N-acylhomoserine lactonas, production method thereof and special recombinant bacterium
A technology of recombinant bacteria and amino acids, applied in the field of genetic engineering, to achieve high-efficiency expression
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0044] Example 1 Acquisition of genetically engineered recombinant bacteria
[0045] 1. Cloning of N-acyl homoserine lactone hydrolase gene
[0046] The genome of Bacillus sp. B546 CGMCC No 3228 was used as a template, and specific primers BT1 (5'-GCGGAATTCATGACAGTAAAGAAGCTTTATTTCG-3') and BT2 (5'-ATAGCGGCCGCCTATATATACTCTGGGAACAC-3') were used (the underlined part is EcoR I and Not, respectively). I enzyme cleavage site), and expanded according to the following conditions: 94 °C for 5 min; 94 °C for 1 min, 58 °C for 30 s, 72 °C for 1 min, a total of 35 cycles; 72 °C for 10 min extension.
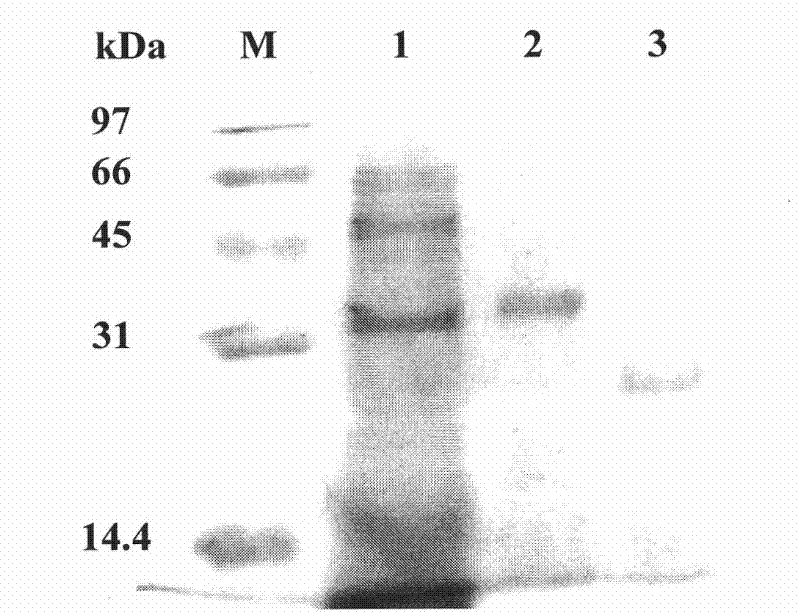
[0047] Amplify a fragment of about 750bp (see figure 1 In 2), the connected pEASY-T3 vector after the fragment recovery was transformed into E.coli TOP 10 competent cells, and the positive clone was picked for sequencing. The sequence shown from position 1 to position 753 at the 5' end, wherein the coding region is from position 1 to position 750 at the 5' end, encodes the protein shown in...
Embodiment 2
[0060] Example 2 High-efficiency expression of AiiA-B546 in P. pastoris AiiA-B546
[0061] 1. Expression and analysis of engineered bacteria at the shake flask level
[0062] The above engineering bacteria and empty vector strain controls were re-inoculated into a 1000mL conical flask with 200mL BMGY medium, cultured at 30°C, 250-280rpm shaker for 48h, centrifuged at 3000×g for 5min, and discarded the supernatant (try to remove as much as possible) , add 200 mL of BMMY medium containing 0.5% methanol to the precipitated cells, and re-induce the culture at 25°C and 250-280 rpm. In order to compensate for the loss of methanol, add methanol solution to the bacterial solution every 12 hours to make the methanol in the bacterial solution. The concentration was kept at 0.5% until induction for 72 h, centrifuged at 12,000 × g for 5 min, and the enzyme activity of the supernatant was determined according to the method provided in Example 1.
[0063] The recombinant E. coli was inocul...
Embodiment 3
[0070] Example 3 Study on the enzymatic properties of yeast expressing AiiA-B546
[0071] Enzymatic reaction system: Except for step 4, in the remaining steps, 3-OXO-C8-HSL was used as the substrate, and the enzymatic properties of N-acyl homoserine lactonase were determined by the active plate method.
[0072] The enzyme solution used in this example is the AiiA-B546 enzyme solution purified in Example 2.
[0073] 1. The pH and temperature adaptability of AiiA-B546.
[0074] The purified AiiA-B546 was subjected to enzymatic reaction at different pH to determine its optimum pH. The buffers used were 50 mM acetate-sodium acetate buffer (pH 5.0 and 5.8), 0.2 M sodium dihydrogen phosphate-disodium hydrogen phosphate buffer (pH 5.8, 6.0, 7.0 and 8.0), 50 mM Tris-HCl buffer (pH 8.0 and 9.0). The pH suitability results of purified AiiA-B546 in different pH buffer systems at 25°C are as follows Figure 4 a (with the highest enzyme activity as 100%, and the ratio of the enzyme act...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More