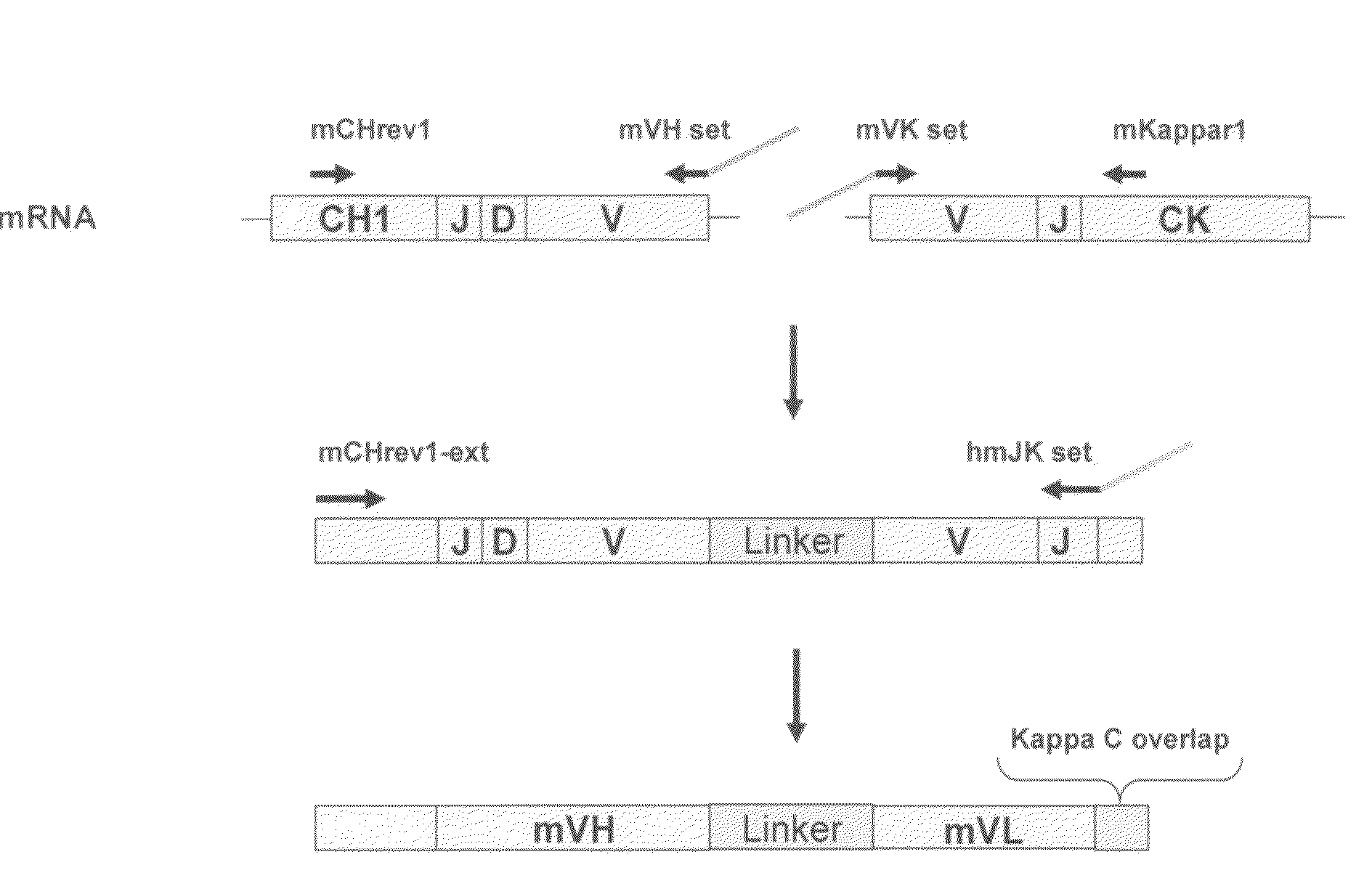
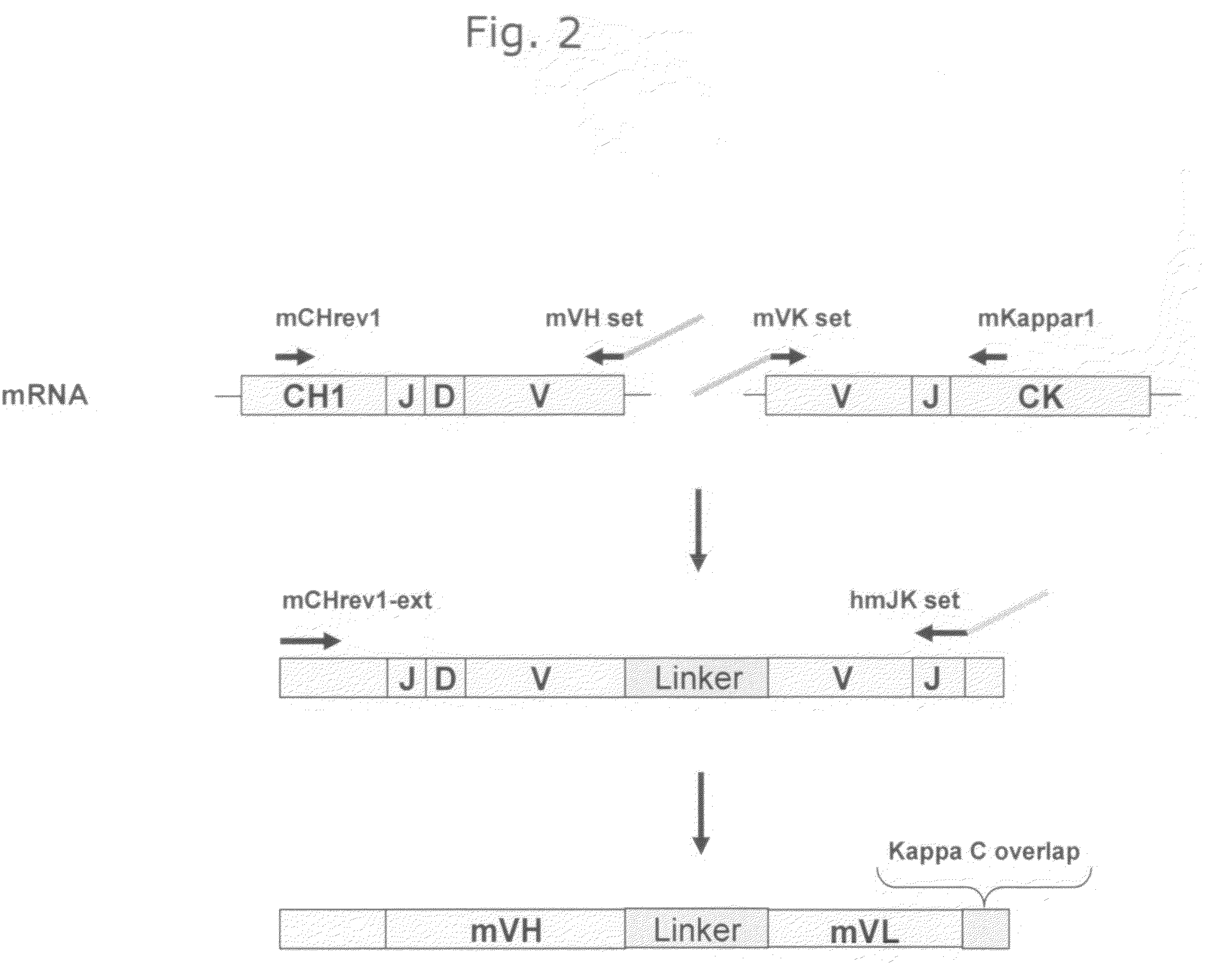
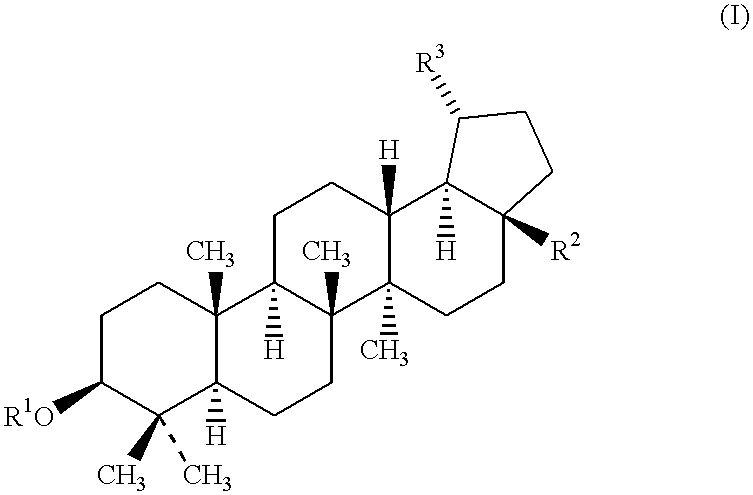
