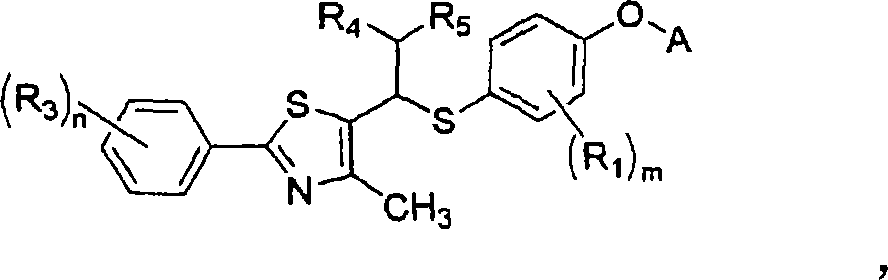
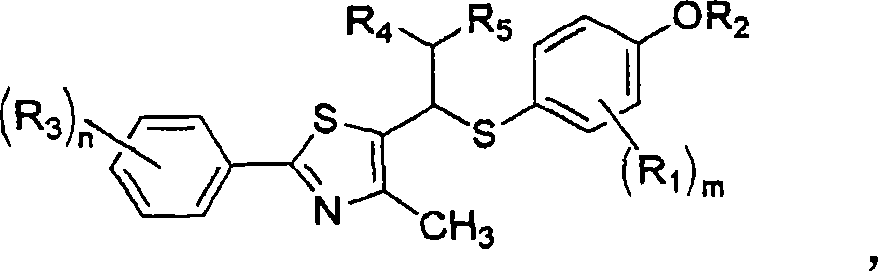
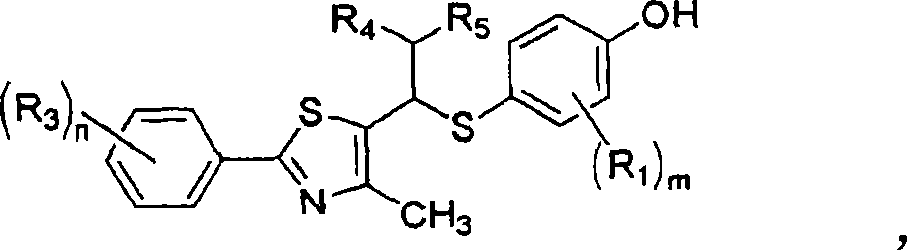
Thiazole derivatives as PPARª€ delta ligands and their manufacturing process
A derivative, thiazole technology, applied in the field of thiazole derivatives as PPARδ ligands and its manufacture, can solve the problem of PPARδ selectivity decline and other issues
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0145] Example 1: Preparation of 4-iodo-2-methyl-phenoxy-tert-butyldimethylsilane (III) [Step A]
[0146] 3.0 g (12.8 mmol) of 4-iodo-2-methylphenol and 1.74 g (25.6 mmol, 2.0 equivalents) of imidazole were completely dissolved in 45 ml of dimethylformamide. 2.12 g (14.1 mmol, 1.1 equivalents) of tert-butyldimethylchlorosilane were slowly added to the solution, and the mixture was stirred at room temperature for 4 hours. After the reaction was completed, the reaction product was extracted with aqueous ammonium chloride and ethyl acetate, and the organic layer was dried with magnesium sulfate. The residue was purified by a silica gel column, and the solvent was distilled off under reduced pressure, thereby obtaining 4.4 g (yield 98%) of the target compound.
[0147] 1 H NMR (300MHz, CDCl 3 )δ7.47(d, 1H, J=0.6Hz), 7.35(dd, 1H, J=8.4, 2.3Hz), 6.54(d, 1H, J=8.4Hz), 2.18(s, 3H), 1.03( s, 9H), 0.22 (s, 6H).
[0148] 13 C NMR (75.5MHz, CDCl 3 )δ 154.3, 139.9, 135.9, 132.3, 121...
Embodiment 2
[0149] Example 2: Preparation of 4-bromo-phenoxy-tert-butyldimethylsilane (III) [Step A]
[0150] 500mg (2.90mmol) of 4-bromophenol and 409mg (6.0mmol, 2.00eq) of imidazole were completely dissolved in dimethylformamide. 436 mg (2.90 mmol, 1.0 equivalent) of tert-butyldimethylchlorosilane was slowly added to the solution, and the mixture was stirred at room temperature for 4 hours. After the reaction was completed, the reaction product was extracted with aqueous ammonium chloride and ethyl acetate, and the organic layer was dried with magnesium sulfate. The residue was purified by a silica gel column, and the solvent was distilled off under reduced pressure, thereby obtaining 811 mg (yield 97%) of the target compound.
[0151] 1 H NMR (300MHz, CDCl 3 )δ7.32(d, 2H, J=8.8Hz), 6.72(d, 2H, J=10.0Hz), 0.98(s, 9H), 0.18(s, 6H)
[0152] 13 C NMR (75.5MHz, CDCl 3 )δ155.3, 132.7, 122.3, 114.0, 26.0, 18.6, -4.1
Embodiment 3
[0153] Example 3: 5-[4-(tert-butyldimethylsilyloxy)-3-methyl-phenylthiomethyl]-4-methyl-2-[(4-trifluoromethyl ) phenyl]-thiazole (V) preparation [step B]
[0154] 1.5g (4.32mmol) of 4-iodo-2-methyl-phenoxy-tert-butyldimethylsilane prepared in Example 1 was dissolved in 120ml of anhydrous tetrahydrofuran under a nitrogen atmosphere, and cooled to - 78°C. 2.54 ml (1.0 equiv) of tert-butyllithium (1.7 M in hexane) was slowly added to the solution. The mixture was stirred for 10 minutes, and then 138 mg (4.32 mmol, 1.0 equivalent) of sulfur in solid phase was added thereto at the same temperature in one portion. Allow the mixture to react for 40 minutes until its temperature reaches 15° C., then slowly add 1.26 g (4.32 mmol, 1.0 equivalents) of 5-chloromethyl-4-methyl represented by formula III dissolved in 10 ml of anhydrous THF -2-[(4-Trifluoromethyl)phenyl]-thiazole. After the reaction was continued for about 1 hour, the reaction was quenched with aqueous ammonium chloride,...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap