Optically active 2-hydroxytetrahydrothienopyridine derivatives and preparation methods and uses thereof
A technology of hydroxytetrahydrothiazide and phenopyridine, which is applied in the field of medicine, can solve the problems of loss of enzyme activity, reduction of oral bioavailability of clopidogrel, high genetic mutation rate, etc., and achieve the goal of increasing oral bioavailability and inhibiting platelet aggregation Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0058] Embodiment 1: the preparation of formula V compound
[0059] Synthetic scheme
[0060]
[0061] Step 1: Synthesis of (R)-(2-chloro-phenyl)-hydroxy-acetic acid methyl ester (2)
[0062]
[0063] To a stirred solution of compound 1 (18.6 g, 0.1 mol) in methanol (100 mL) was added concentrated sulfuric acid (2 mL). The mixture was then heated at reflux for 3 h and excess methanol was removed under vacuum. The oily residue was taken up in 200 mL of dichloromethane, then washed with 10% potassium carbonate in water (240 mL), the organic solvent was extracted, dried, and then concentrated in vacuo to give compound 2 (19 g, 95%) as a colorless of oil.
[0064] 1 HNMR: (Y0859-04567-023, CDCl 3 ,400MHz)δ7.52-7.50(m,1H),7.45-7.43(m,1H),7.39-7.32(m,2H),6.36-6.34(d,J=6.4Hz,1H),5.43-5.42( d,J=6.4Hz,1H),3.62(s,3H).
[0065] Step 2: Synthesis of (R)-(2-chloro-phenyl)-(4-nitro-benzenesulfonyloxy)-acetic acid methyl ester (4)
[0066]
[0067] at 0°C and N 2 Next, to...
Embodiment 2
[0083] Embodiment 2: the preparation of formula VI compound
[0084] Preparation scheme:
[0085]
[0086] Step 1: (S)-(2-Chloro-phenyl)-[2-(di-tert-butoxy-phosphoryloxymethoxy)-6,7-dihydro-4H-thiophene[3, Synthesis of 2-c]pyridin-5-yl]-acetic acid methyl ester (10)
[0087]
[0088] at -78°C and N 2 Add LHMDS (4.23 mL, 1.0 M , 2.85mmol), then the mixture was stirred at room temperature for 30 minutes, then compound 9 (540mg, 2.11mmol) was added, and stirred for another 10 hours. use NH 4 The reaction was quenched with Cl solution and extracted with EA. with NaHCO 3 solution and brine, washed the organic layer with anhydrous Na 2 SO 4 Dry and concentrate to dryness. The residue was purified with silica gel column (PE:EA=50:1-3:1) to give compound 10 (52 mg, 7%) as a white solid.
[0089] 1 HNMR: (CDCl 3 ,400MHz)δ7.69-7.66(m,1H),7.41-7.39(m,1H),7.30-7.24(m,2H),6.11(s,1H),5.44(s,1H),5.41(s, 1H),4.89(s,1H),3.72(s,3H),3.62-3.58(m,1H),3.49-3.46(m,1H),2.87-2.85(...
Embodiment 3
[0096] Embodiment 3: Pharmacokinetic test in vivo
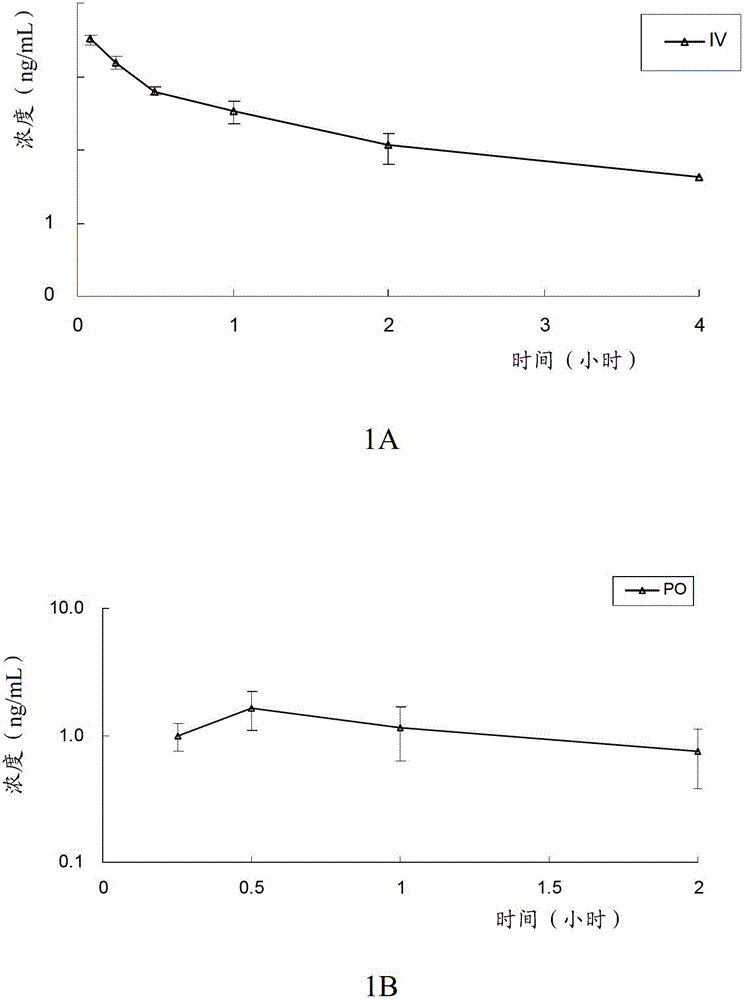
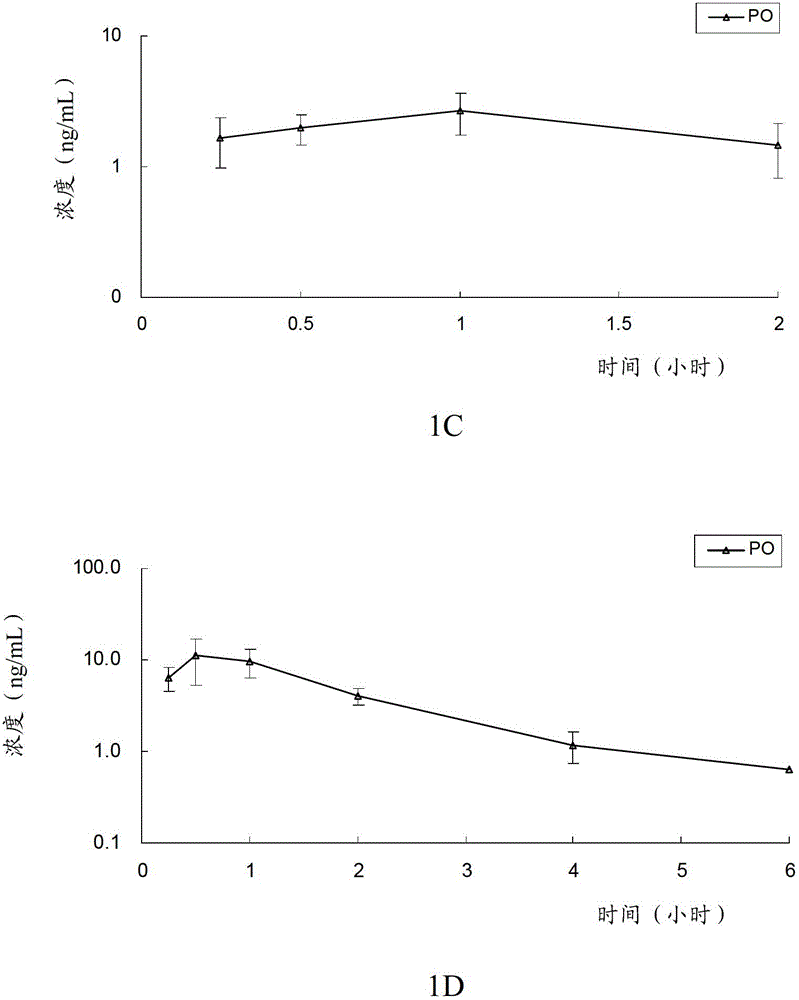
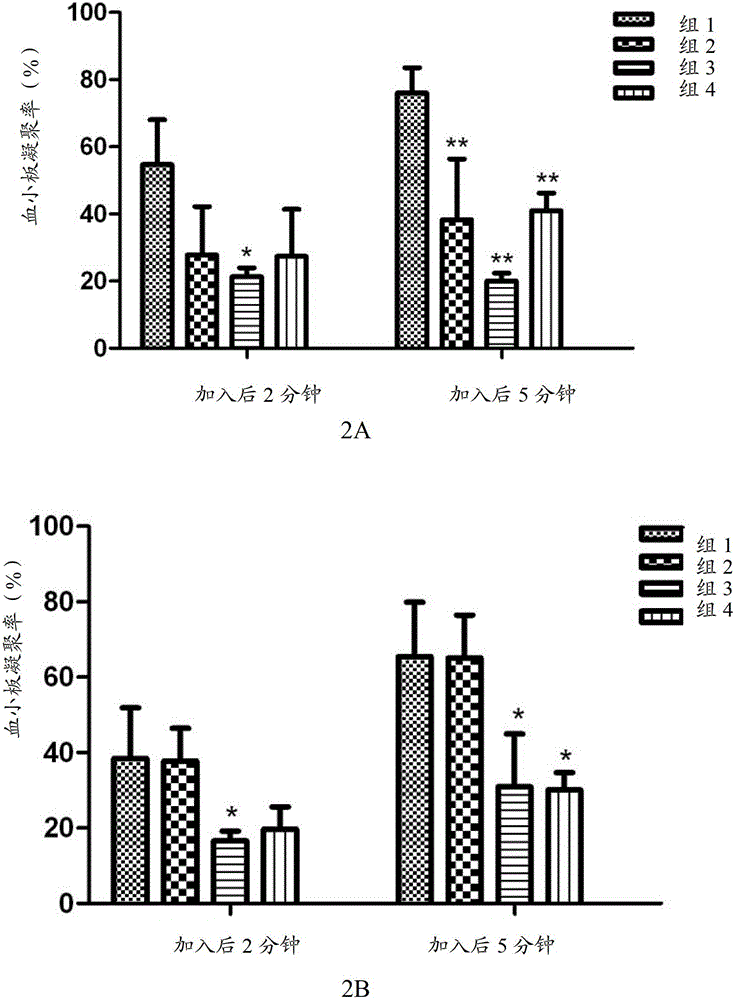
[0097] The in vivo pharmacokinetics of the compound of formula V, the compound of formula VI, clopidogrel (formula VII) and the first metabolite of clopidogrel (formula VIII) of the present invention were detected. Specifically, the compounds of formula V, compounds of formula VI and clopidogrel and their first metabolites were given orally or intravenously to rats, and the pharmacokinetic characteristics of the compounds of the present invention and clopidogrel in rats were evaluated, and the The conversion of the compound of the present invention into the first metabolite in vivo, and by measuring the blood concentration of the first metabolite (formula VIII) in rats at a certain time, compare the bioavailability of the compound of the present invention and clopidogrel Spend.
[0098] The experimental animals were male SD rats, aged 6 to 8 weeks, weighing 190-215 grams, purchased from Beijing Weili Tonghua Experimental A...
PUM
| Property | Measurement | Unit |
|---|---|---|
| wavelength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap