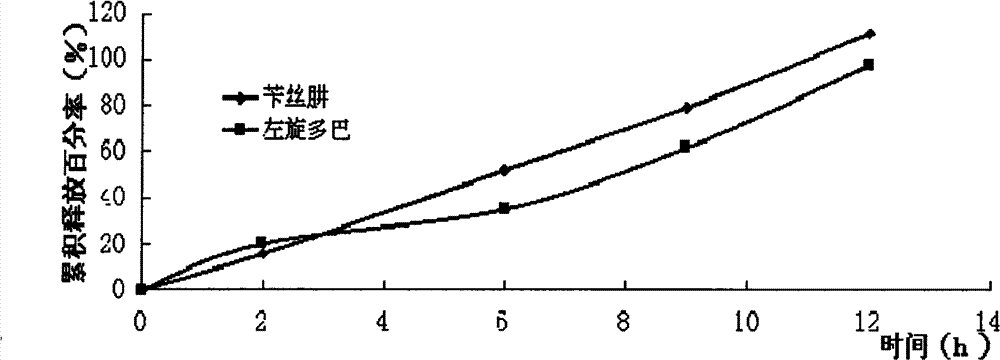
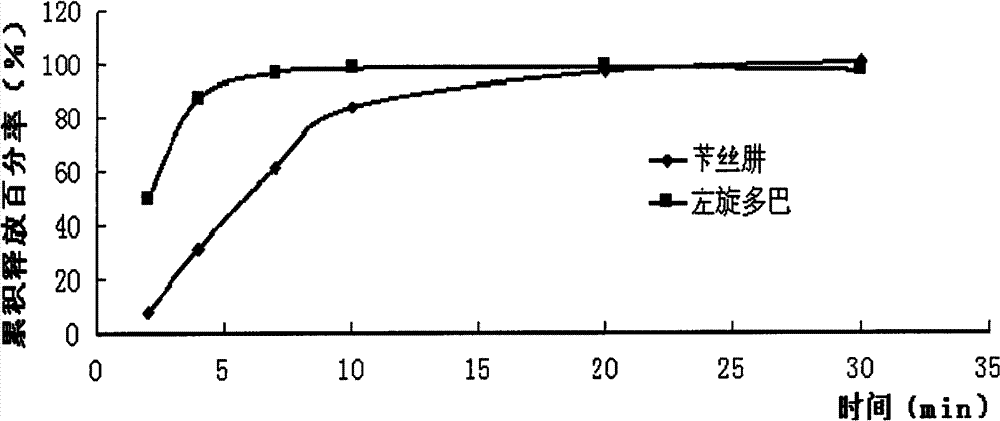
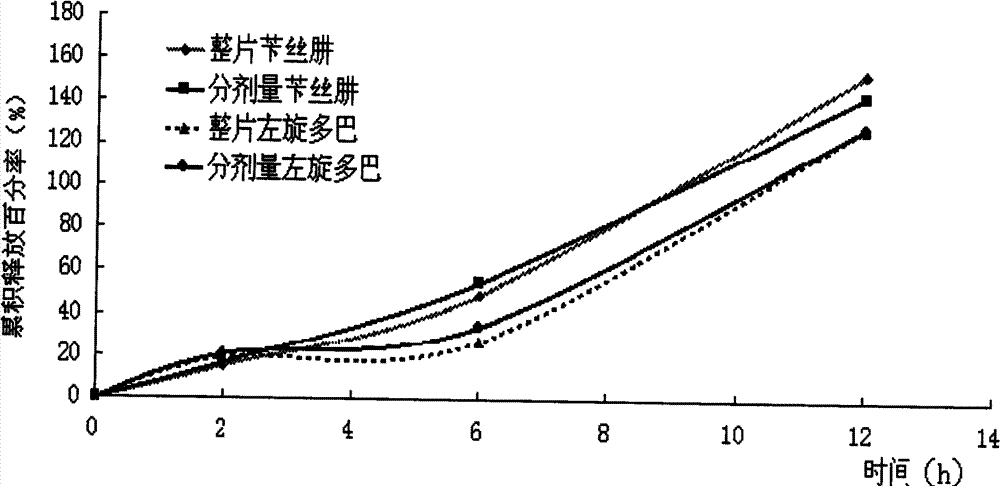
Compound Levodopa Microcapsule Floating Tablets
A technology of levodopa microcapsules and floating tablets, which is applied in the field of medicine, can solve the problems of low bioavailability of ordinary tablets, complicated drug regimens, and many times of administration, etc., so as to prolong the residence time in the microcapsules and improve bioavailability The effect of high degree and flexible clinical application
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0025] Example 1: The following methods are adopted to prepare compound levodopa microcapsule floating tablets:
[0026] The preparation of microcapsules includes the following steps:
[0027] 1) Levodopa is passed through a 400-mesh sieve for use.
[0028] 2) Weigh:
[0029] Levodopa-3g;
[0030] Ethyl cellulose with a viscosity of 10mPa·s——1.8g;
[0031] Ethyl cellulose with a viscosity of 20 mPa·s-7.2 g.
[0032] Ethyl cellulose of different viscosities was dissolved in 112.5 mL of methylene chloride, and the concentration of the capsule material after dissolution was 8%. After the ethyl cellulose is completely dissolved, add levodopa, stir at 400r / min for 30min in a 10℃ water bath, and add the resulting suspension to 1125mL aqueous solution containing 0.01~0.08g sodium lauryl sulfate at room temperature 400 / min. Min stirring for 1 hour, filtered under reduced pressure to obtain the microcapsule intermediate. The intermediate was washed with diluted hydrochloric acid at a ratio of 1...
Embodiment 2
[0048] Embodiment 2: The difference from embodiment 1 is:
[0049] Weigh in step A:
[0050] Levodopa-45.6g;
[0051] Ethyl cellulose with a viscosity of 10mPa·s——1.2g;
[0052] Ethyl cellulose with a viscosity of 20 mPa·s-1.2 g.
[0053] And ethyl cellulose of different viscosities was dissolved in 225 mL of methylene chloride, and the concentration of the capsule material after dissolution was 9%. In the end, 48 g of levodopa microcapsules were obtained.
[0054] Weigh in step B:
[0055] Benserazide hydrochloride-6.64g;
[0056] Vitamin C-1.36g.
[0057] Polyvinylpyrrolidone K30——3.6g;
[0058] Ethyl cellulose with a viscosity of 10 mPa·s-0.4 g.
[0059] Finally, 12 g of benserazide hydrochloride solid dispersion was obtained.
[0060] Weigh in step C:
[0061] Stearic acid-30g;
[0062] Acrylic resin IV-6.67g;
[0063] Hydroxypropyl methylcellulose K4M-3.33g.
[0064] Finally, 40g of auxiliary materials are obtained.
[0065] The levodopa microcapsules, benserazide hydrochloride solid dispersi...
Embodiment 3
[0066] Embodiment 3: The difference from embodiment 1 is:
[0067] Weigh in step A:
[0068] Levodopa-8g;
[0069] Ethyl cellulose with a viscosity of 10mPa·s——2g;
[0070] Ethyl cellulose with a viscosity of 20mPa·s-6g.
[0071] And ethyl cellulose of different viscosities was dissolved in 225 mL of methylene chloride, and the concentration of the capsule material after dissolution was 9%. In the end, a total of 16 g of levodopa microcapsules were obtained.
[0072] Weigh in step B:
[0073] Benserazide hydrochloride-2.14g;
[0074] Vitamin C-1.36g.
[0075] Polyvinylpyrrolidone K30——1.11g;
[0076] Ethyl cellulose with a viscosity of 10 mPa·s-0.22 g.
[0077] Finally, 4 g of benserazide hydrochloride solid dispersion was obtained.
[0078] Weigh in step C:
[0079] Stearic acid-20g;
[0080] Acrylic resin IV-40g;
[0081] Hydroxypropyl methylcellulose K4M-20g.
[0082] Finally, 80g of auxiliary materials are obtained.
[0083] The levodopa microcapsules, benserazide hydrochloride solid dispersio...
PUM
| Property | Measurement | Unit |
|---|---|---|
| hardness | aaaaa | aaaaa |
| viscosity | aaaaa | aaaaa |
| viscosity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap