Kit and detection method for detecting hepatitis B virus core gene promoter base mutation
A technology of hepatitis B virus and kit, applied in the field of molecular biology, can solve problems such as difficulty in popularization and application, too strict requirements on hybridization temperature, etc., and achieve high accuracy
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
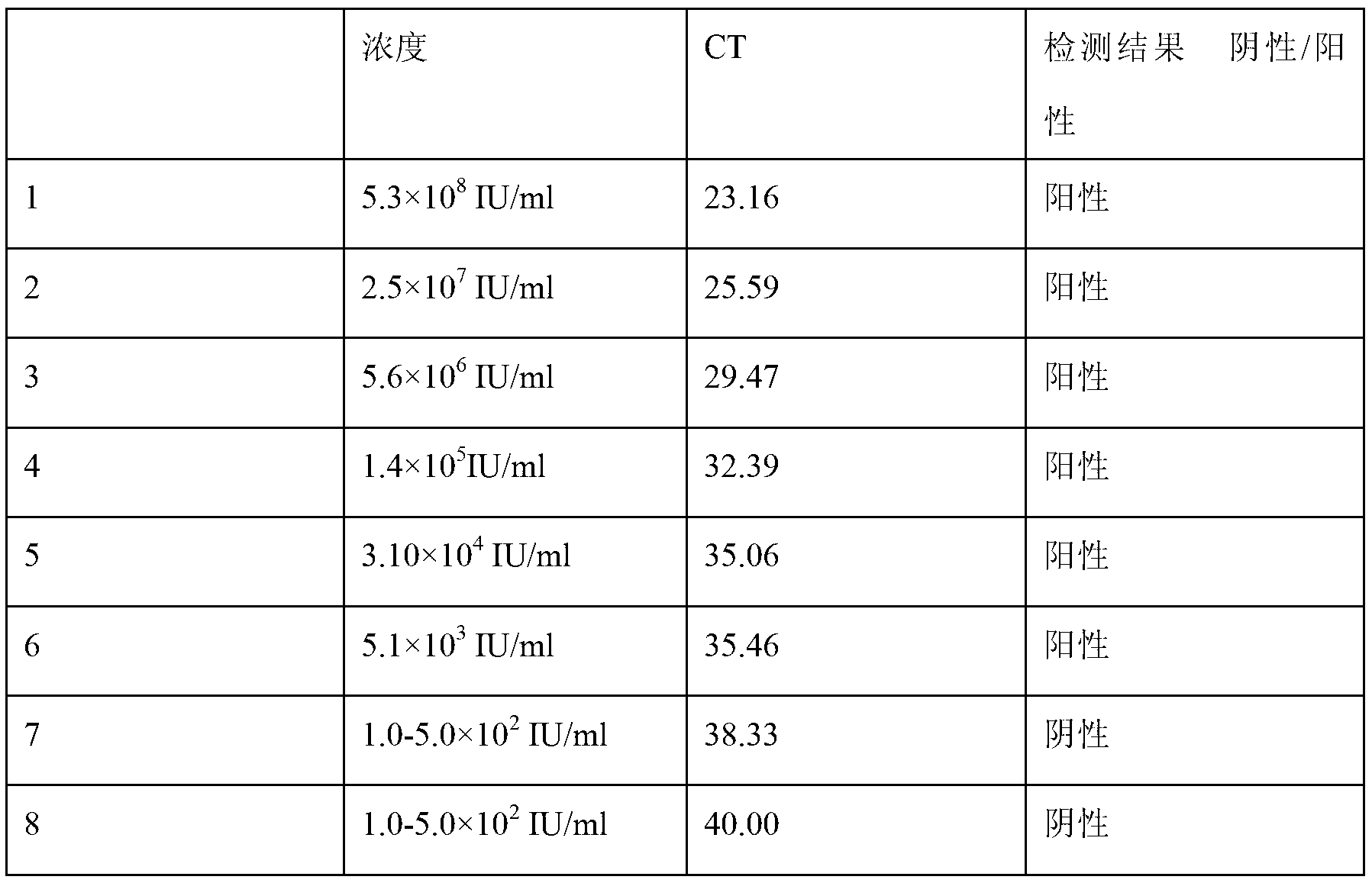
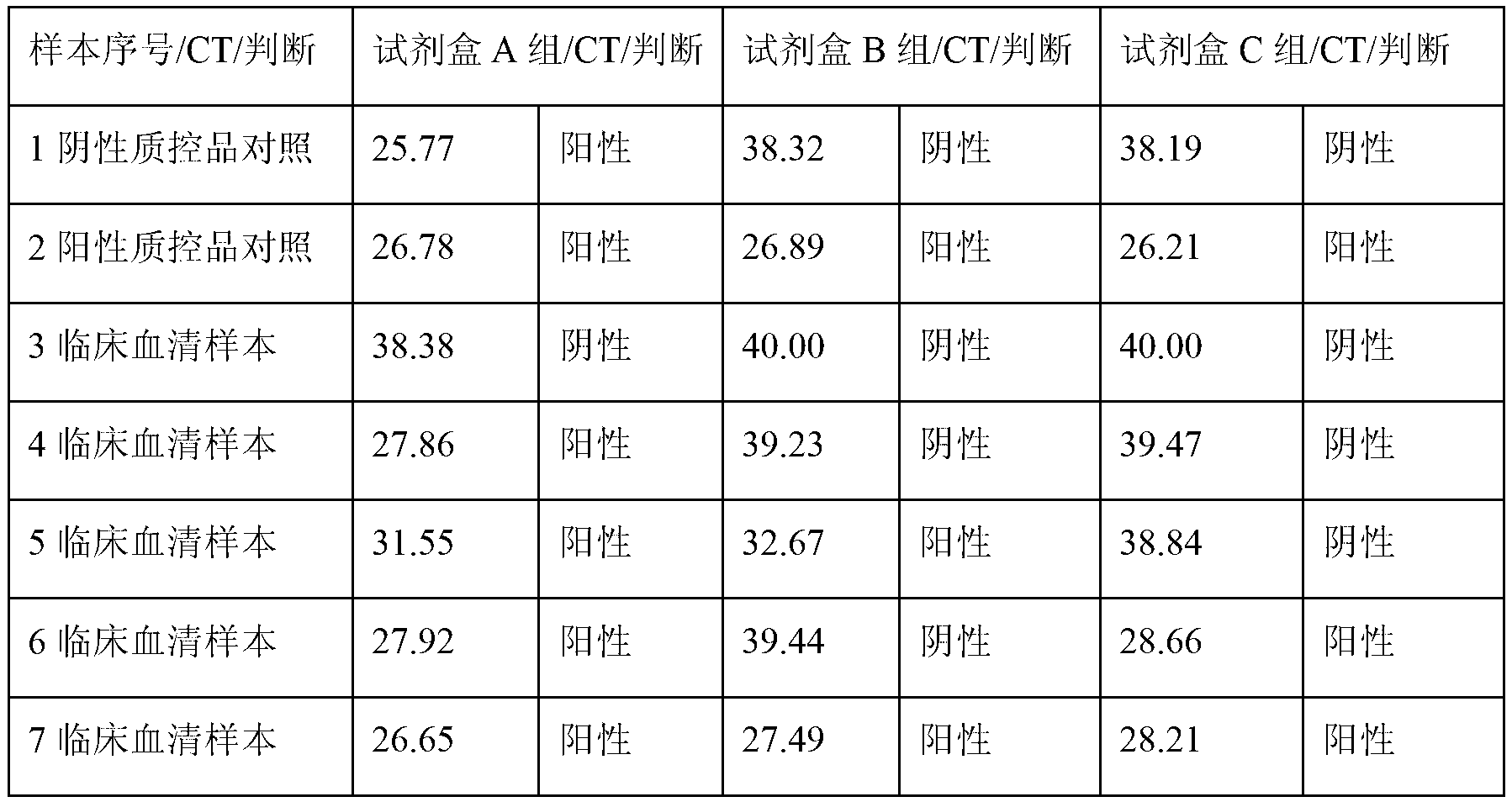
[0060] Embodiment 1: the detection reagent of group A of PCR of hepatitis B virus core gene promoter DNA in the kit, to the positive quality control material of application national regulation HBV standard substance, people's negative HBV serum, negative quality control substance and different dilution concentration, carry out negative And Positive Cutoff Test, Minimum Detection Concentration Test.
[0061] Both wild-type and mutant types of hepatitis B virus bases can be successfully amplified by PCR, including the following components:
[0062] Human negative HBV serum: no hepatitis B virus.
[0063] Negative quality control: No A1762T and Gl764A base variant wild-type gene clone strain and A1762T and G1764A base variant gene clone strain, wild-type gene clone strain (Escherichia coli DH5α) plasmid DNA, nucleotide sequence Yes:
[0064] gtttaaagac tgggaggagt tgggggagga gattaggtta aaggtctttg tactaggagg ctgtaggcat aaattggtct gttcaccagc accatgcaac tttttcacct ctgcctaatc atctca...
Embodiment 2
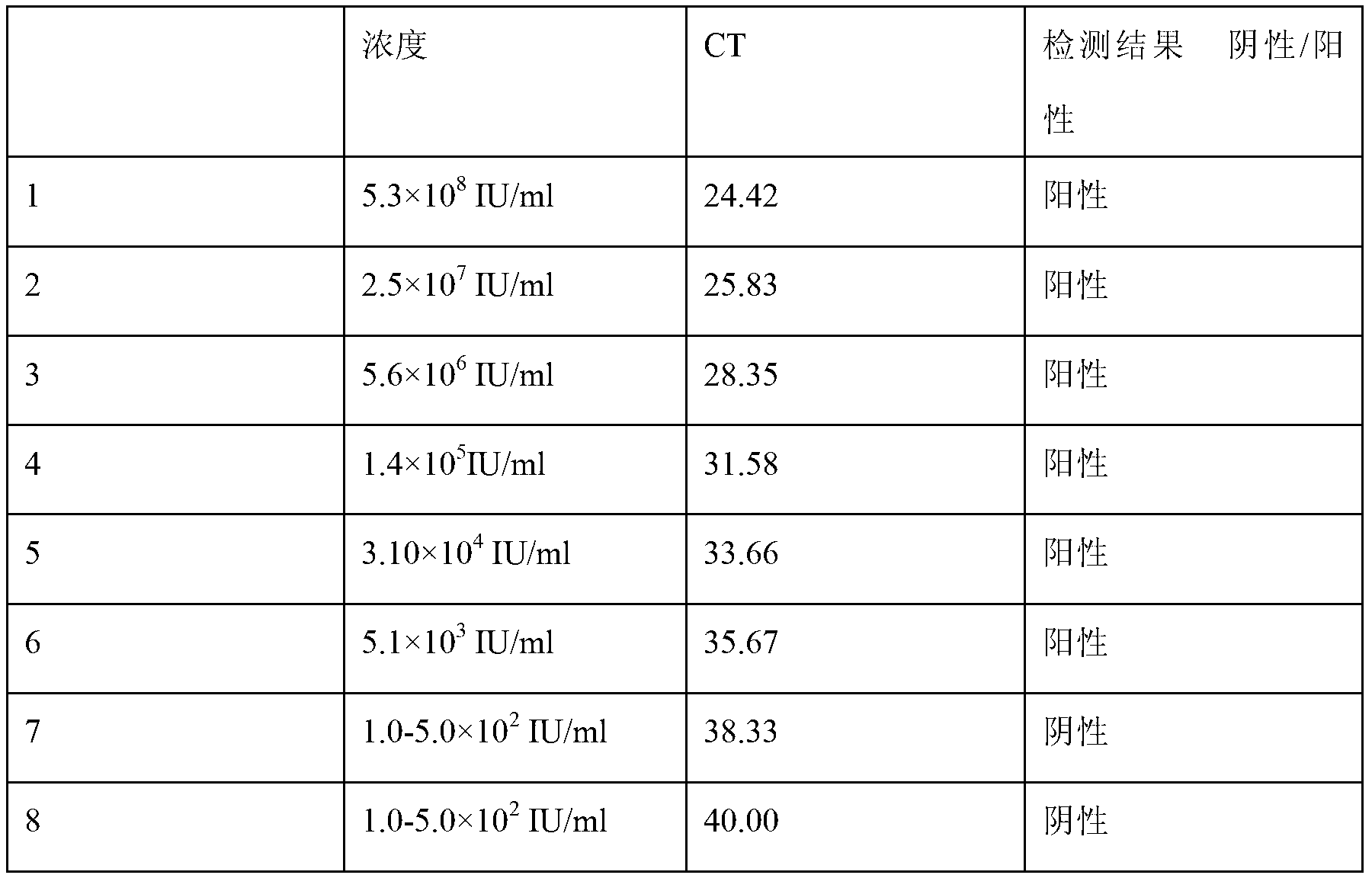
[0088] Embodiment 2: A group of detection reagents of hepatitis B virus core gene promoter DNA in the kit can carry out PCR amplification smoothly to the A1762T mutant type of hepatitis B virus base, including the following components:
[0089] The negative quality control substance that detection test is used: with embodiment 1.
[0090] The positive quality control substance that detection test is used: with embodiment 1.
[0091] PCR reaction solution B: PCR buffer (1×), TaqDNA polymerase 1.5u, dNTP0.3μmol / L, and:
[0092] HBV-B1 upstream primer 5′-tgggg gagga gatta ggtta at-3′ 0.3 μmol / L
[0093] HBV-B2 upstream antisense primer 5′-tttaa cctaa tctcc tcccc ca-3′ 0.03 μmol / L
[0094] HBV-D common downstream primer 5′-agcca cccaa ggcac agctt g-3′; 0.3 μmol / L
[0095] HBV-Y fluorescent probe: 5′-FAM-tgtag gcata aattg gtctg tt-TAMARA-3′; 0.2μmol / L
[0096] Specific implementation steps:
[0097] 1) Sample DNA treatment: Take the negative quality control and positive qualit...
Embodiment 3
[0107] Embodiment 3: a group of detection reagents of hepatitis B virus core gene promoter DNA in the kit can carry out PCR amplification smoothly to the Gl764A mutant type of hepatitis B virus base, including the following components:
[0108] The negative quality control substance that detection test is used: with embodiment 1.
[0109] The positive quality control substance that detection test is used: with embodiment 1.
[0110] PCR reaction solution C: PCR buffer (1×), TaqDNA polymerase 1.5u, dNTP0.3μmol / L, and:
[0111] HBV-C1 upstream primer 5′-ggagg agatt aggtt aaaga-3′0.3μmol / L
[0112] HBV-C2 upstream antisense primer 5′-ccttt aacct aatct cctcc-3′0.03μmol / L
[0113] HBV-D common downstream primer 5′-agcca cccaa ggcac agctt g-3′0.3μmol / L
[0114] HBV-Y fluorescent probe: 5′-FAM-tgtag gcata aattg gtctg tt-TAMARA-3′0.2μmol / L
[0115] Specific implementation steps:
[0116] 1) Sample DNA treatment: Take the negative quality control and positive quality control, dilu...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap