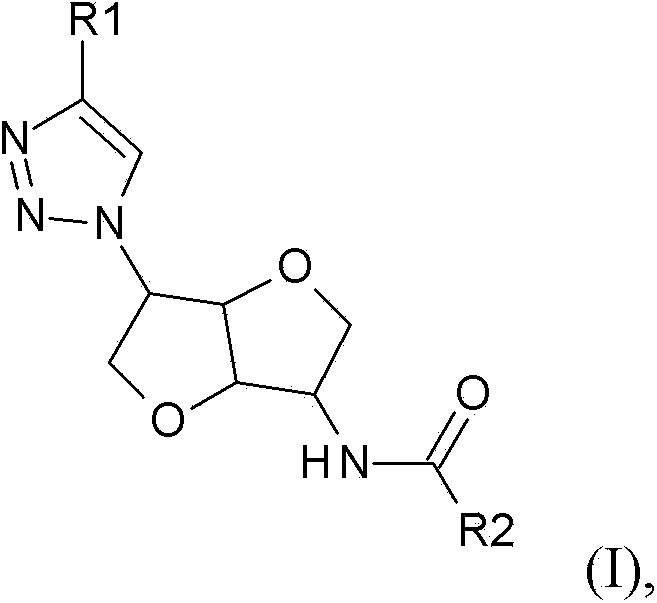
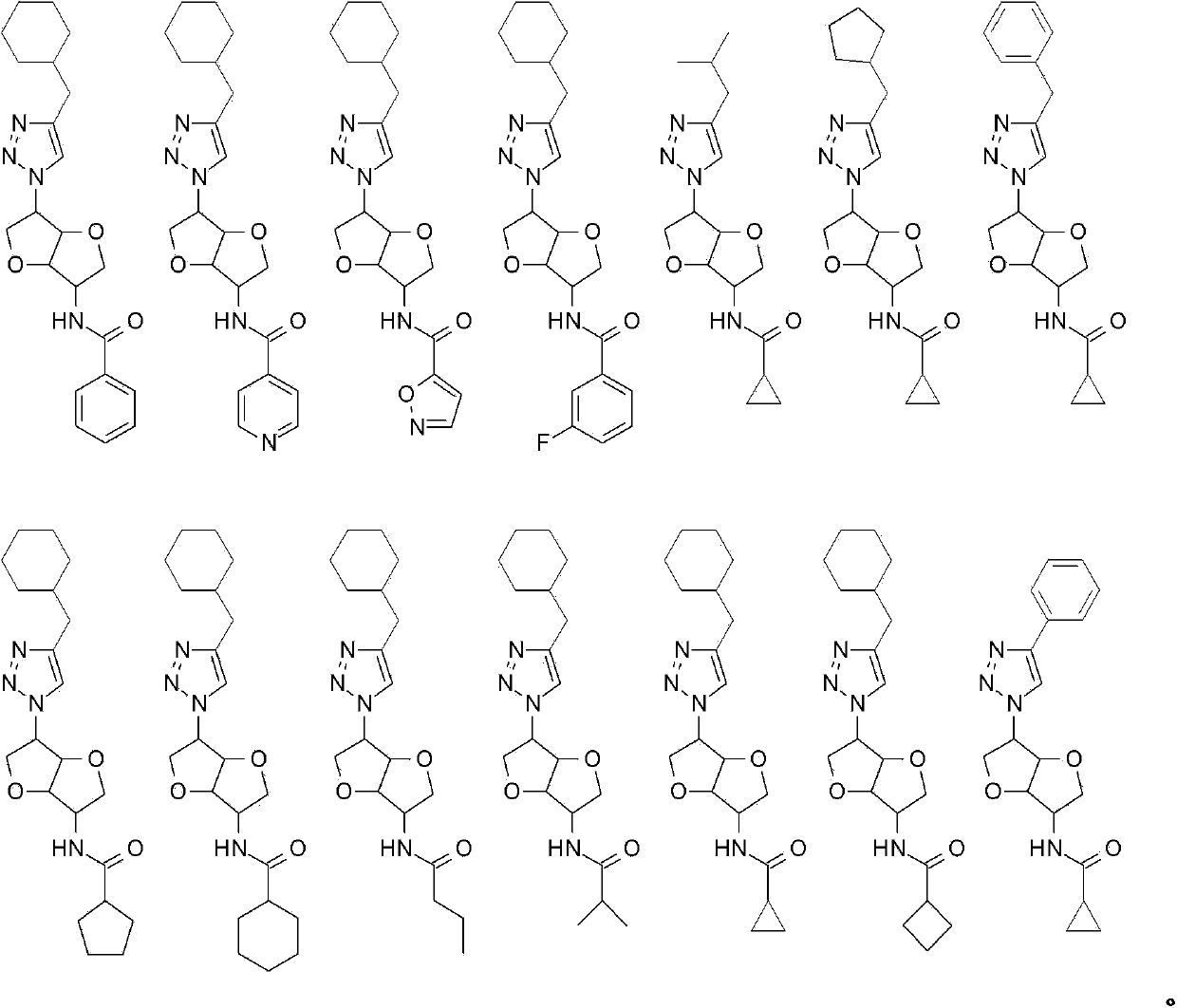
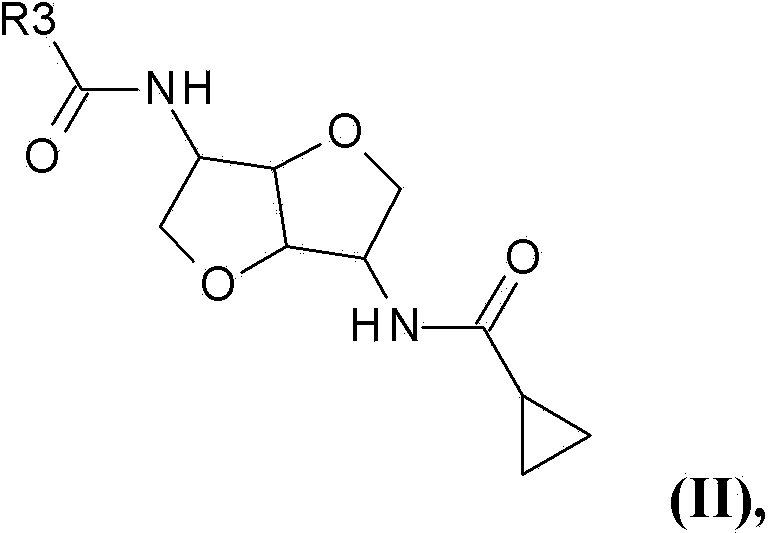
Chemosensory receptor ligand-based therapies
A chemical sensing and ligand technology, applied in organic chemistry, chemical instruments and methods, preparation of organic compounds, etc., can solve the problems of unclear state of acid receptor recognition and no testing
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1a
[0989] Example 1a: Upper GI (Upper GI) administration of a chemosensory receptor ligand in diabetic rats.
[0990] Many well-established rat models exist for evaluating the efficacy of diabetes treatments. As detailed in the Examples below, a single chemosensory receptor ligand (eg, sweet) can be tested for diabetes treatment in an established rat model of diabetes.
[0991] Diabetic rats and Wistar rats are selected and chemosensory receptor ligands such as sucralose are administered to treat diabetes. Animals are grouped by dose and escalating doses (range 0.01-100 mg / kg) are used. Chemosensory receptor ligands were instilled into the animals via a silastic tube inserted orally into the duodenum of the lightly anesthetized animals.
[0992] Optionally, dipeptidyl peptidase IV (DPP IV) is inhibited in a given group of the test animals, or in all groups, to prevent degradation of the hormone of interest by endogenous peptidases. DPP IV inhibition was achieved by coadministr...
Embodiment 1b
[1000] Example 1b: Optionally, in the experimental procedure described above, the chemosensory receptor ligand is administered together with a cognate metabolite if no metabolic change occurs. For example in an alternative experimental approach, sucralose is administered together with glucose. Escalating doses of the ligand may be administered relative to a fixed dose of the cognate metabolite; and vice versa.
Embodiment 1c
[1001] Example 1c: Optionally, the above-described experimental method was performed using industry standard diet-induced obese rats and applicable controls (healthy rats). Parameters unique to this obesity system were modified based on known standard experimental conditions. Samples were collected and hormone assays were performed as described above. Other hormones such as glucagon and uroguanylin may be measured.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com