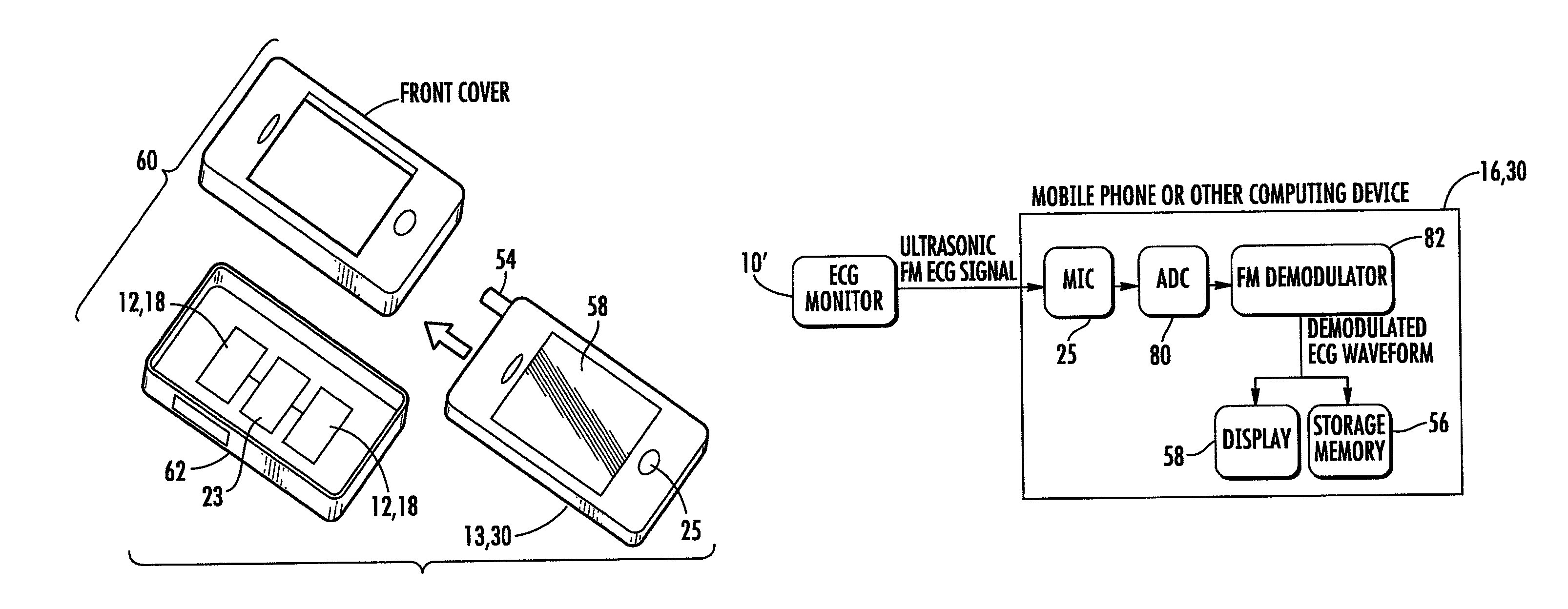
Ultrasonic digital communication of biological parameters
Medical sensing devices and systems that transmit digital data from a first device via an ultrasonic digital modem to a receiver such as a smartphone. Methods of transmitting digital biological data by ultrasound are also described.
Owner:ALIVECOR
Medical ultrasound system and handpiece and methods for making and tuning
InactiveUS20070232926A1Small sizeIncrease displacementUltrasonic/sonic/infrasonic diagnosticsSurgeryUltrasonographyUltrasonic sensor
Several embodiments of medical ultrasound handpieces are described each including a medical ultrasound transducer assembly. An embodiment of a medical ultrasound system is described, wherein the medical ultrasound system includes a medical ultrasound handpiece having a medical ultrasound transducer assembly and includes an ultrasonically-vibratable medical-treatment instrument which is attachable to a distal end of the transducer assembly. An embodiment of a medical ultrasound system is described, wherein the medical ultrasound system has a handpiece including a medical ultrasound transducer assembly and including a housing or housing component surrounding the transducer assembly. A method for tuning a medical ultrasound handpiece includes machining at least a distal non-threaded portion of an instrument-attachment stud of the transducer assembly to match a measured fundamental frequency to a desired fundamental frequency to within a predetermined limit. A method for making a medical ultrasound transducer assembly determines acceptable gains for gain stages of the transducer assembly.
Owner:CILAG GMBH INT
Medical ultrasound system and handpiece and methods for making and tuning
InactiveUS20070232928A1Small sizeIncrease displacementUltrasonic/sonic/infrasonic diagnosticsSurgeryUltrasonographyUltrasonic sensor
Several embodiments of medical ultrasound handpieces are described each including a medical ultrasound transducer assembly. An embodiment of a medical ultrasound system is described, wherein the medical ultrasound system includes a medical ultrasound handpiece having a medical ultrasound transducer assembly and includes an ultrasonically-vibratable medical-treatment instrument which is attachable to a distal end of the transducer assembly. An embodiment of a medical ultrasound system is described, wherein the medical ultrasound system has a handpiece including a medical ultrasound transducer assembly and including a housing or housing component surrounding the transducer assembly. A method for tuning a medical ultrasound handpiece includes machining at least a distal non-threaded portion of an instrument-attachment stud of the transducer assembly to match a measured fundamental frequency to a desired fundamental frequency to within a predetermined limit. A method for making a medical ultrasound transducer assembly determines acceptable gains for gain stages of the transducer assembly.
Owner:CILAG GMBH INT +1
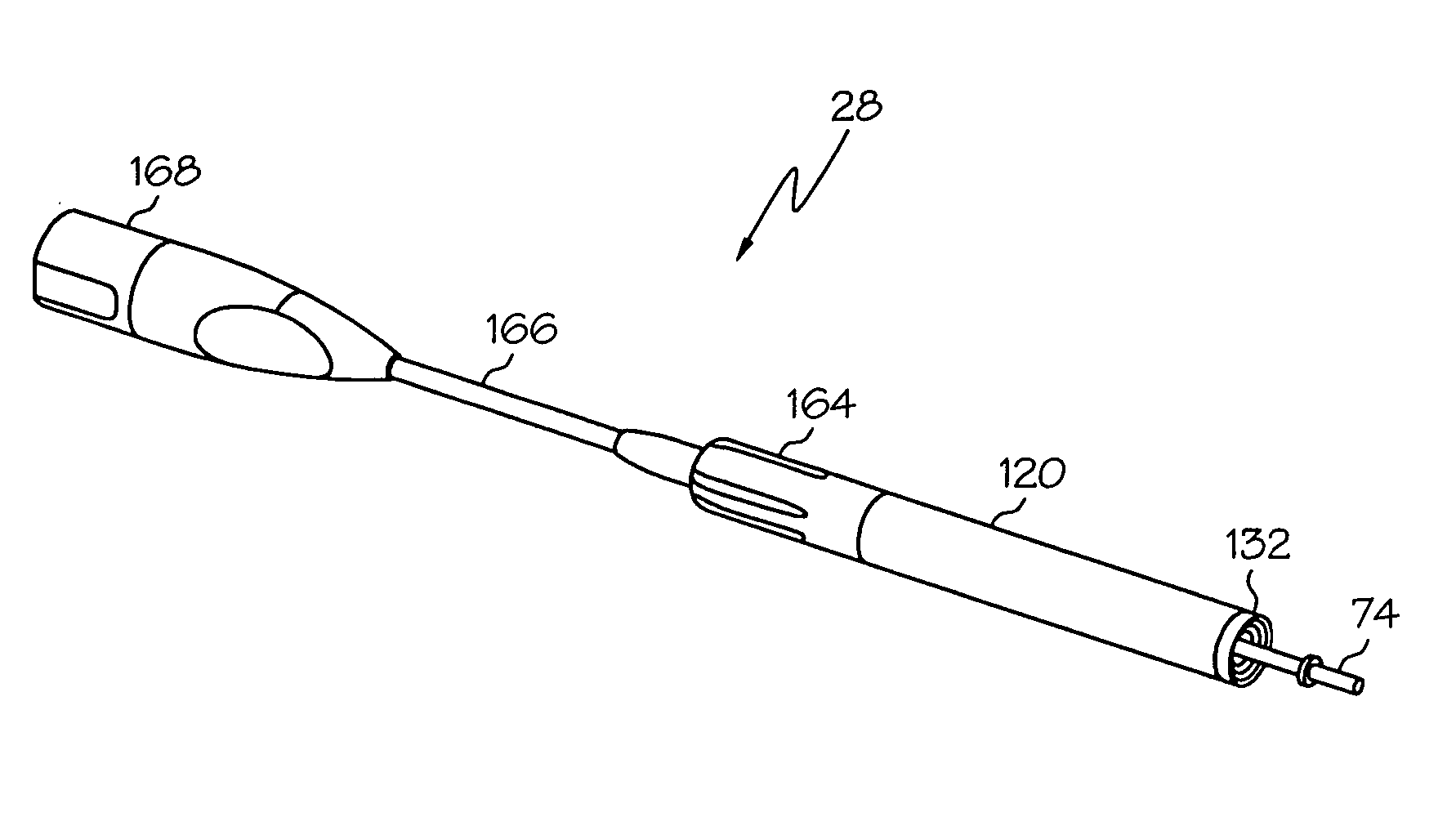
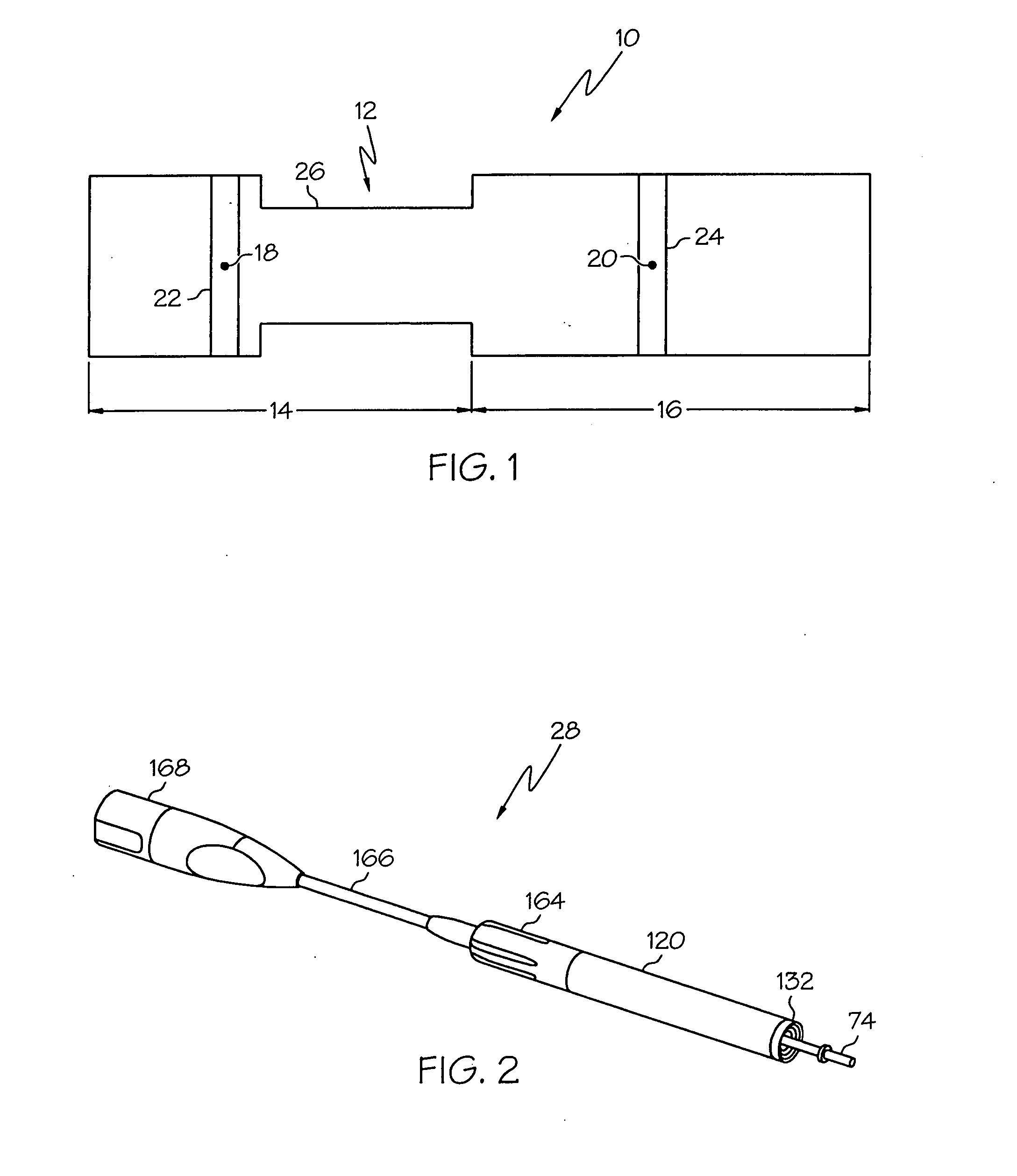
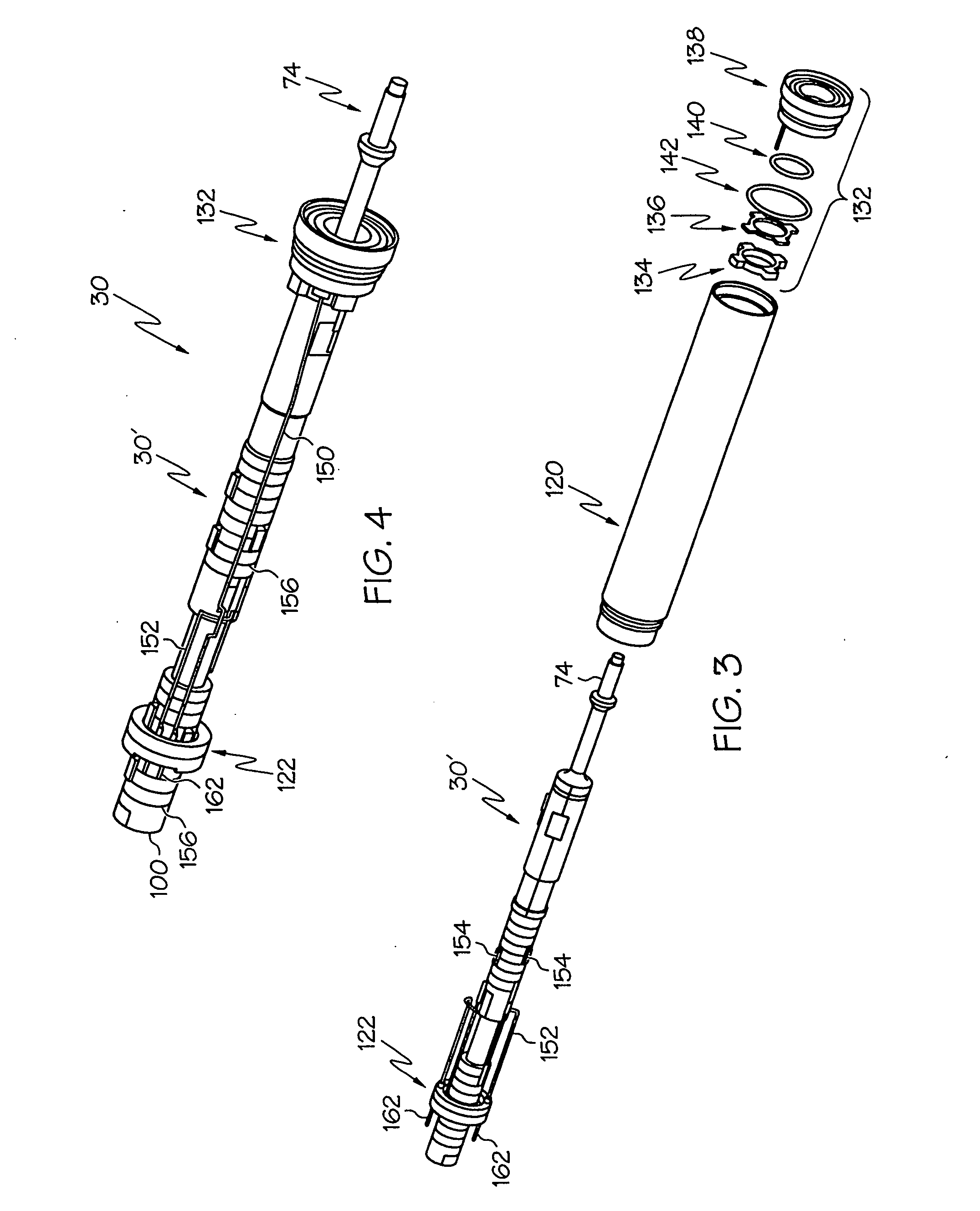
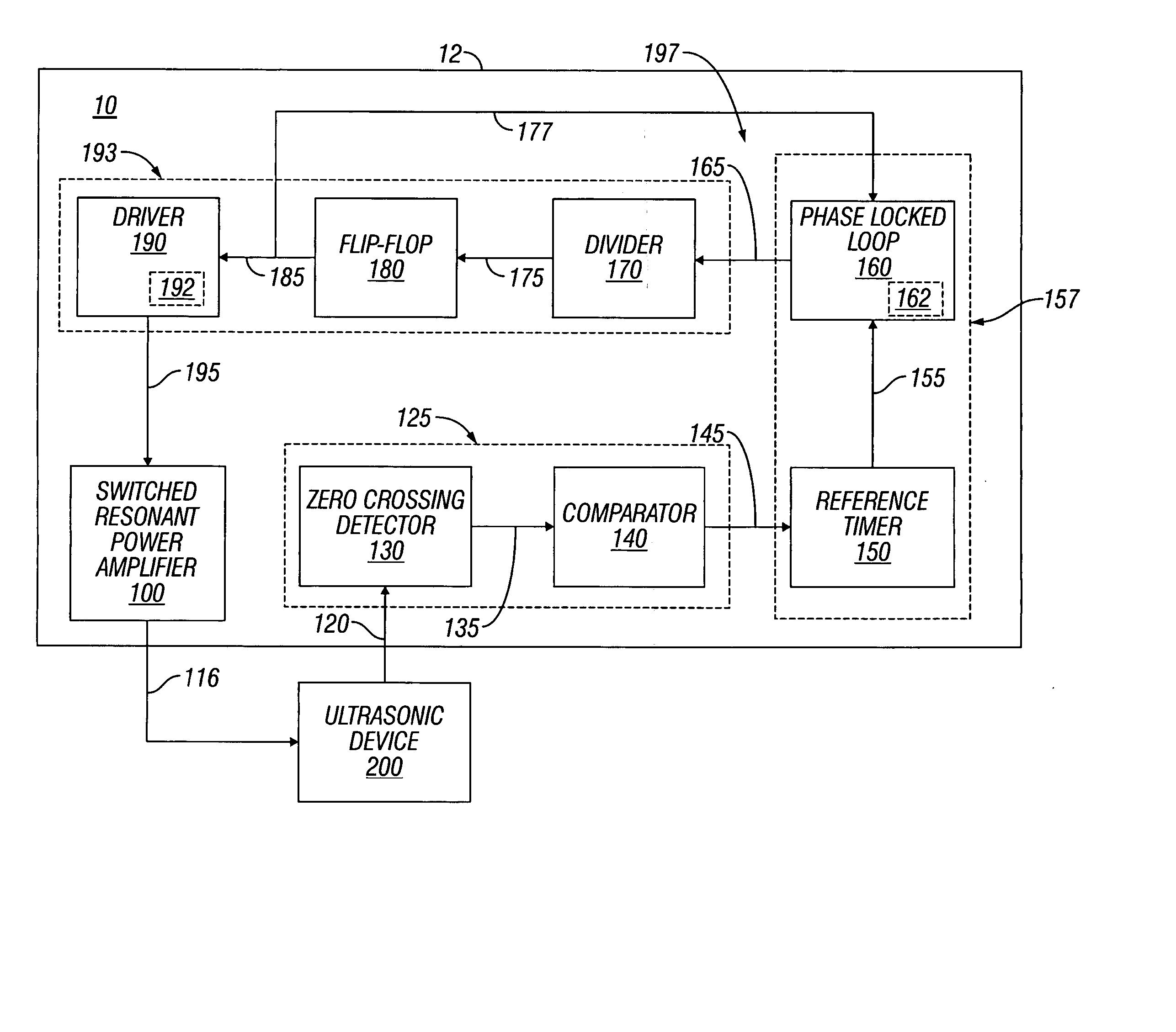
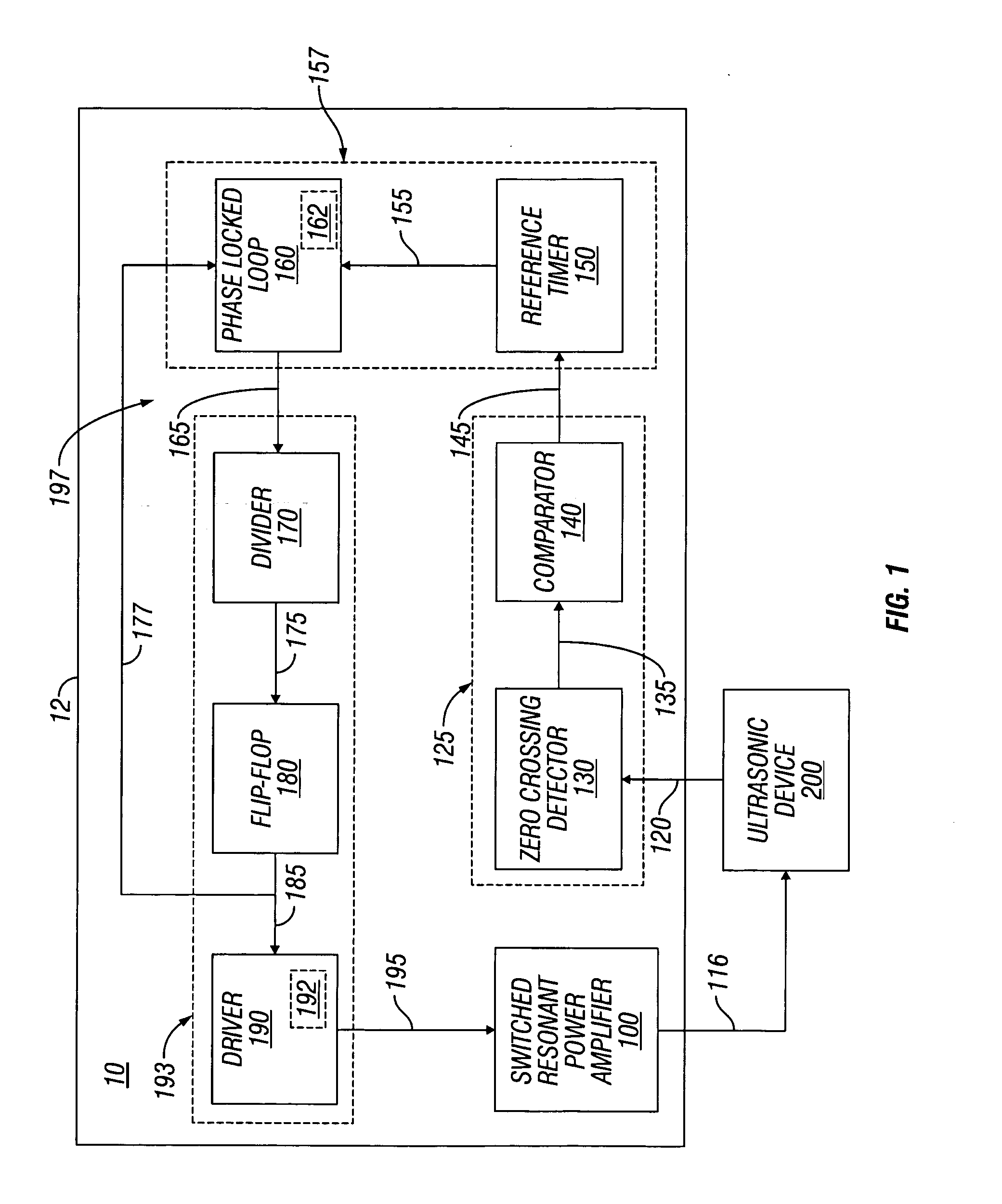
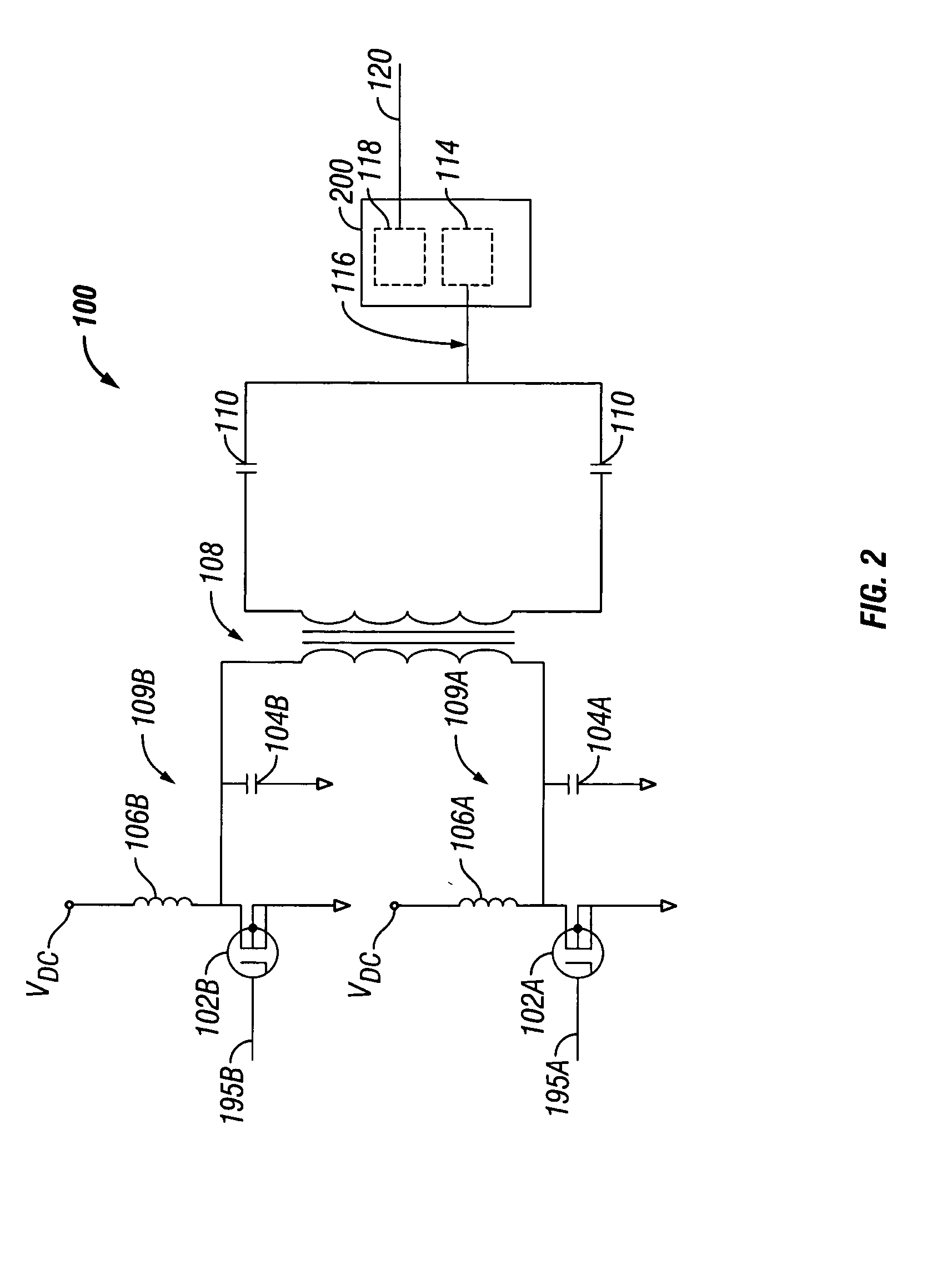
Switched resonant ultrasonic power amplifier system
ActiveUS20050149151A1Reduce frequencyControl outputUltrasound therapyAmplifier modifications to raise efficiencySonificationAudio power amplifier
A switched resonant power amplifier system for ultrasonic transducers is disclosed. The system includes an amplifier that receives and processes a driver output signal for generating a drive signal that is provided to an ultrasonic device for controlling output of the ultrasonic device. An output control circuit receives and processes a signal related to a feedback signal generated by the ultrasonic device and a divider reference signal, and generates a compensated clock signal that is adjusted for at least one of phase and frequency differences between the received feedback signal and the divider reference signal. A compensated drive circuit receives and processes the compensated clock signal for generating the divider reference signal, and for generating the driver output signal.
Owner:COVIDIEN AG
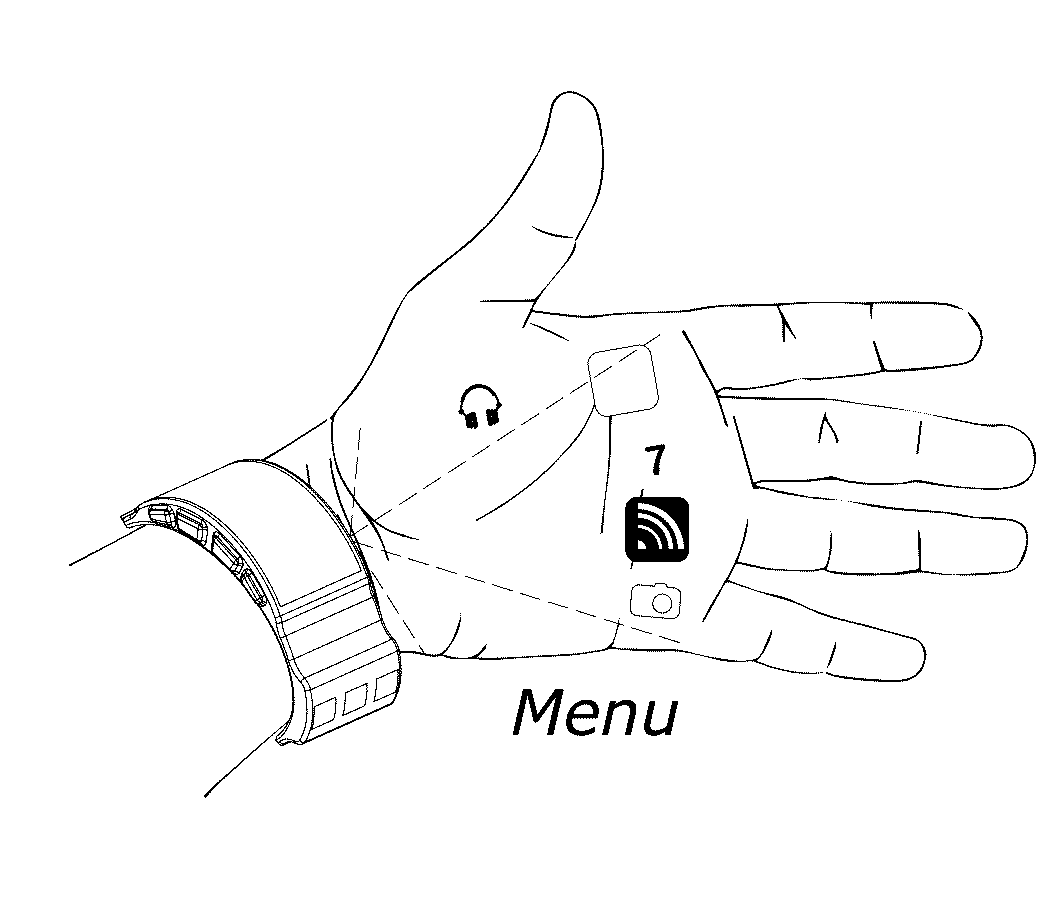
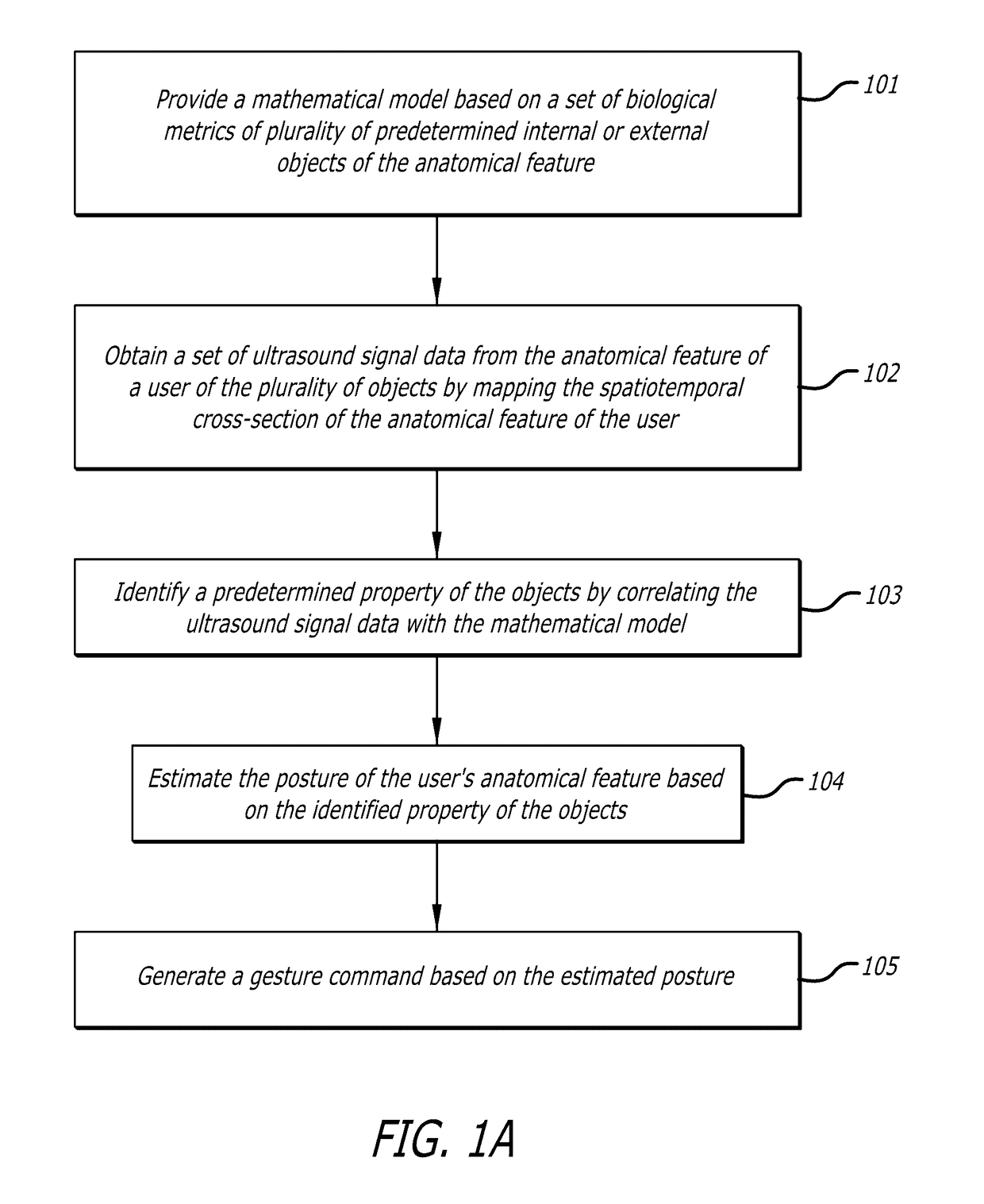
System and methods for on-body gestural interfaces and projection displays
ActiveUS20170123487A1Input/output for user-computer interactionDetails for portable computersSonificationTransceiver
A wearable system with a gestural interface for wearing on, for instance, the wrist of a user. The system comprises an ultrasonic transceiver array structure and may comprise a pico projector display element for displaying an image on a surface. User anatomical feature inputs are received in the form of ultrasound signals representative of a spatio-temporal cross-section of the wrist of the user by articulating wrist, finger and hand postures, which articulations are translated into gestures. Inputs from inertial and other sensors are used by the system as part of the anatomical feature posture identification method and device. Gestures are recognized using a mathematically-modeled, simulation-based set of biological metrics of tissue objects, which gestures are converted to executable computer instructions. Embodiments of the system disclosed herein may also be used to monitor biometric and health data over computer networks or using onboard systems.
Owner:OSTENDO TECH INC
Apparatus and method for non-invasive and minimally-invasive sensing of venous oxygen saturation and pH levels
InactiveUS20060224053A1Easy to useCost effectiveOrgan movement/changes detectionSensorsSonificationScattering cross-section
Medical diagnostic apparatus and methods are disclosed. Ultrasound radiation pressure selectively modulates a target area within a body. One or more pulses of radiation containing temporally correlated groups of photons are generated. The photons are characterized by two or more different wavelengths that are selected to have specific interaction with a target chromophore. The two or more different wavelengths are also selected to have substantially similar scattering cross-sections and anisotropy parameters in the target and its surroundings. The pulses of radiation are injected into the body proximate the target area being modulated by the radiation pressure field. Photon groups at each of the different wavelengths that are backscattered from the target area are detected in temporal coincidence. Time-gated background-free amplification of the return signal is used to exclude photons which could not by virtue of their arrival time have interacted with the radiation-pressure-modulated target. Photon groups are selected with a modulation component at the modulation frequency of the radiation pressure modulation field, or at a harmonic of the modulation frequency. From the arrival rate of the detected temporally correlated photon pairs or multiplets, chemical information about the target area, such as an oxygenation or pH level can be inferred. Cardiac output may be computed from measurements of venous and / or arterial oxygenation using this technique.
Owner:SKYLINE BIOMEDICAL
Method and apparatus for contextual voice cues
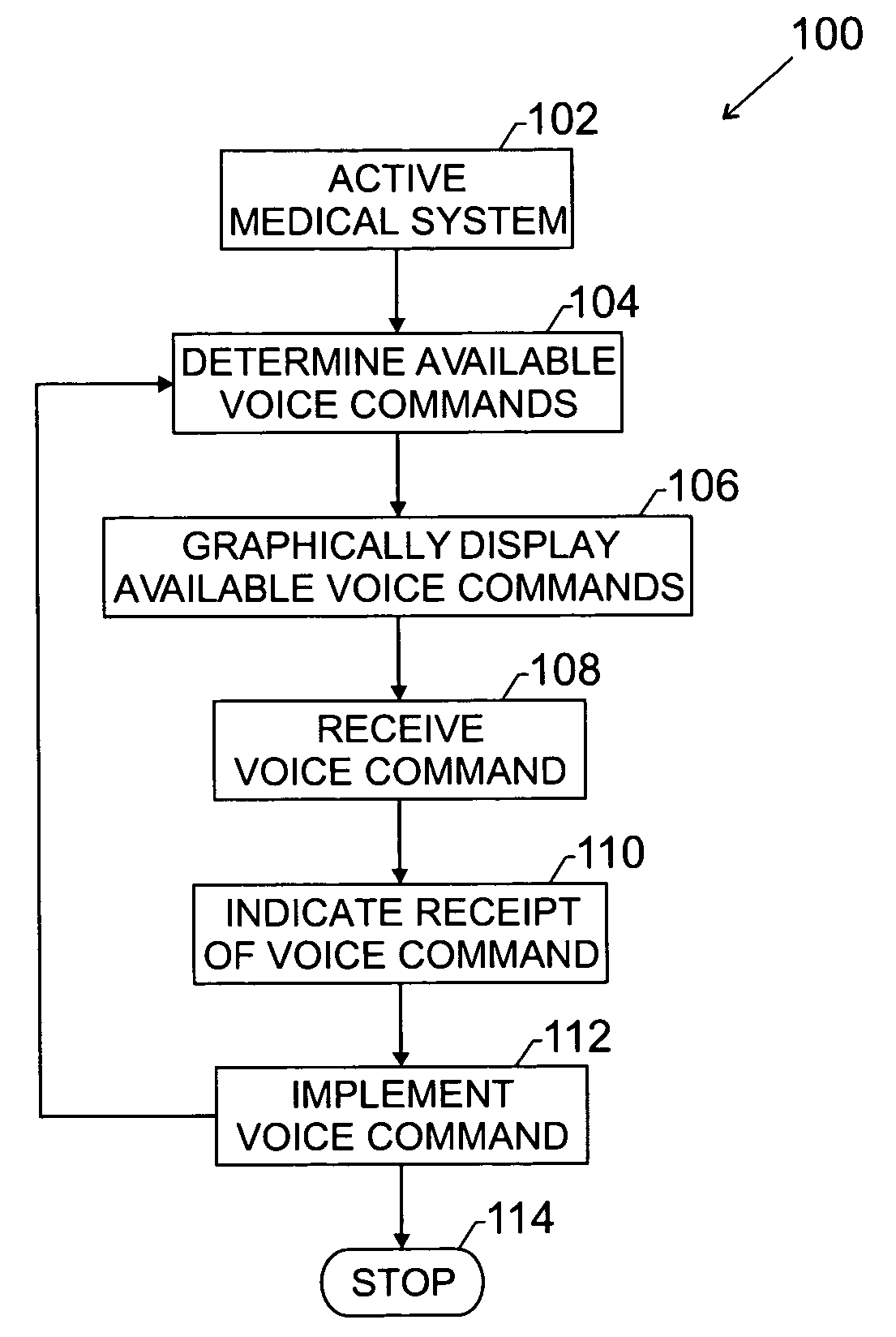
InactiveUS20050114140A1Increase adoptionImprove advancedLocal control/monitoringSpeech recognitionGraphicsSonification
The present invention provides a novel technique designed to provide a front-end graphical user interface for voice interaction, displaying a list of voice commands that can be used within a control scope of a medical system and that change depending on where the user is in the system. The user is presented with a quick reference guide to available commands without being overwhelmed. “Contextual voice cues” (CVC) provide a non-intrusive dynamic list of available commands to the user which automatically pop-up and change depending on the screen or mode the user is in. An indicator, such as a feedback light, may show whether a voice command is accepted. The technique may be utilized with medical information and diagnostic systems such as picture archival communication systems (PACS), ultrasound modalities, and so forth. Implementation of the technique should increase clinician adoption rates of voice recognition control and thus advance improvements in clinician workflow.
Owner:GE MEDICAL SYST INFORMATION TECH
Wireless power transmission apparatus using ultrasound
ActiveUS20150003207A1Improve efficiencyMultiple-port networksElectromagnetic wave systemElectric power transmissionSonification
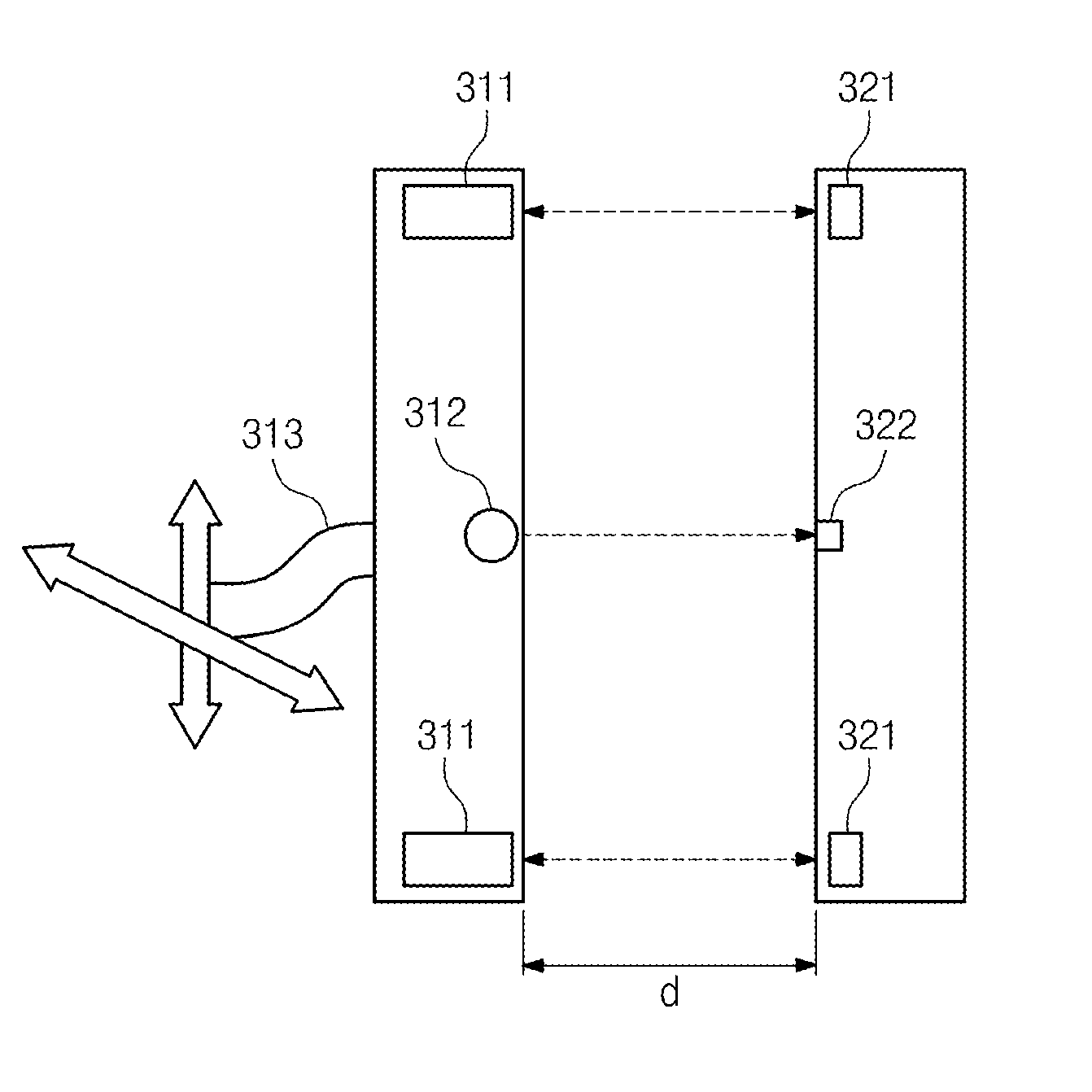
Provided is a wireless power transmission apparatus. The apparatus includes an ultrasound transmitter generating an ultrasound signal in response to an external source voltage and transmitting the generated ultrasound signal to a medium layer and an ultrasound receiver receiving the ultrasound signal through the medium layer and converting the received ultrasound signal into a driving voltage. The ultrasound transmitter and receiver are manufactured to control impedance values thereof to be matched with each other and a distance between the ultrasound transmitter and receiver is controlled according to predetermined distance conditions.
Owner:ELECTRONICS & TELECOMM RES INST
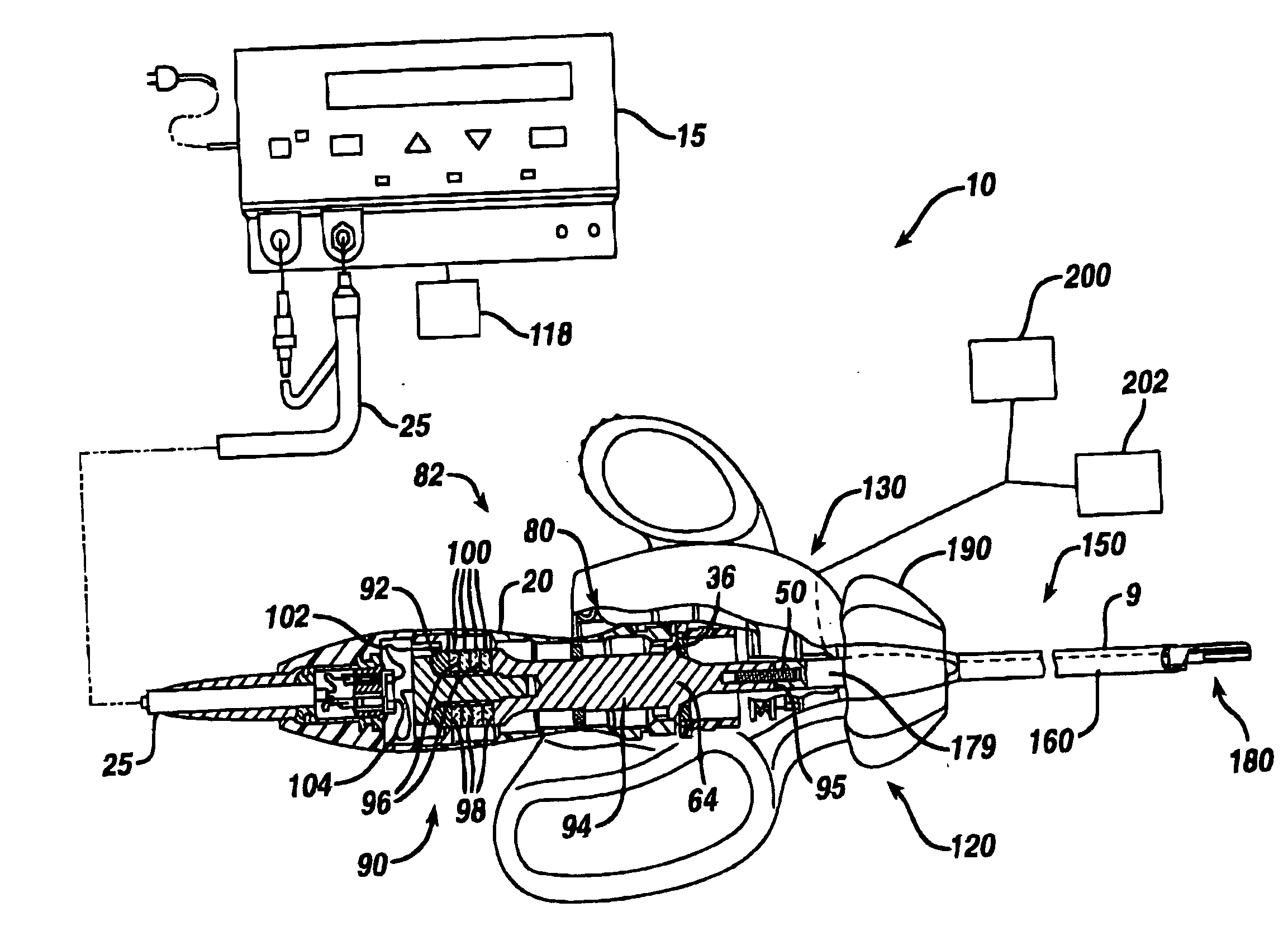
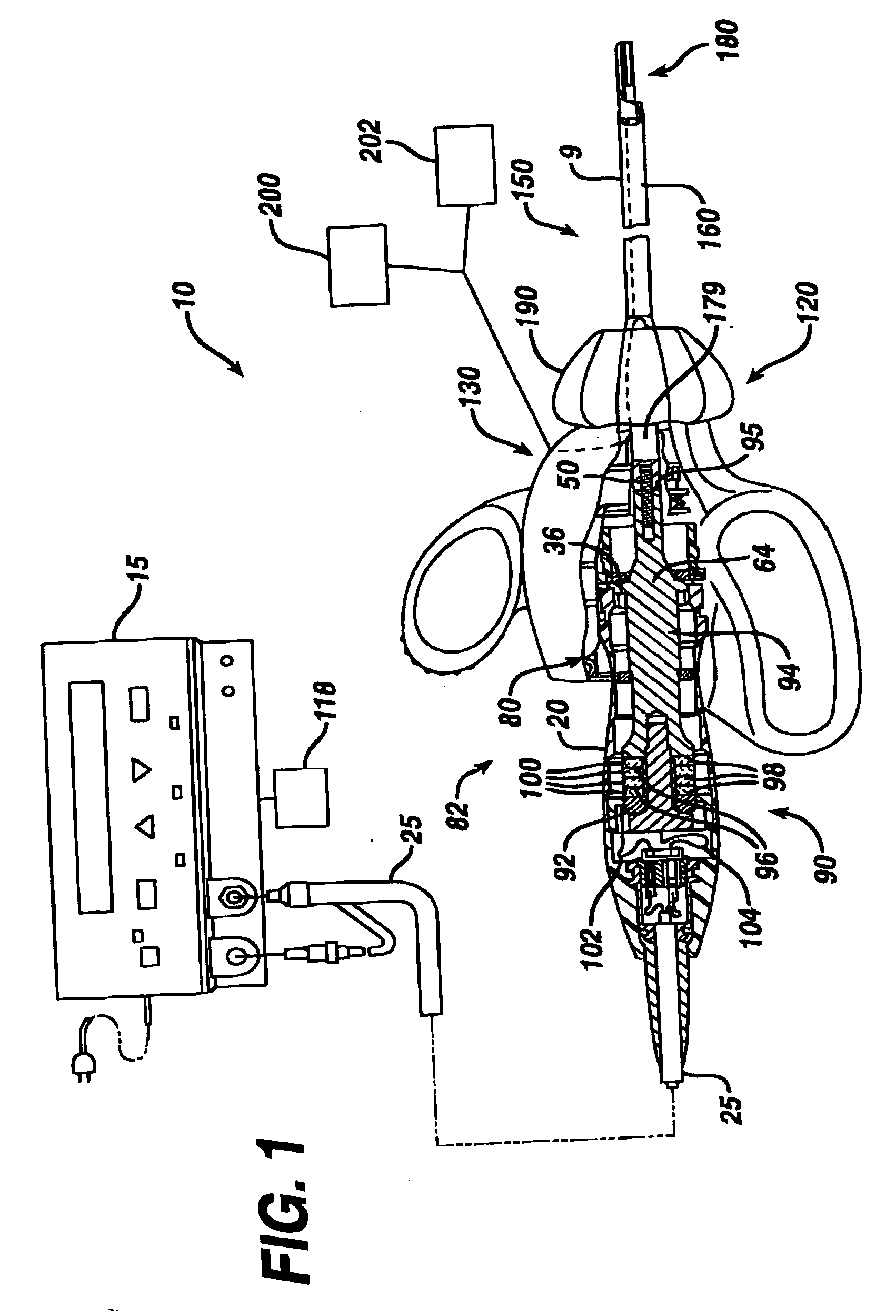
Ultrasonic surgical instrument incorporating fluid management
ActiveUS20050049546A1Load minimizationEfficient removalDiaphragm valvesEngine diaphragmsSurgical bladeSonification
Disclosed is an ultrasonic surgical device having a distally / proximally movable fluid management system consisting of single lumen or multiple lumens. The invention provides for the delivery of irrigation fluid or the removal of fluid, debris or vapor from the tissue-effecting portion of the blade while minimizing the loading on the blade. The blades of the surgical device, when excited at a natural blade system frequency, will have modal shapes characterized by longitudinal, transverse and / or torsional motion and will have nodal locations for these motions at positions along the tissue effecting length of the blade. The instrument is designed to allow for the fluid management system to be positioned at one or more motion nodes to facilitate efficient removal of tissue or fluid, which tends to accumulate at such nodes of the ultrasonic surgical blades.
Owner:ETHICON ENDO SURGERY INC
Systems and methods for characterization of materials and combinatorial libraries with mechanical oscillators
InactiveUS6182499B1Optical radiation measurementMaterial nanotechnologySonificationVisual perception
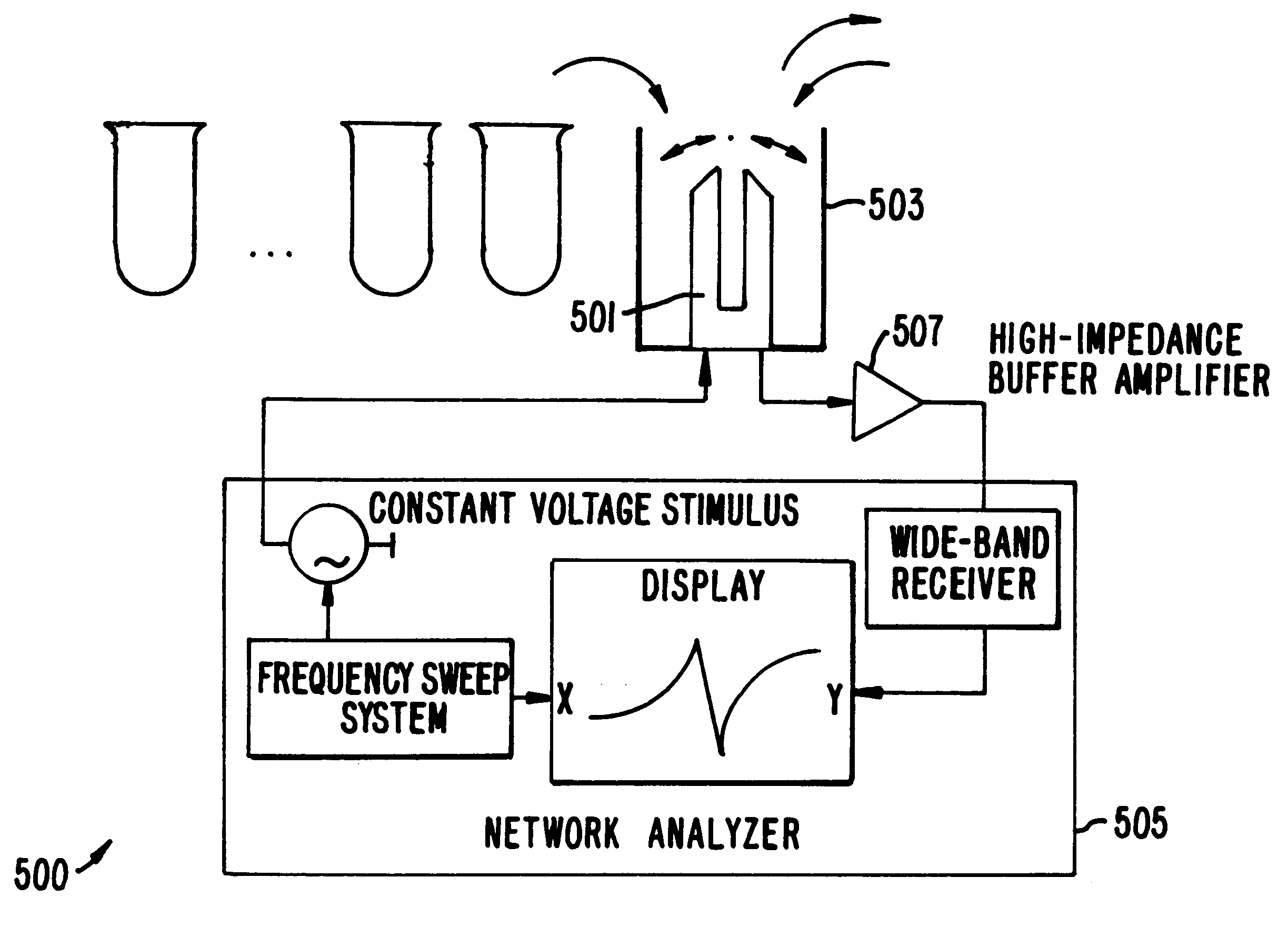
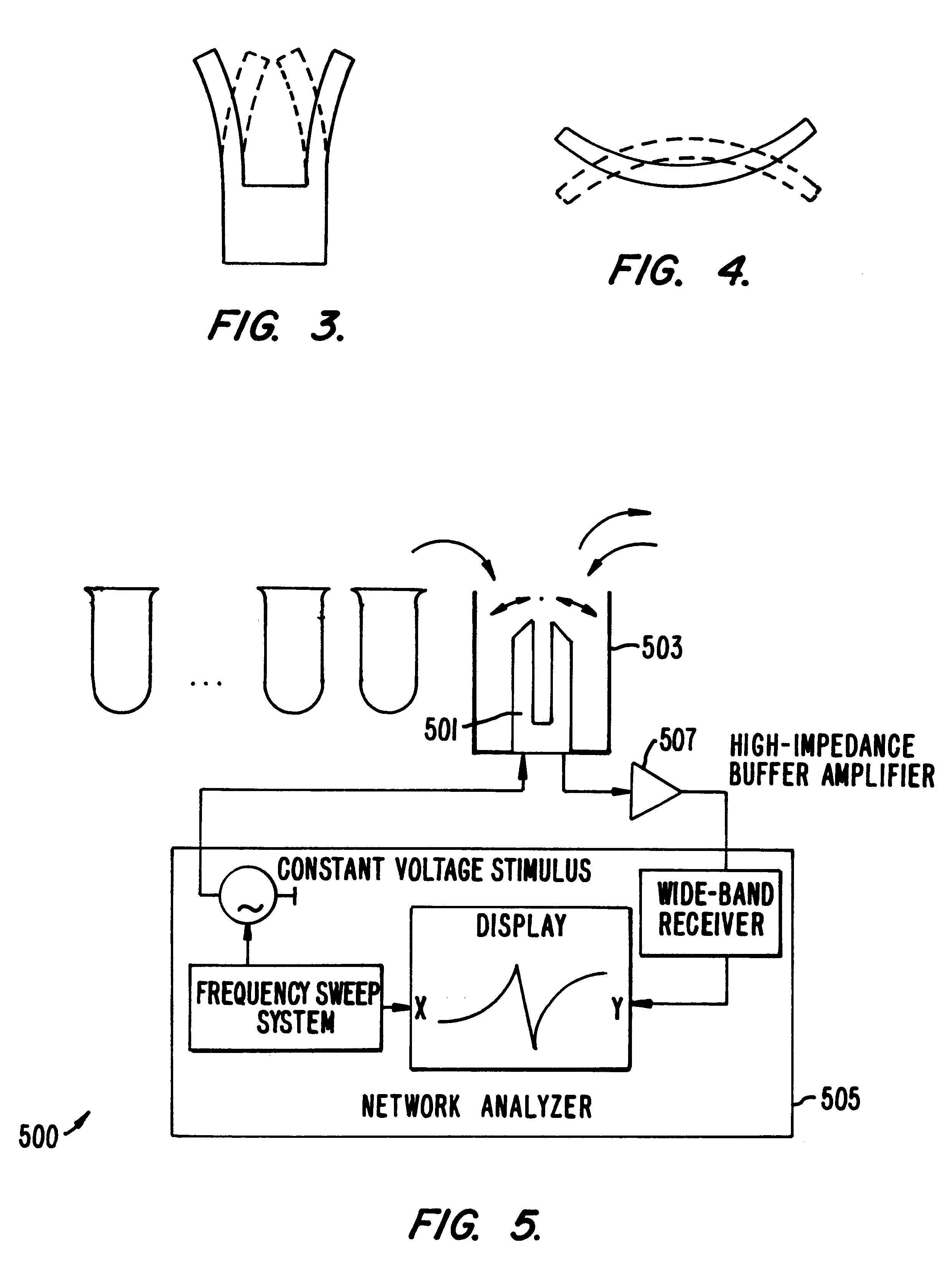
Methods and apparatus for screening diverse arrays of materials are provided. In one aspect, systems and methods are provided for imaging a library of materials using ultrasonic imaging techniques. The system includes one or more devices for exciting an element of the library such that acoustic waves are propagated through, and from, the element. The acoustic waves propagated from the element are detected and processed to yield a visual image of the library element. The acoustic wave data can also be processed to obtain information about the elastic properties of the library element. In another aspect, systems and methods are provided for generating acoustic waves in a tank filled with a coupling liquid. The library of materials is then placed in the tank and the surface of the coupling liquid is scanned with a laser beam. The structure of the liquid surface disturbed by the acoustic wave is recorded, the recorded disturbance being representative of the physical structure of the library. In another aspect of the invention, a mechanical resonator is used to evaluate various properties (e.g., molecular weight, viscosity, specific weight, elasticity, dielectric constant, conductivity, etc.) of the individual liquid elements of a library of materials. The resonator is designed to ineffectively excite acoustic waves. The frequency response of the resonator is measured for the liquid element under test, preferably as a function of time. By calibrating the resonator to a set of standard liquids with known properties, the properties of the unknown liquid can be determined. An array of library elements can be characterized by a single scanning transducer or by using an array of transducers corresponding to the array of library elements. Alternatively, multiple resonators of differing design may be used to evaluate each element of a library of elements, thus providing improved dynamic range and sensitivity.
Owner:FREESLATE
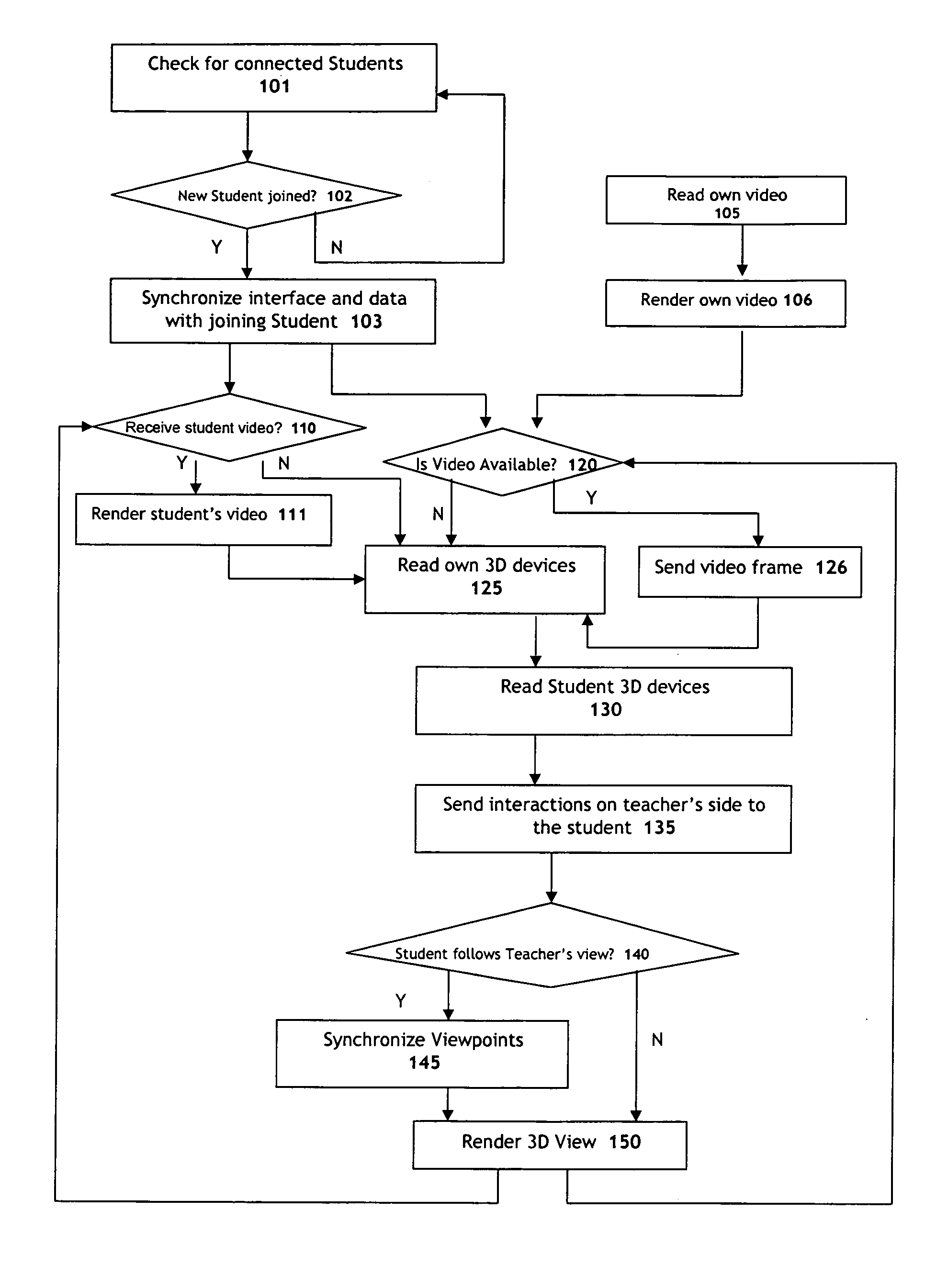
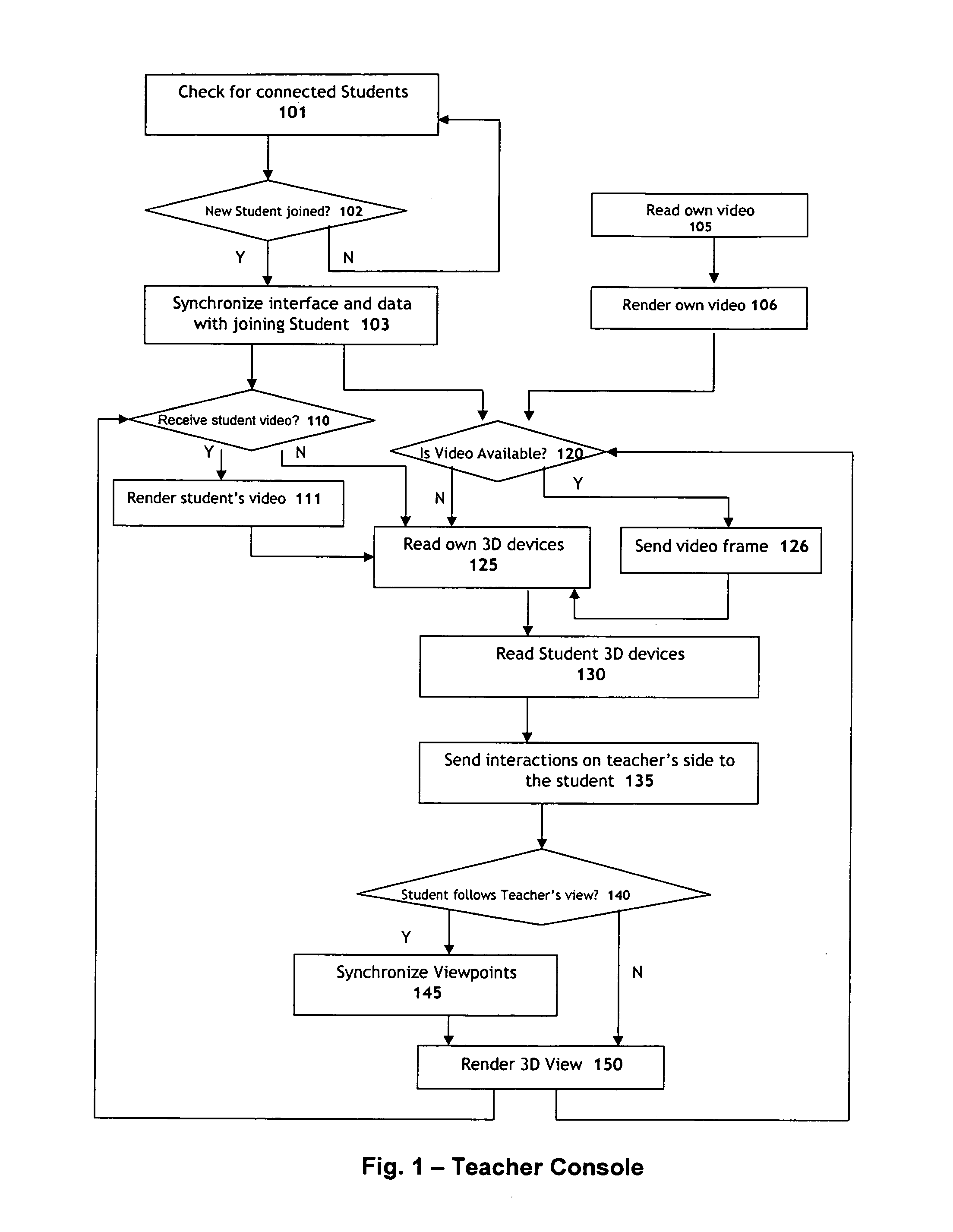
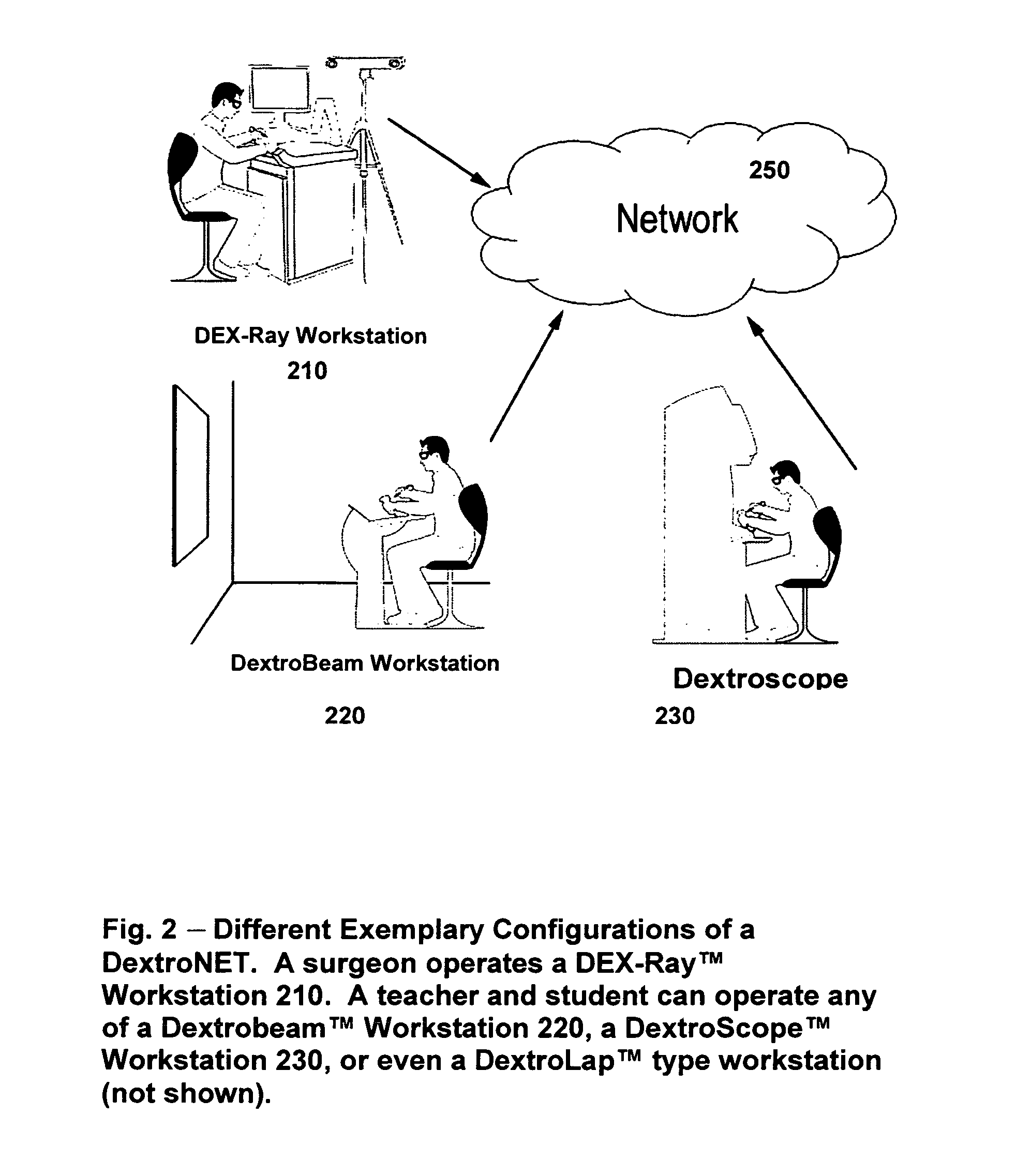
Systems and methods for collaborative interactive visualization of 3D data sets over a network ("DextroNet")
Owner:BRACCO IMAGINIG SPA
User interface for handheld imaging devices
InactiveUS20060116578A1Reduce total usageMinimize timeBlood flow measurement devicesLocal control/monitoringUltrasonographyData display
A Graphical User Interface (GUI) for an ultrasound system. The ultrasound system has operational modes and the GUI has corresponding icons, tabs, and menu items image and information fields. The User Interface (UI) provides several types of graphical elements with intelligent behavior, such as being context sensitive and adaptive, called active objects, for example, tabs, menus, icons, windows of user interaction and data display and an alphanumeric keyboard. In addition the UI may also be voice activated. The UI further provides for a touchscreen for direct selection of displayed active objects. In an embodiment, the UI is for a medical ultrasound handheld imaging instrument. The UI provides a limited set of hard and soft keys with adaptive functionality that can be used with only one hand and potentially with only one thumb.
Owner:ZONARE MEDICAL SYST
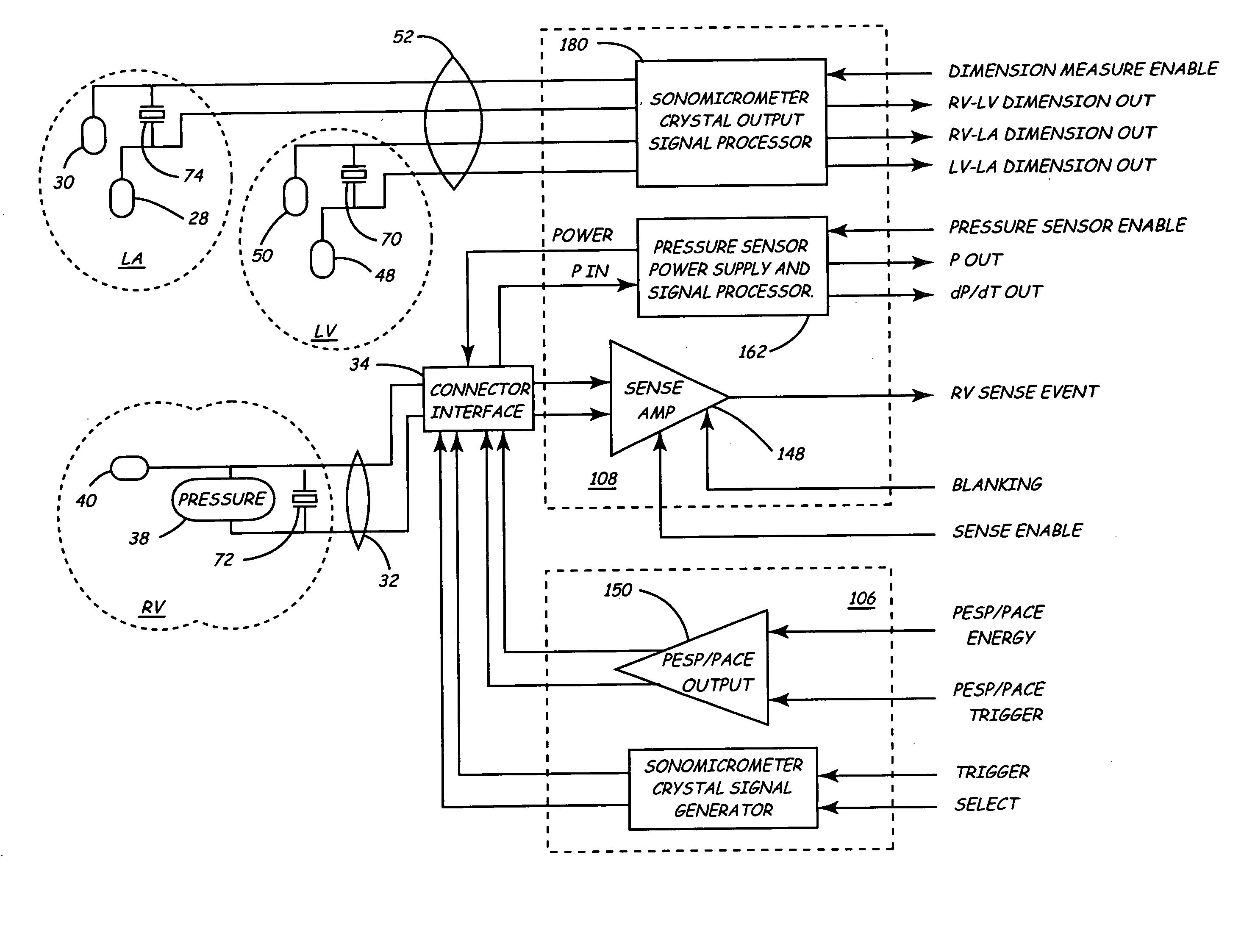
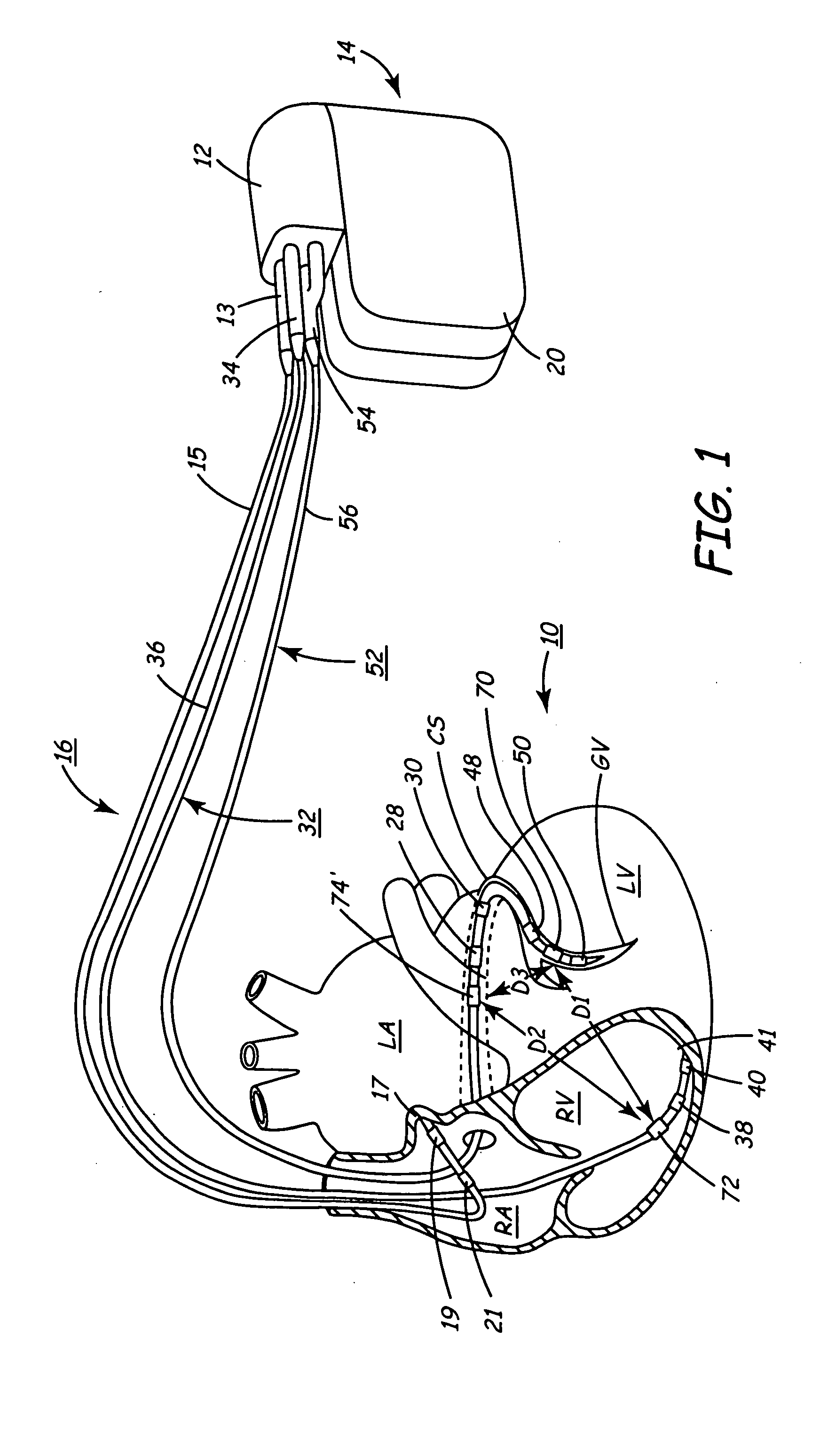
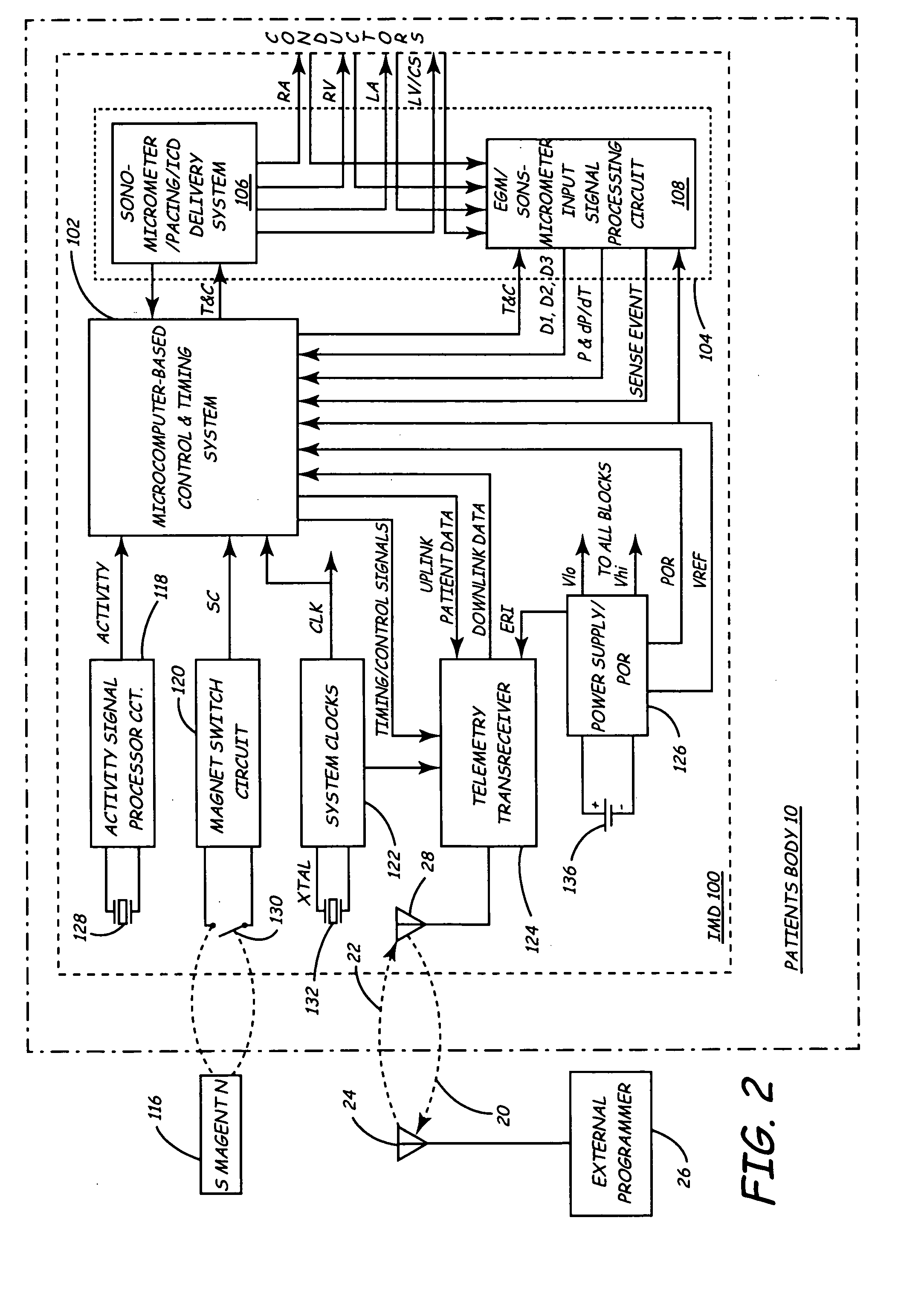
Implantable medical device for monitoring cardiac blood pressure and chamber dimension
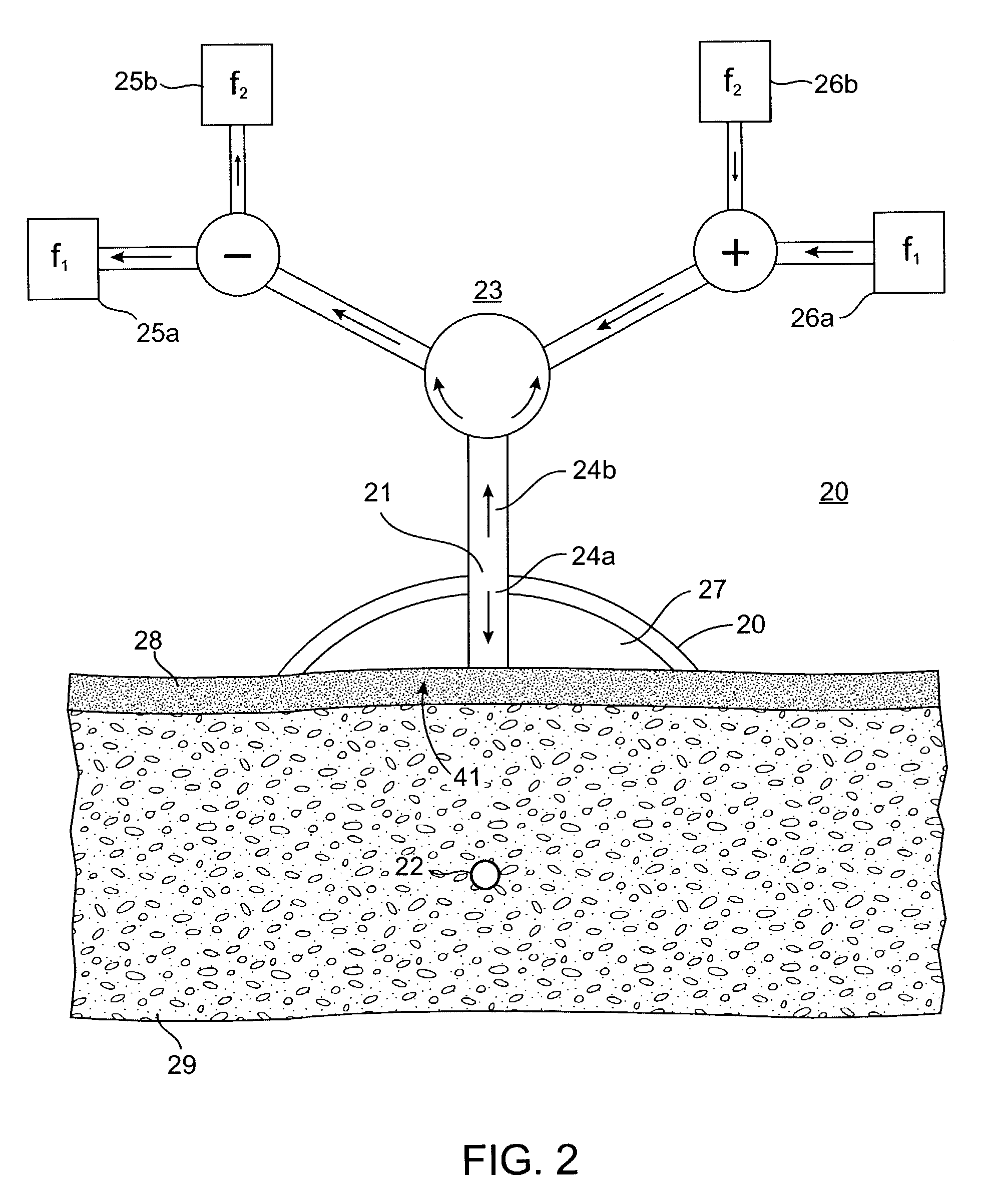
InactiveUS20050027323A1Maximize cardiac outputConvenient timeCatheterHeart stimulatorsSonificationHeart chamber
Implantable medical devices (IMDs) for monitoring signs of acute or chronic cardiac heart failure by measuring cardiac blood pressure and mechanical dimensions of the heart and providing multi-chamber pacing optimized as a function of measured blood pressure and dimensions are disclosed. The dimension sensor or sensors comprise at least a first sonomicrometer piezoelectric crystal mounted to a first lead body implanted into or in relation to one heart chamber that operates as an ultrasound transmitter when a drive signal is applied to it and at least one second sonomicrometer crystal mounted to a second lead body implanted into or in relation to a second heart chamber that operates as an ultrasound receiver. The ultrasound receiver converts impinging ultrasound energy transmitted from the ultrasound transmitter through blood and heart tissue into an electrical signal. The time delay between the generation of the transmitted ultrasound signal and the reception of the ultrasound wave varies as a function of distance between the ultrasound transmitter and receiver which in turn varies with contraction and expansion of a heart chamber between the first and second sonomicrometer crystals. One or more additional sonomicrometer piezoelectric crystal can be mounted to additional lead bodies such that the distances between the three or more sonomicrometer crystals can be determined. In each case, the sonomicrometer crystals are distributed about a heart chamber such that the distance between the separated ultrasound transmitter and receiver crystal pairs changes with contraction and relaxation of the heart chamber walls.
Owner:MEDTRONIC INC
Synchronization of ultrasound imaging data with electrical mapping
ActiveUS20070106146A1Improve diagnostic accuracyUltrasonic/sonic/infrasonic diagnosticsElectrocardiographyUltrasound imagingElectricity
An image of an electro-anatomical map of a body structure having cyclical motion is overlaid on a 3D ultrasonic image of the structure. The electro-anatomical data and anatomic image data are synchronized by gating both electro-anatomical data acquisition and an anatomic image at a specific point in the motion cycle. Transfer of image data includes identification of a point in the motion cycle at which the 3-dimensional image was captured or is to be displayed.
Owner:BIOSENSE WEBSTER INC
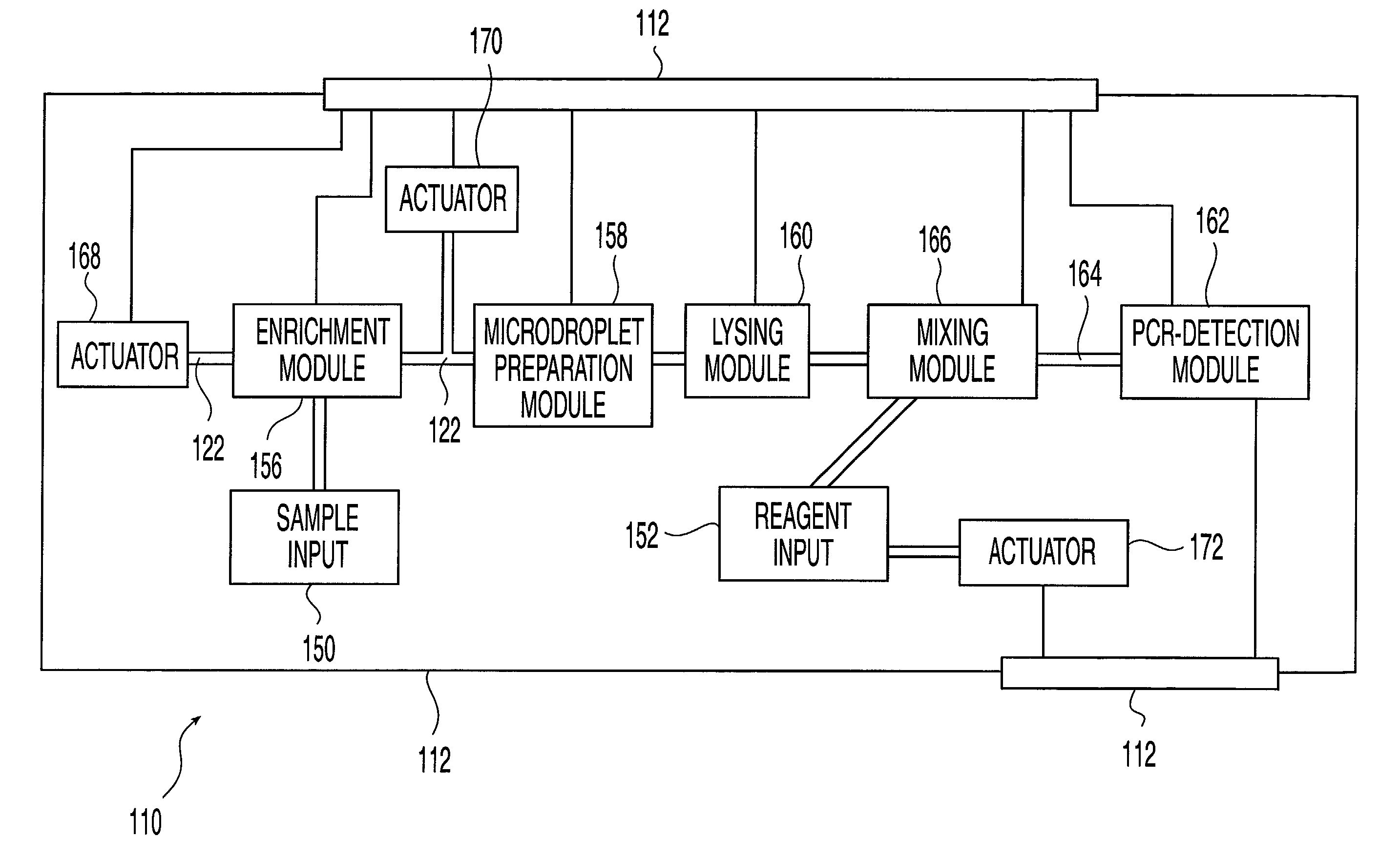
Methods and systems for releasing intracellular material from cells within microfluidic samples of fluids
InactiveUS7192557B2Bioreactor/fermenter combinationsFixed microstructural devicesSonificationCompound (substance)
The present invention relates to a microfluidic system for processing a cell-containing liquid. The system includes a lysing zone to receive the cell-containing sample and a positioning element to position the cell-containing sample in a lysing position in the vicinity of a lysing mechanism. The lysing mechanism releases intracellular material, such as DNA or RNA, from the cells. In one embodiment, the lysing mechanism includes electrodes for generating an electric field sufficient to release intracellular contents from the cells. Alternatively, the lysing mechanism may lyse the cells using chemical, heat and / or ultrasonic techniques or any combination of these techniques.
Owner:HANDLAB INC
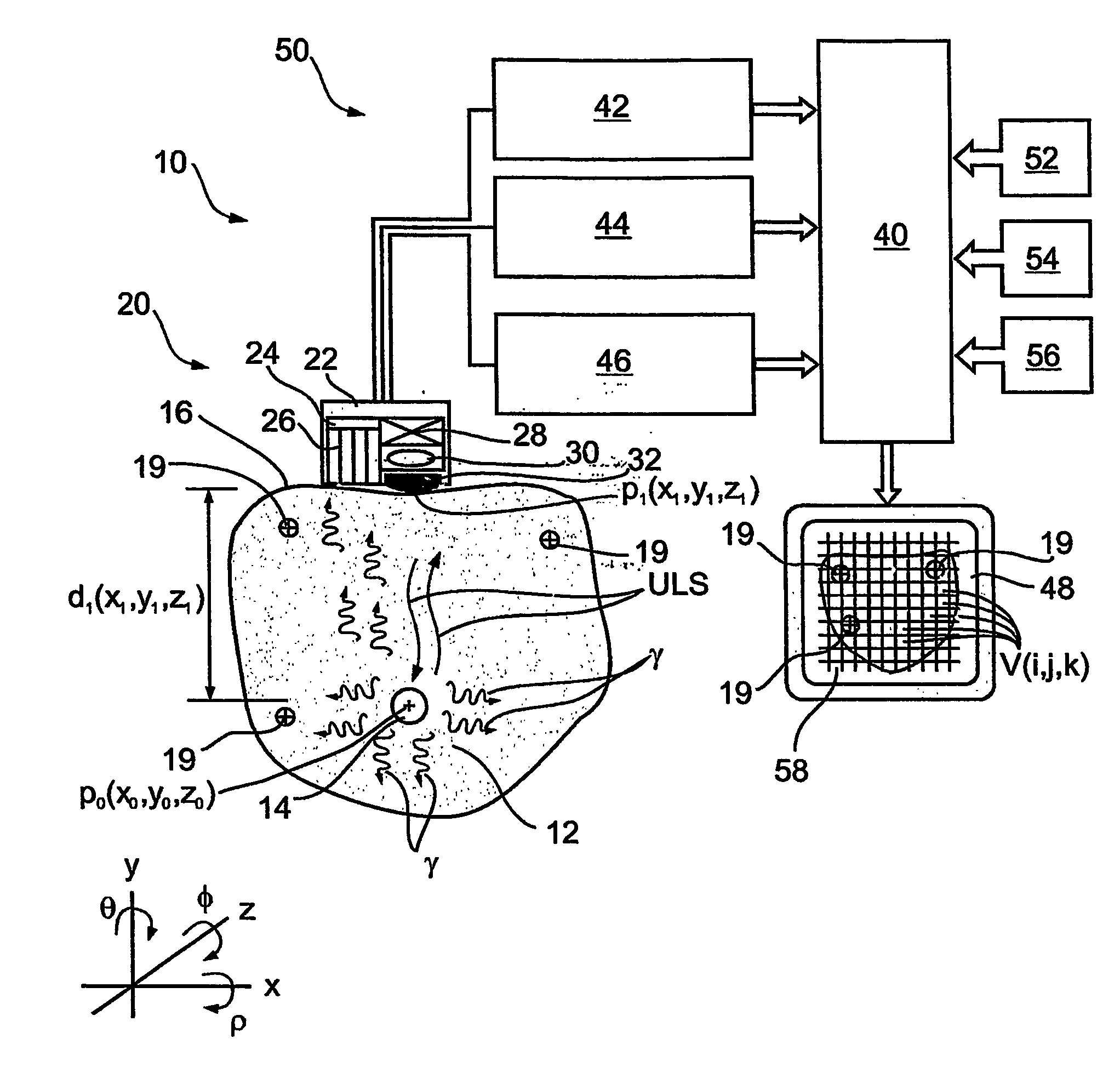
Apparatus and methods for imaging and attenuation correction
Imaging apparatus, is provided, comprising a first device, for obtaining a first image, by a first modality, selected from the group consisting of SPECT, PET, CT, an extracorporeal gamma scan, an extracorporeal beta scan, x-rays, an intracorporeal gamma scan, an intracorporeal beta scan, an intravascular gamma scan, an intravascular beta scan, and a combination thereof, and a second device, for obtaining a second, structural image, by a second modality, selected from the group consisting of a three-dimensional ultrasound, an MRI operative by an internal magnetic field, an extracorporeal ultrasound, an extracorporeal MRI operative by an external magnetic field, an intracorporeal ultrasound, an intracorporeal MRI operative by an external magnetic field, an intravascular ultrasound, and a combination thereof, and wherein the apparatus further includes a computerized system, configured to construct an attenuation map, for the first image, based on the second, structural image. Additionally, the computerized system is configured to display an attenuation-corrected first image as well as a superposition of the attenuation-corrected first image and the second, structural image. Furthermore, the apparatus is operative to guide an in-vivo instrument based on the superposition.
Owner:SPECTRUM DYNAMICS MEDICAL LTD
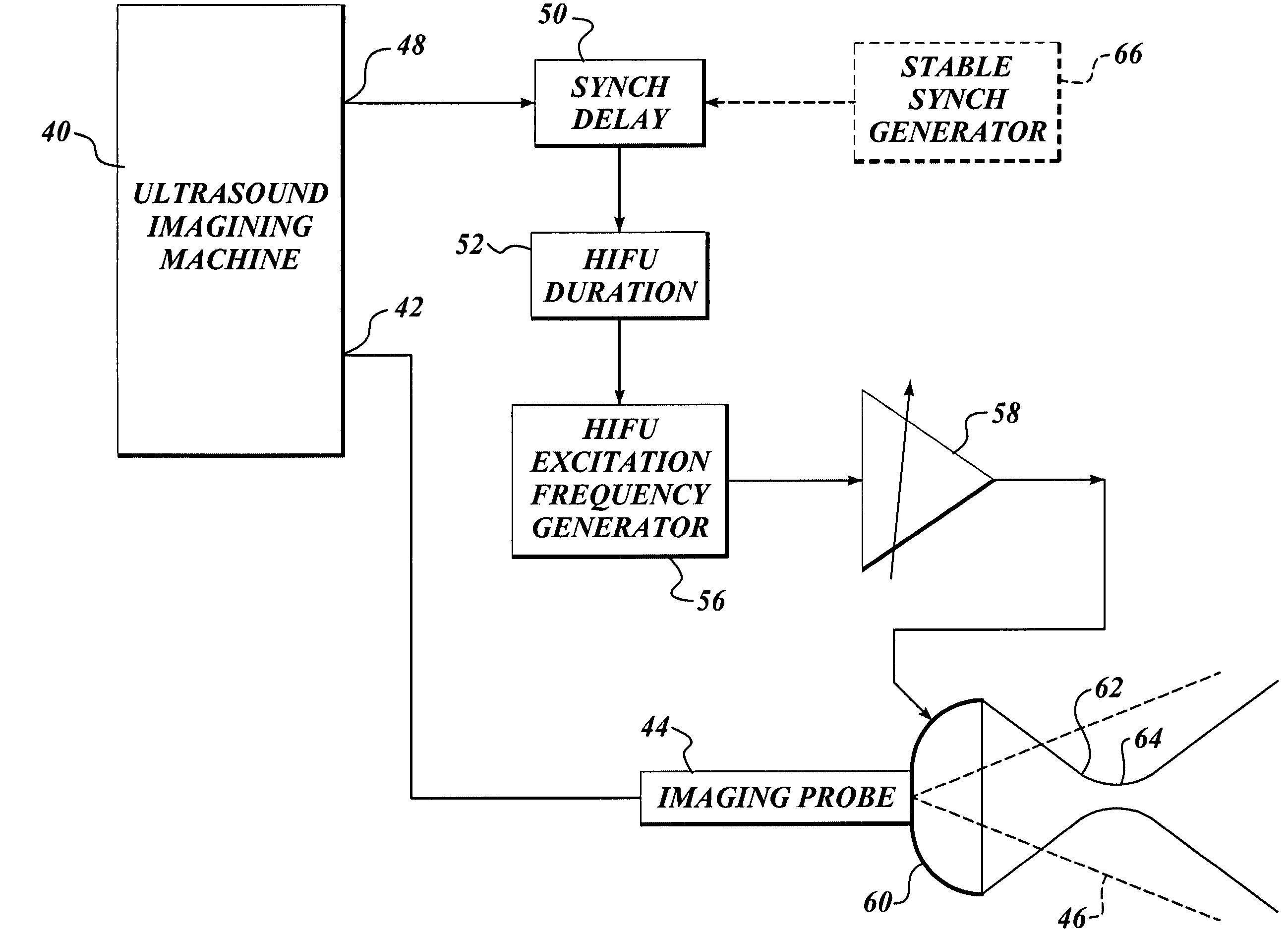
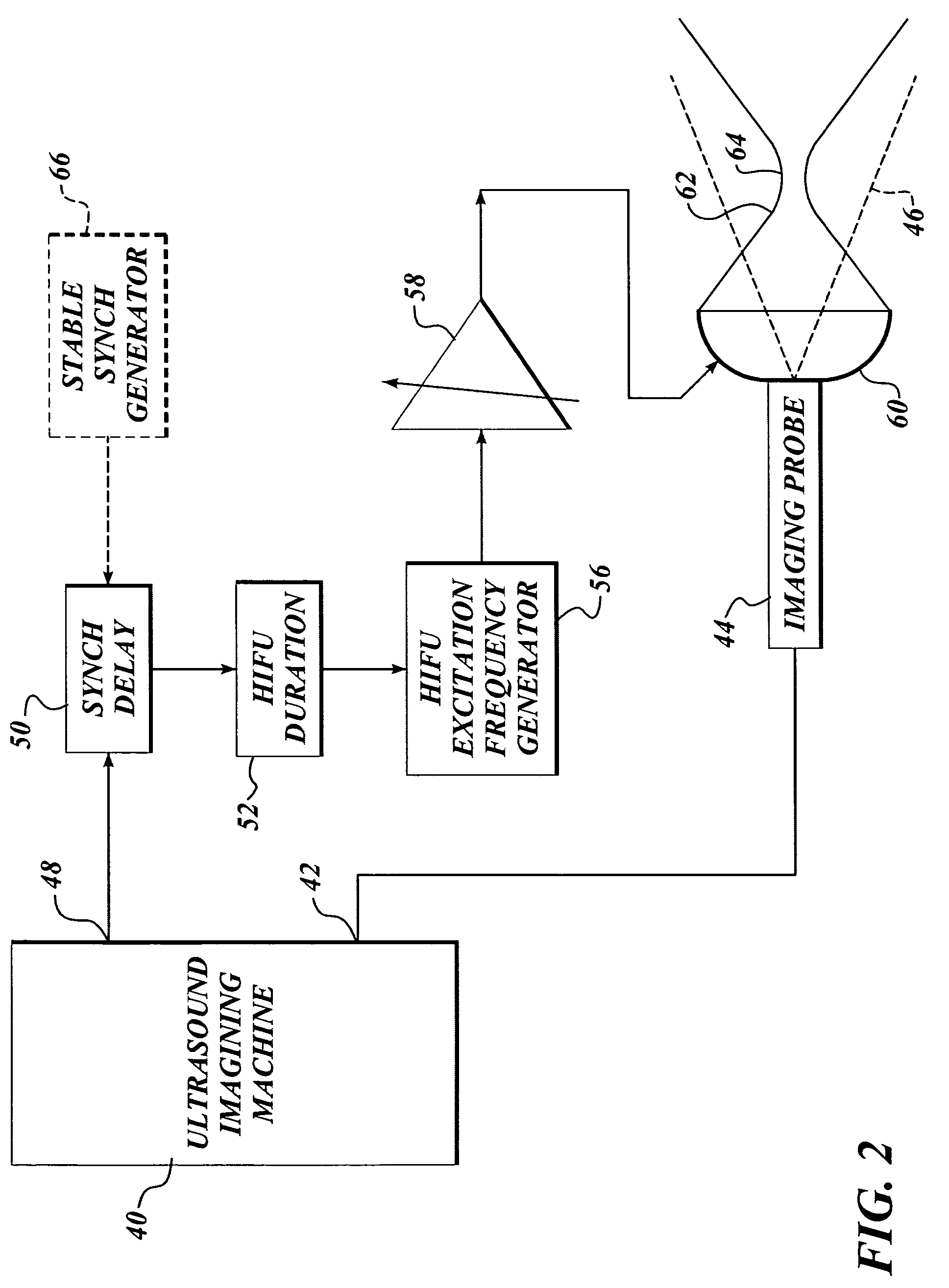
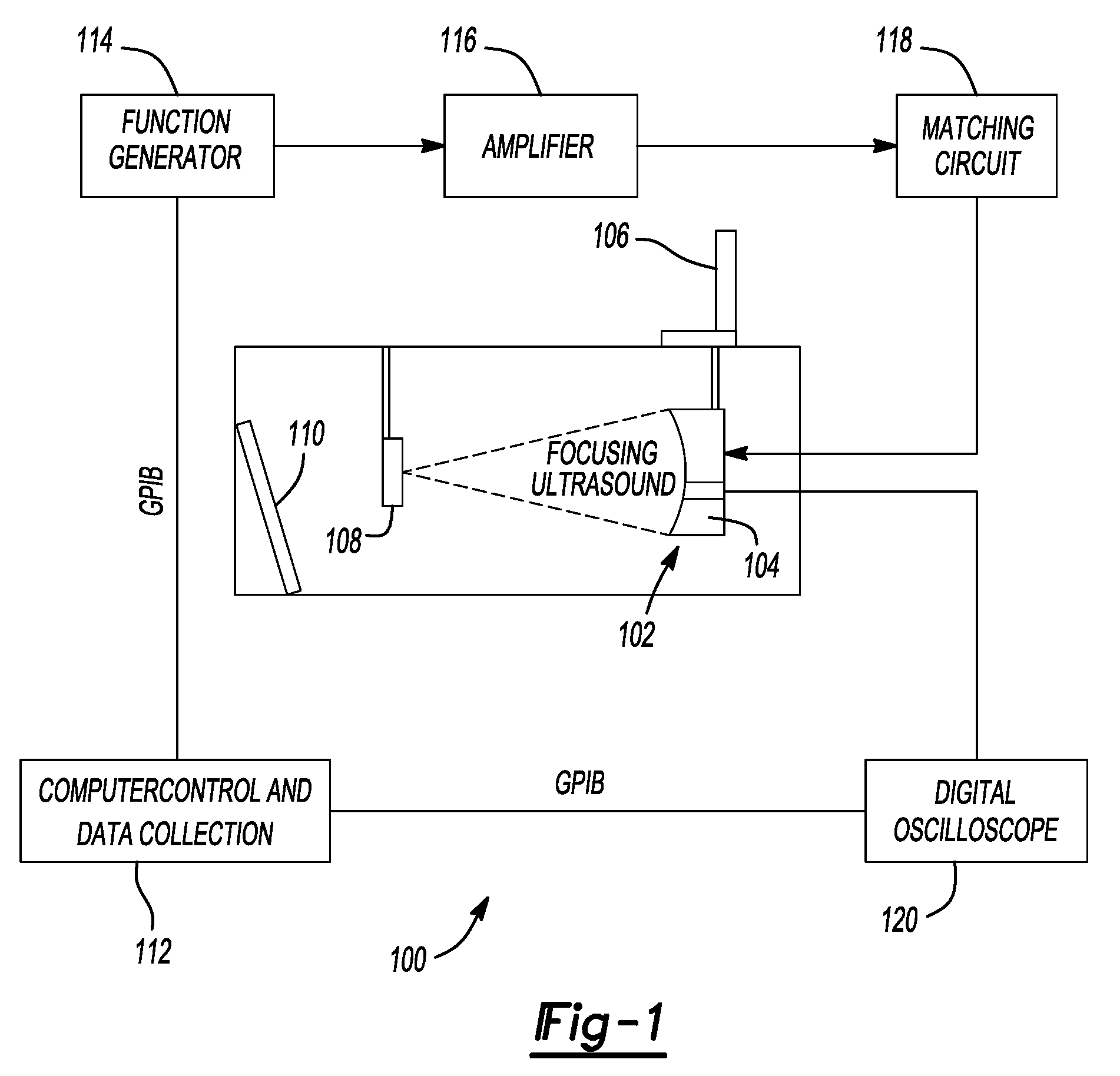
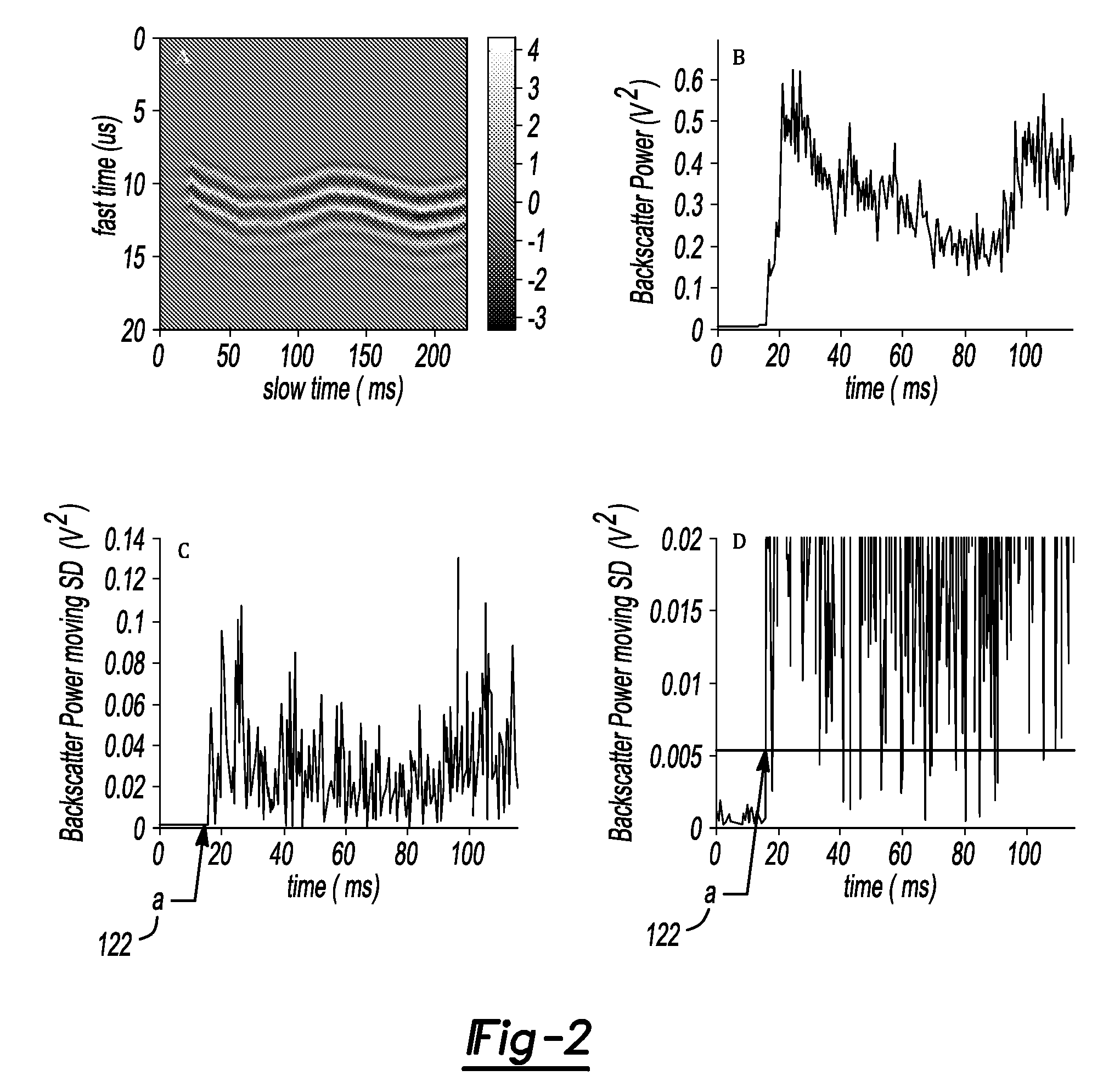
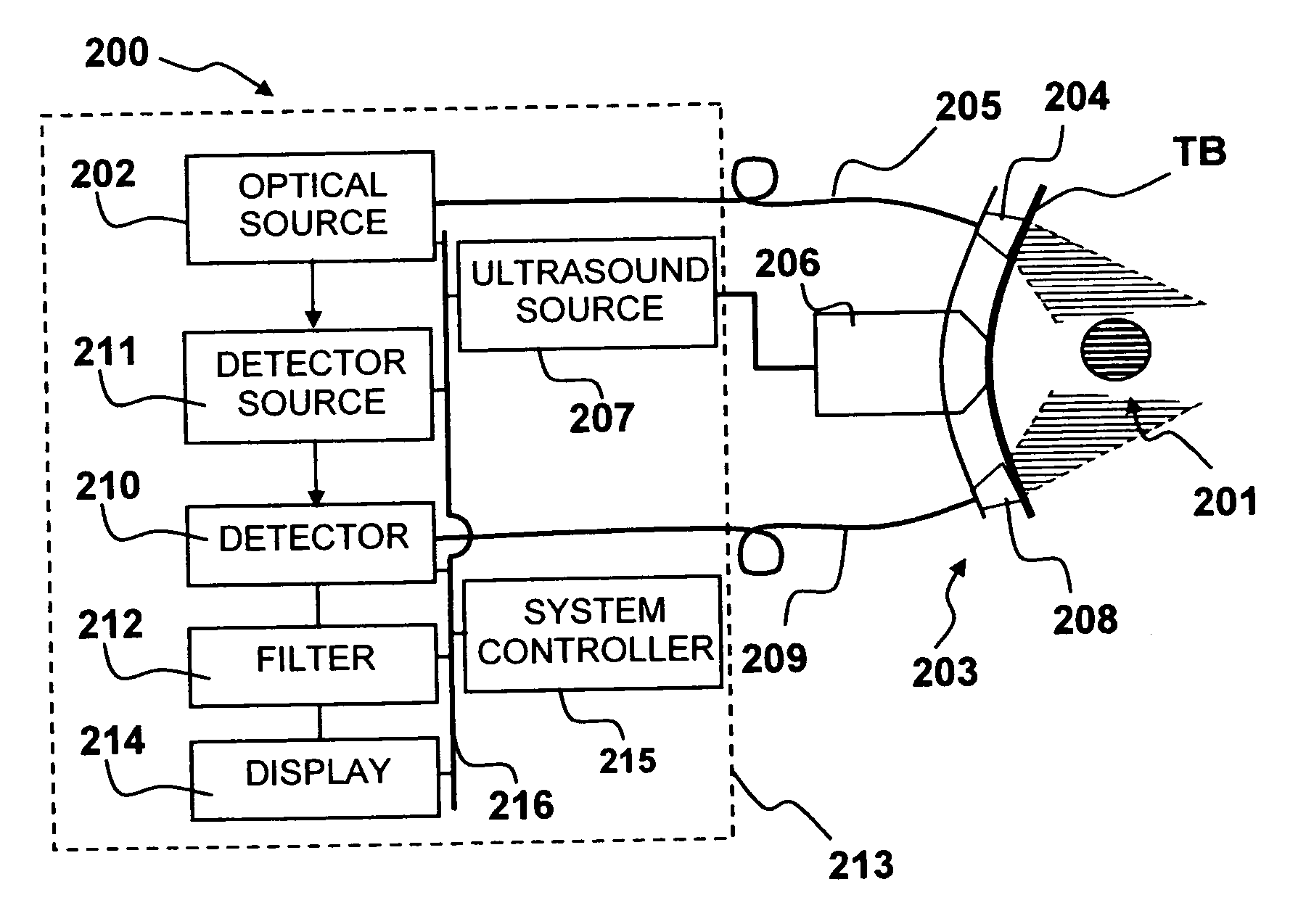
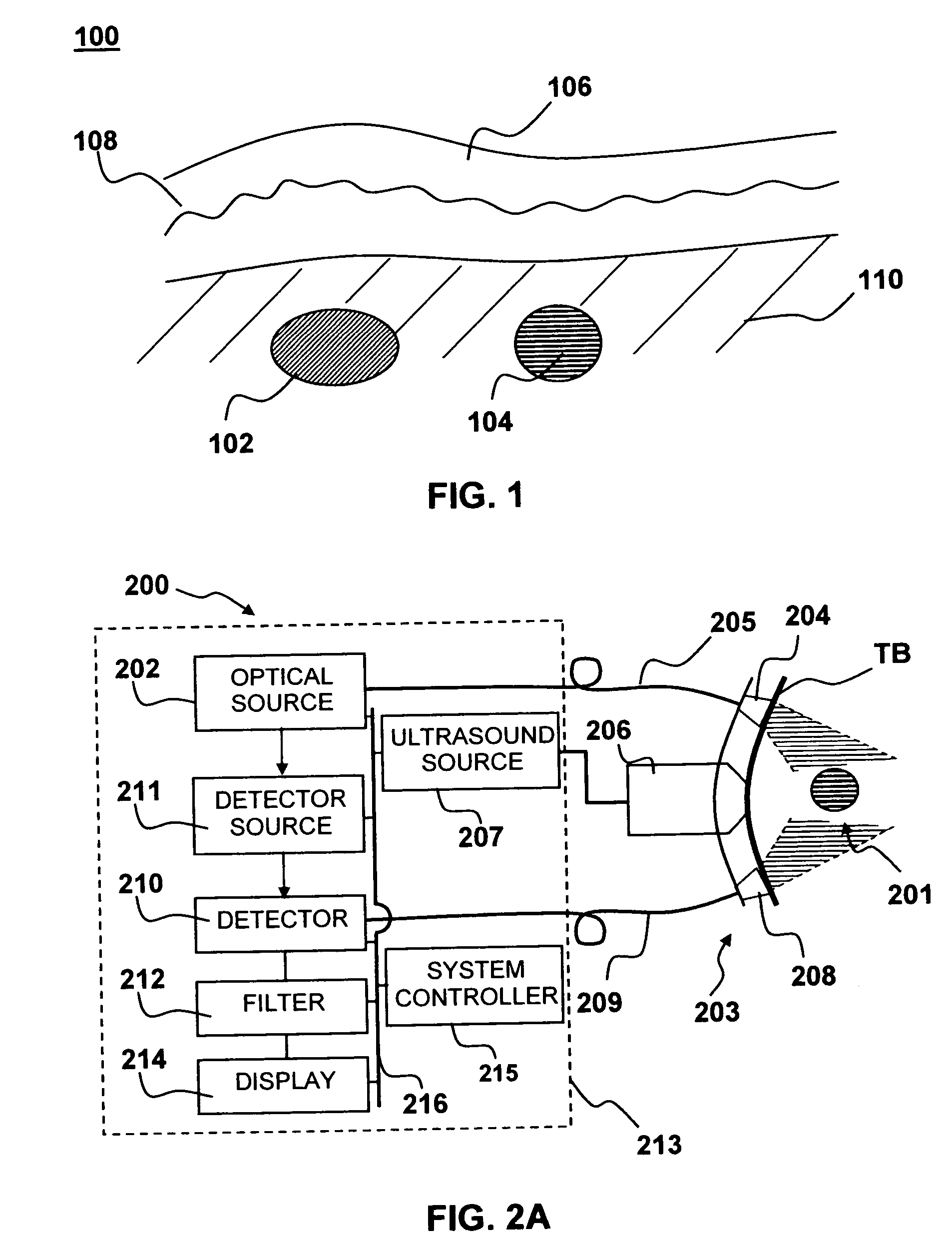
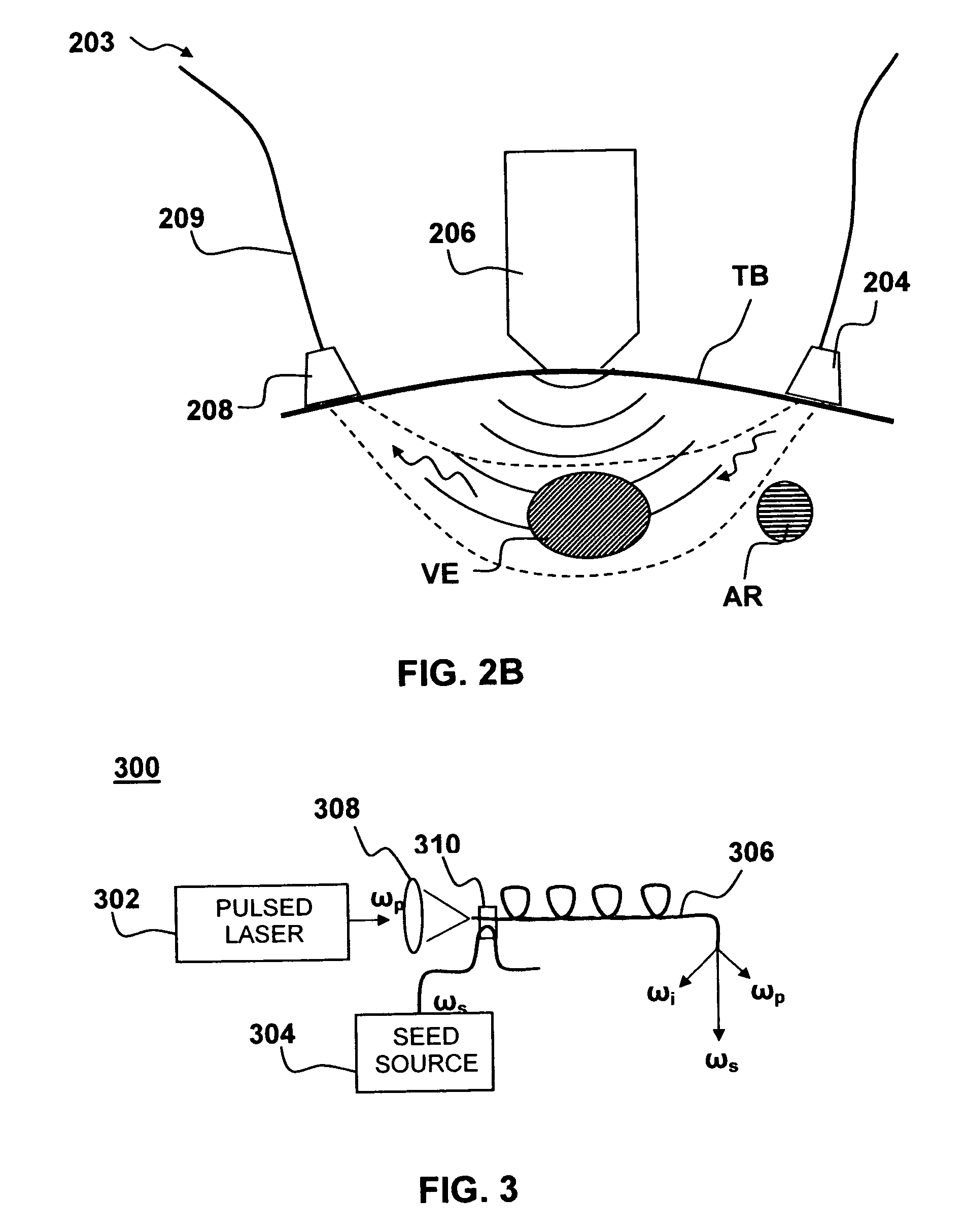
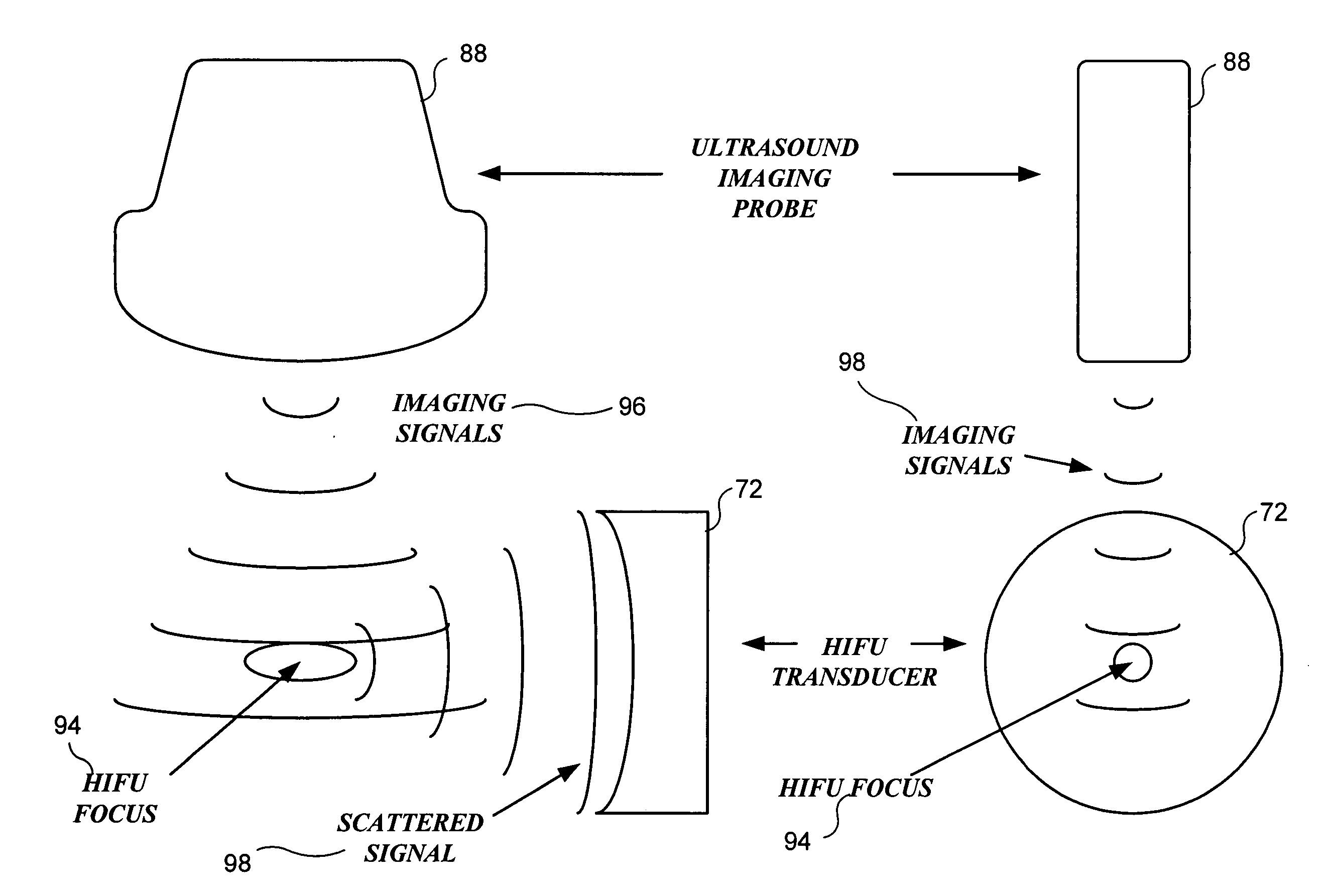
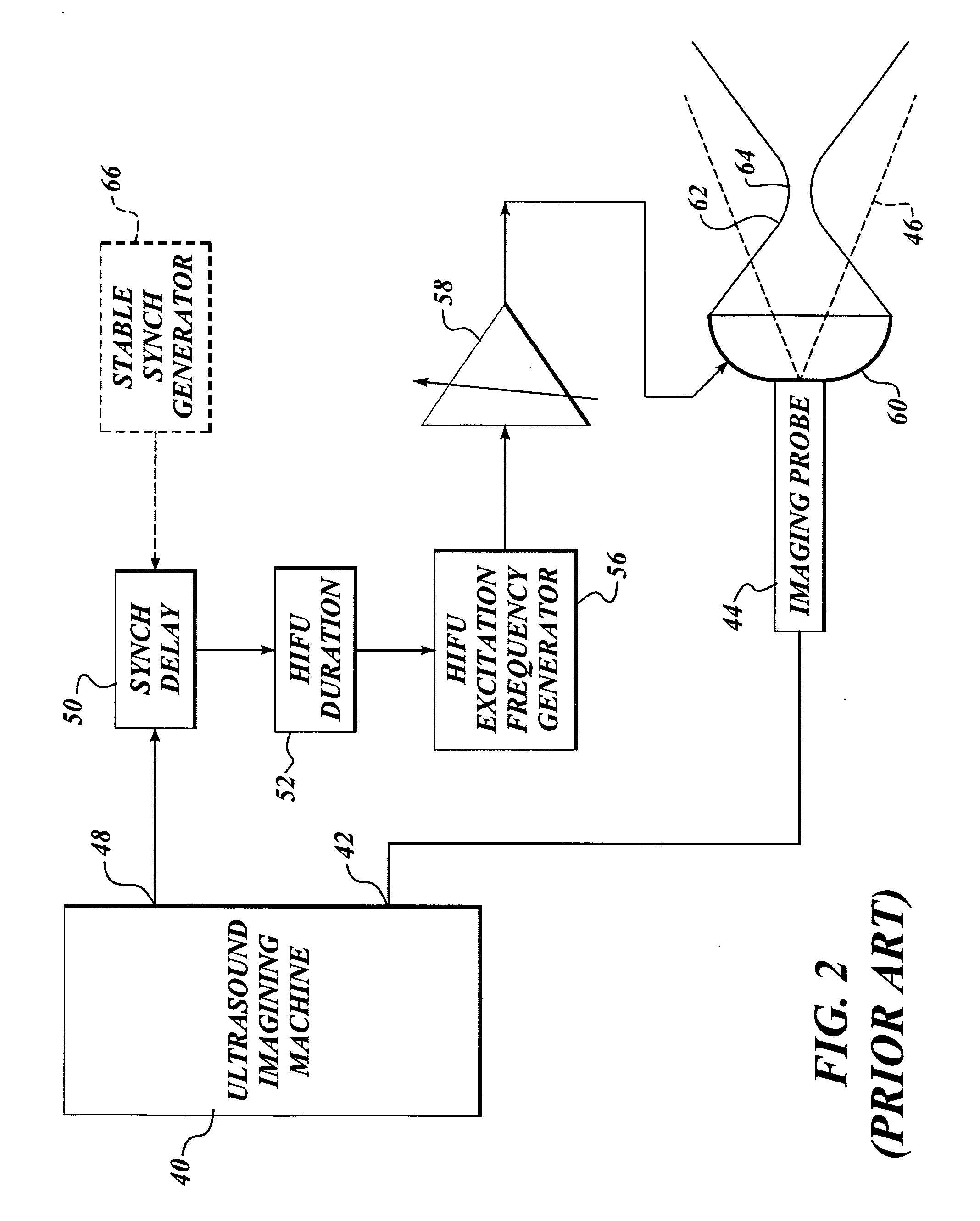
Method and system to synchronize acoustic therapy with ultrasound imaging
ActiveUS20070055155A1Synchronization is simpleGood synchronizationUltrasonic/sonic/infrasonic diagnosticsUltrasound therapyUltrasound imagingSonification
Interference in ultrasound imaging when used in connection with high intensity focused ultrasound (HIFU) is avoided by employing a synchronization signal to control the HIFU signal. Unless the timing of the HIFU transducer is controlled, its output will substantially overwhelm the signal produced by ultrasound imaging system and obscure the image it produces. The synchronization signal employed to control the HIFU transducer is obtained without requiring modification of the ultrasound imaging system. Signals corresponding to scattered ultrasound imaging waves are collected using either the HIFU transducer or a dedicated receiver. A synchronization processor manipulates the scattered ultrasound imaging signals to achieve the synchronization signal, which is then used to control the HIFU bursts so as to substantially reduce or eliminate HIFU interference in the ultrasound image. The synchronization processor can alternatively be implemented using a computing device or an application-specific circuit.
Owner:UNIV OF WASHINGTON
Wireless, ultrasonic personal health monitoring system
A personal monitoring device has a sensor assembly configured to sense physiological signals upon contact with a user's skin. The sensor assembly produces electrical signals representing the sensed physiological signals. A converter assembly, integrated with, and electrically connected to the sensor assembly, converts the electrical signals generated by the sensor assembly to a frequency modulated inaudible ultrasonic sound signal. The ultrasonic signal is demodulated from an aliased signal produced by undersampling.
Owner:ALIVECOR
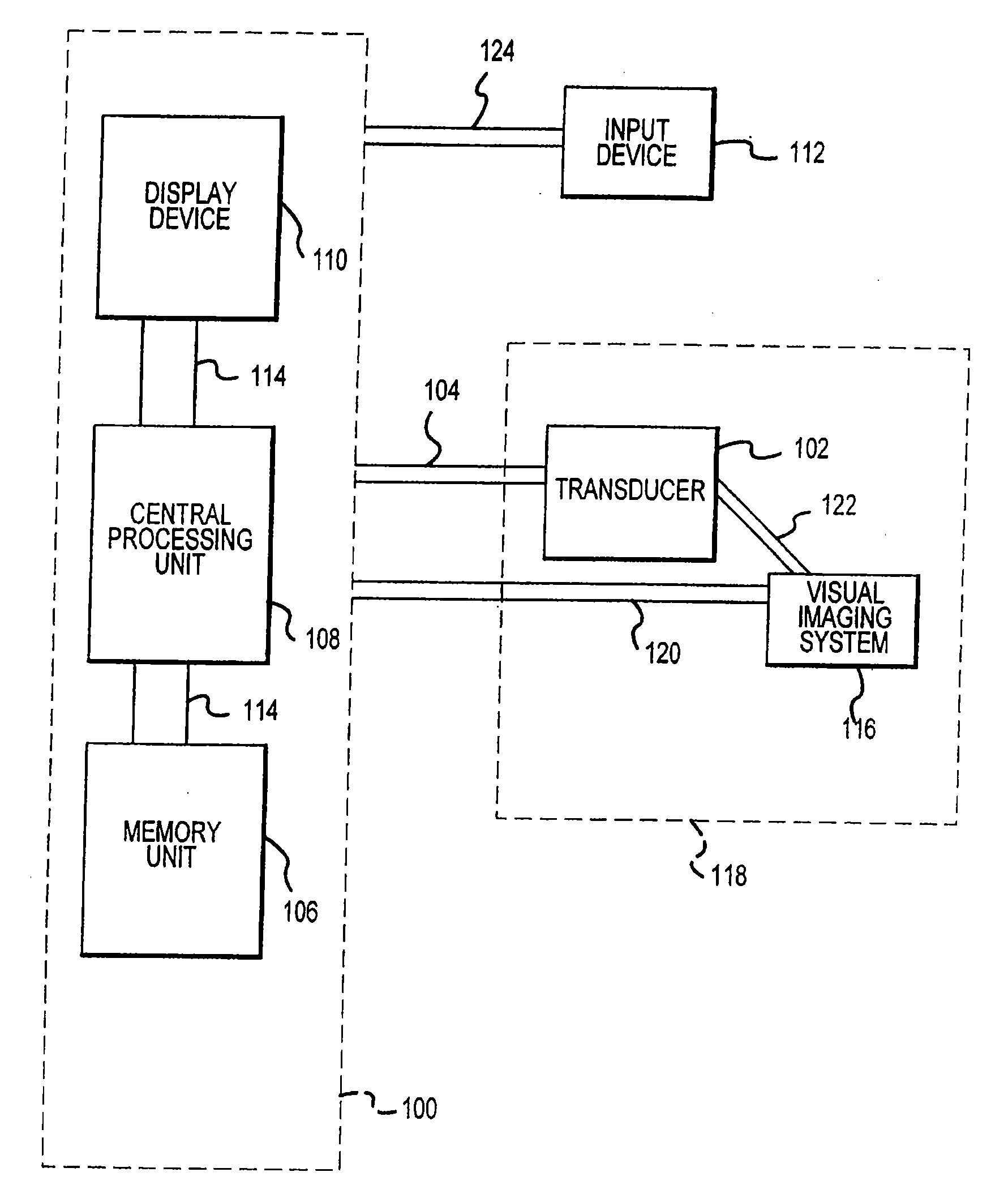
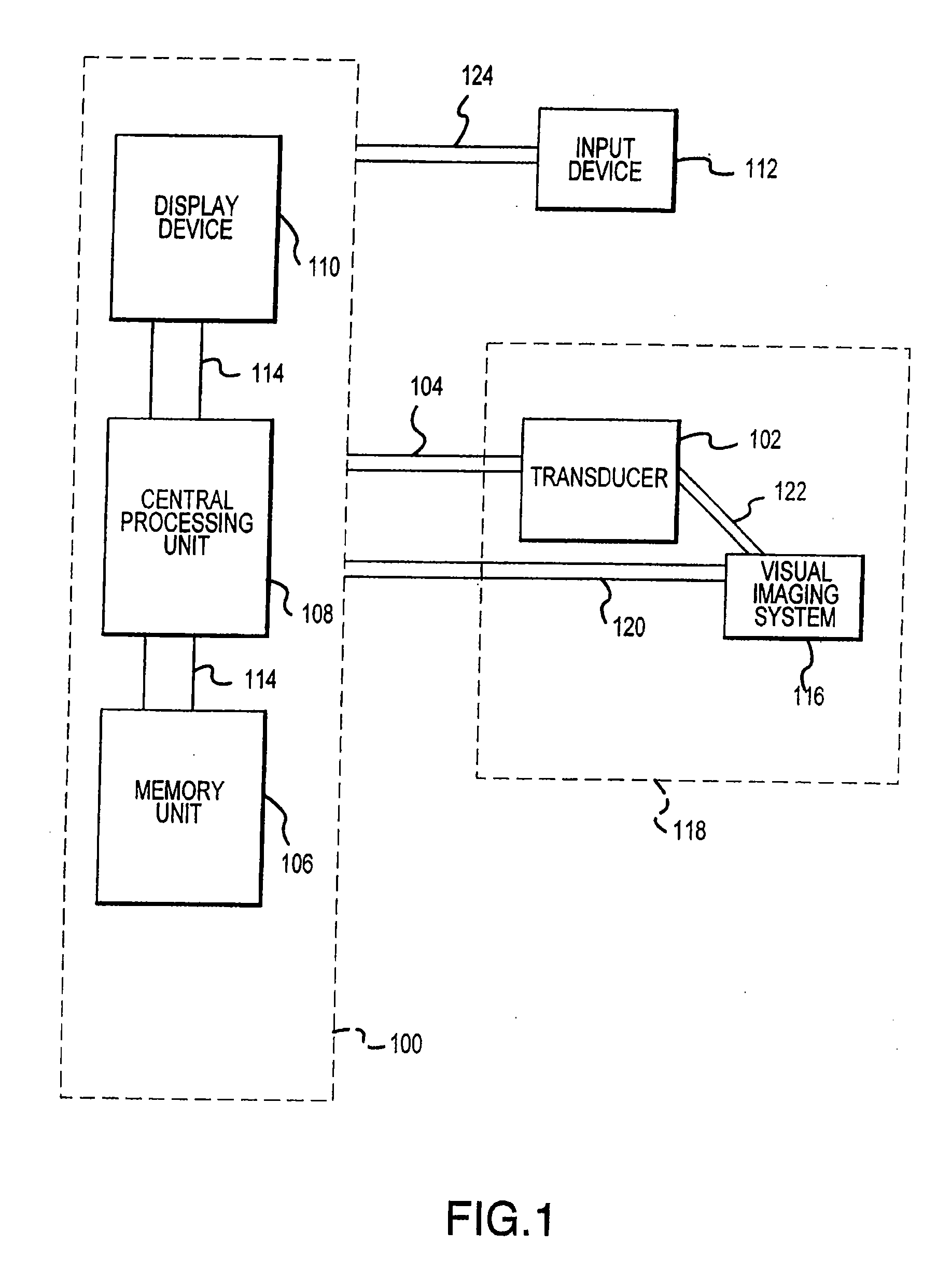
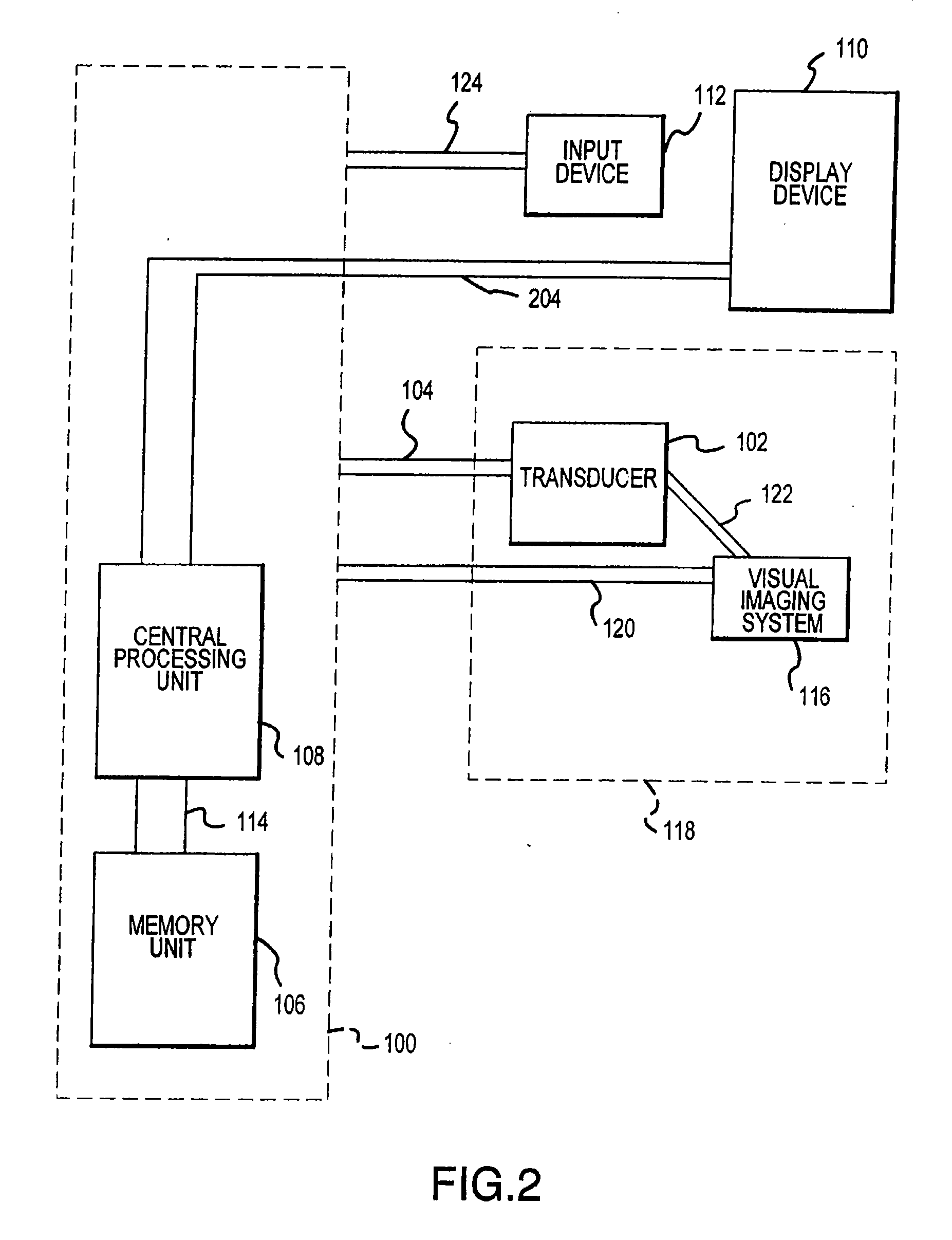
Visual imaging system for ultrasonic probe
InactiveUS20070167709A1Optimize locationEasy to determineDiagnostic probe attachmentInfrasonic diagnosticsSonificationNon invasive
A non-invasive visual imaging system is provided, wherein the imaging system procures an image of a transducer position during diagnostic or therapeutic treatment. In addition, the system suitably provides for the transducer to capture patient information, such as acoustic, temperature, or ultrasonic images. For example, an ultrasonic image captured by the transducer can be correlated, fused or otherwise combined with the corresponding positional transducer image, such that the corresponding images represent not only the location of the transducer with respect to the patient, but also the ultrasonic image of the region of interest being scanned. Further, a system is provided wherein the information relating to the transducer position on a single patient may be used to capture similar imaging planes on the same patient, or with subsequent patients. Moreover, the imaging information can be effectively utilized as a training tool for medical practitioners.
Owner:GUIDED THERAPY SYSTEMS LLC
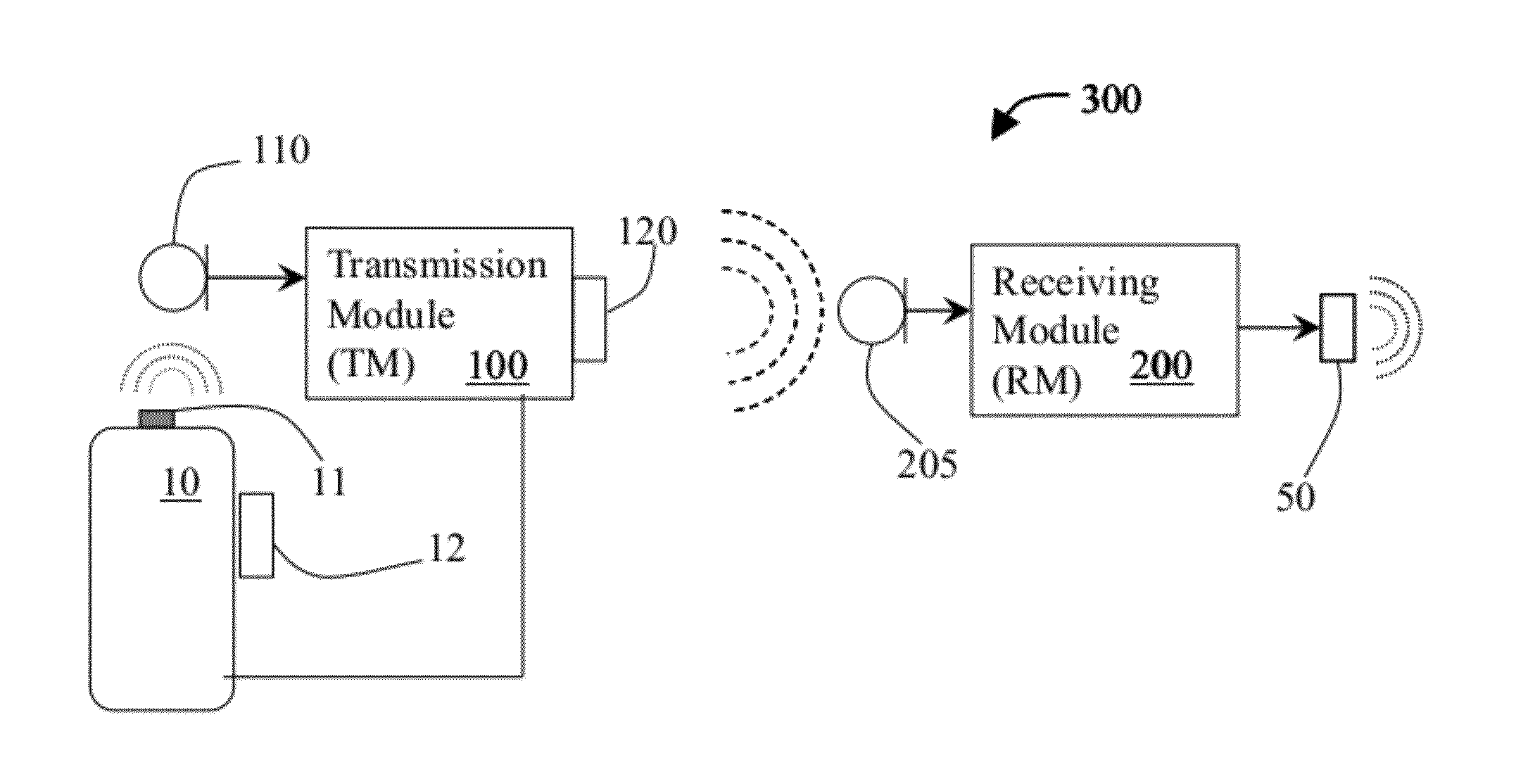
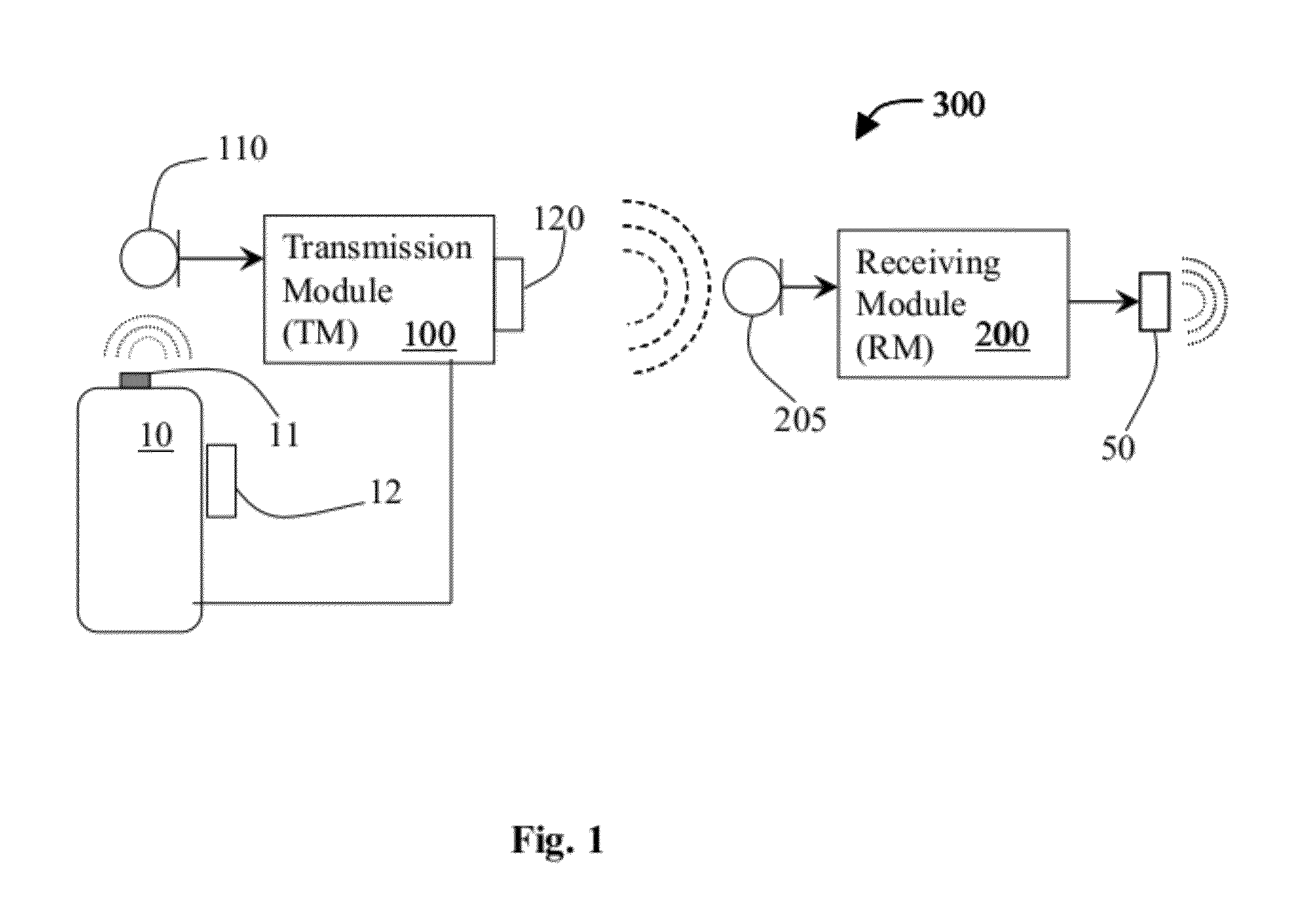
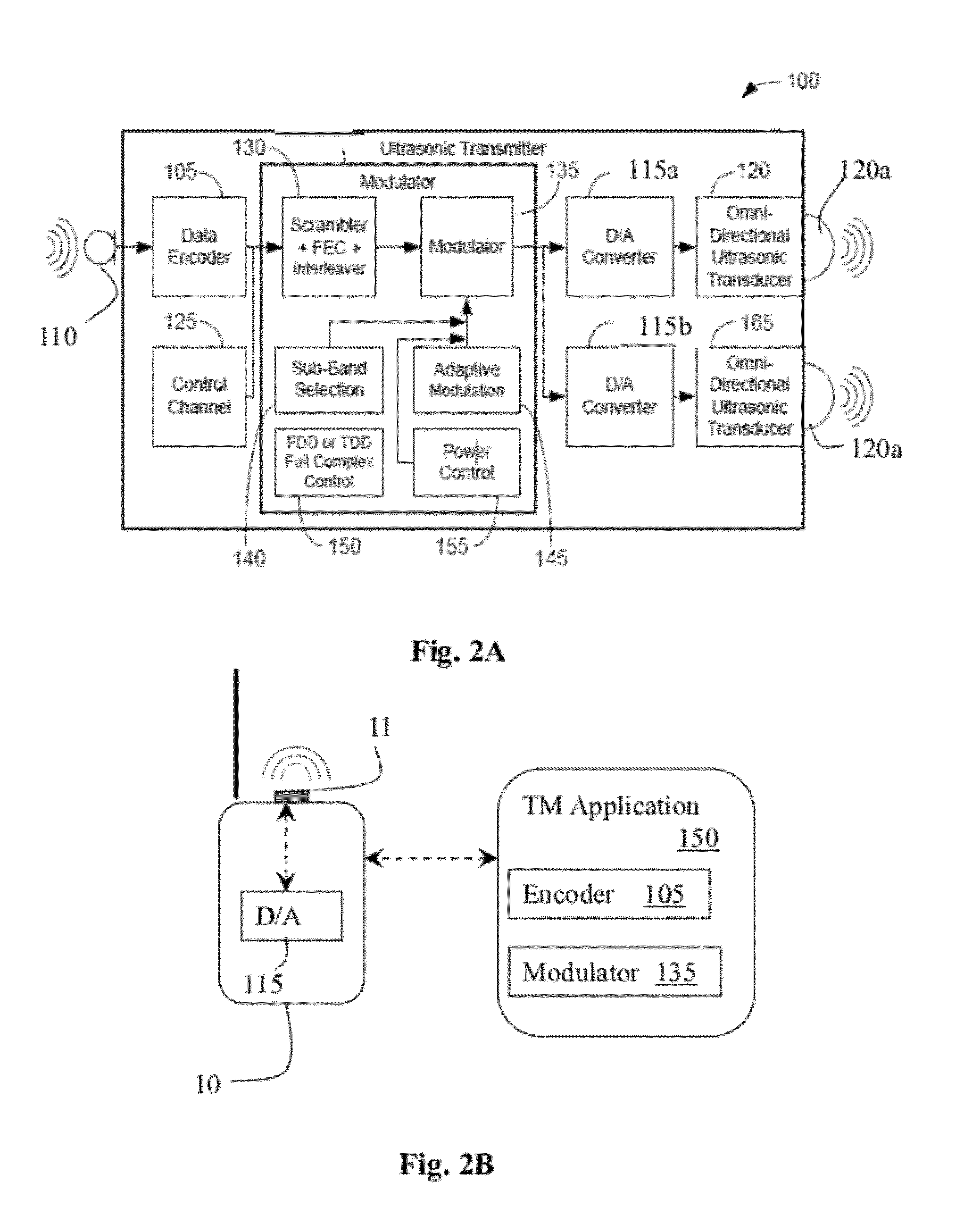
System and method for using ultrasonic communication
InactiveUS20120171963A1Sonic/ultrasonic/infrasonic transmissionRadio transmissionSonificationOutput device
A system for wireless communication using ultrasonic signals that includes a transmission module, which receives input signals from a wireless device, modifies the received input signals in a manner that converts each received input signal into a corresponding ultrasonic signal and wirelessly transmits each said ultrasonic signal over an ultrasonic link, and a receiving module, which receives the transmitted ultrasonic signals, recovers the corresponding input signals therefrom and enables outputting each respective input signal through one or more output devices. Modification of the input signals may include compressing, encoding and modulating the input signals. The input signals may be voice audio signals for enabling to use the system for supporting phone calls by enabling ultrasonic communication between for instance, a wireless headset and a mobile phone. Transmission and receiving modules may be associated with a wireless headset and mobile phone for allowing ultrasonic and optionally RF communication therebetween.
Owner:TSFATY YOSSEF