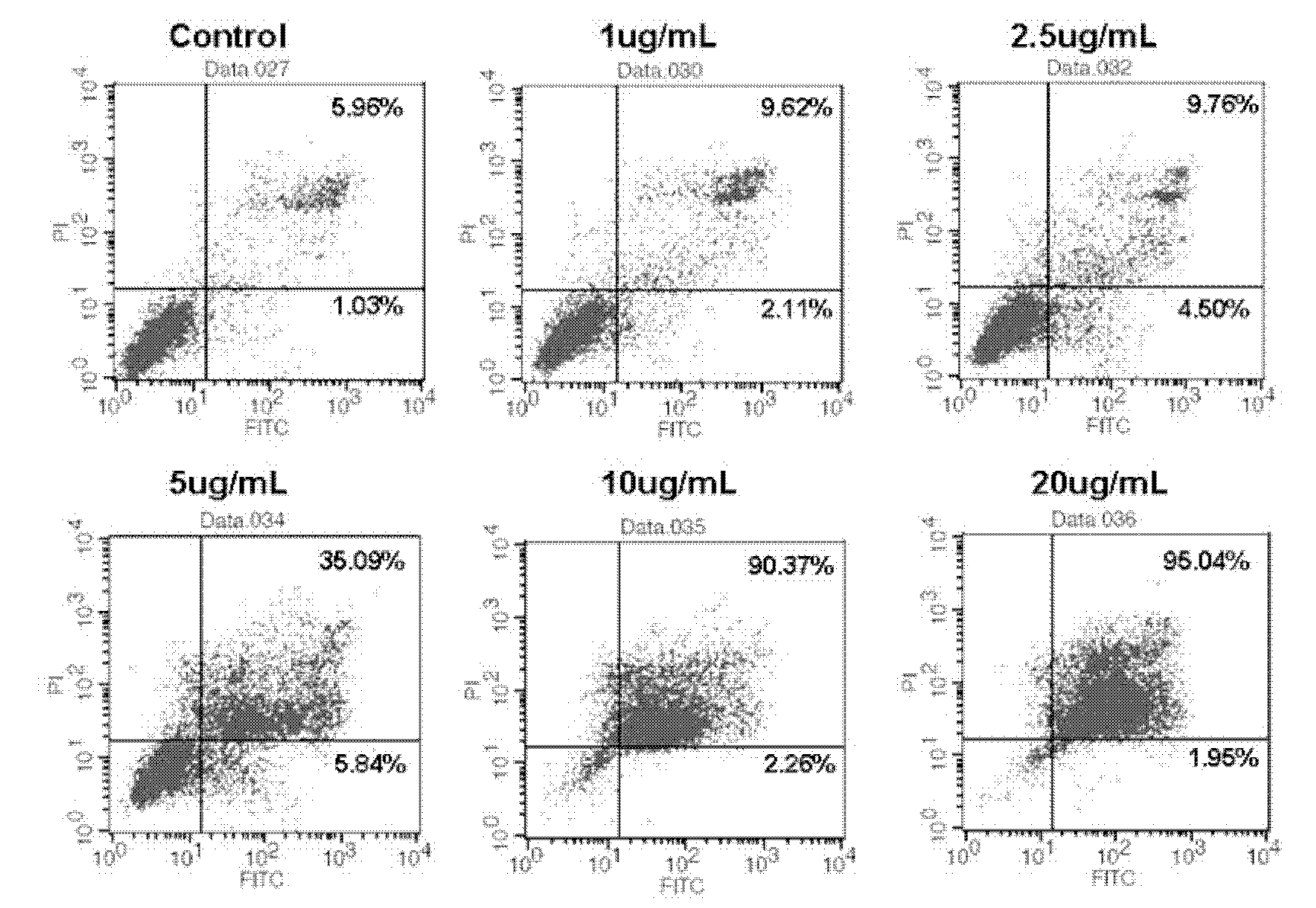
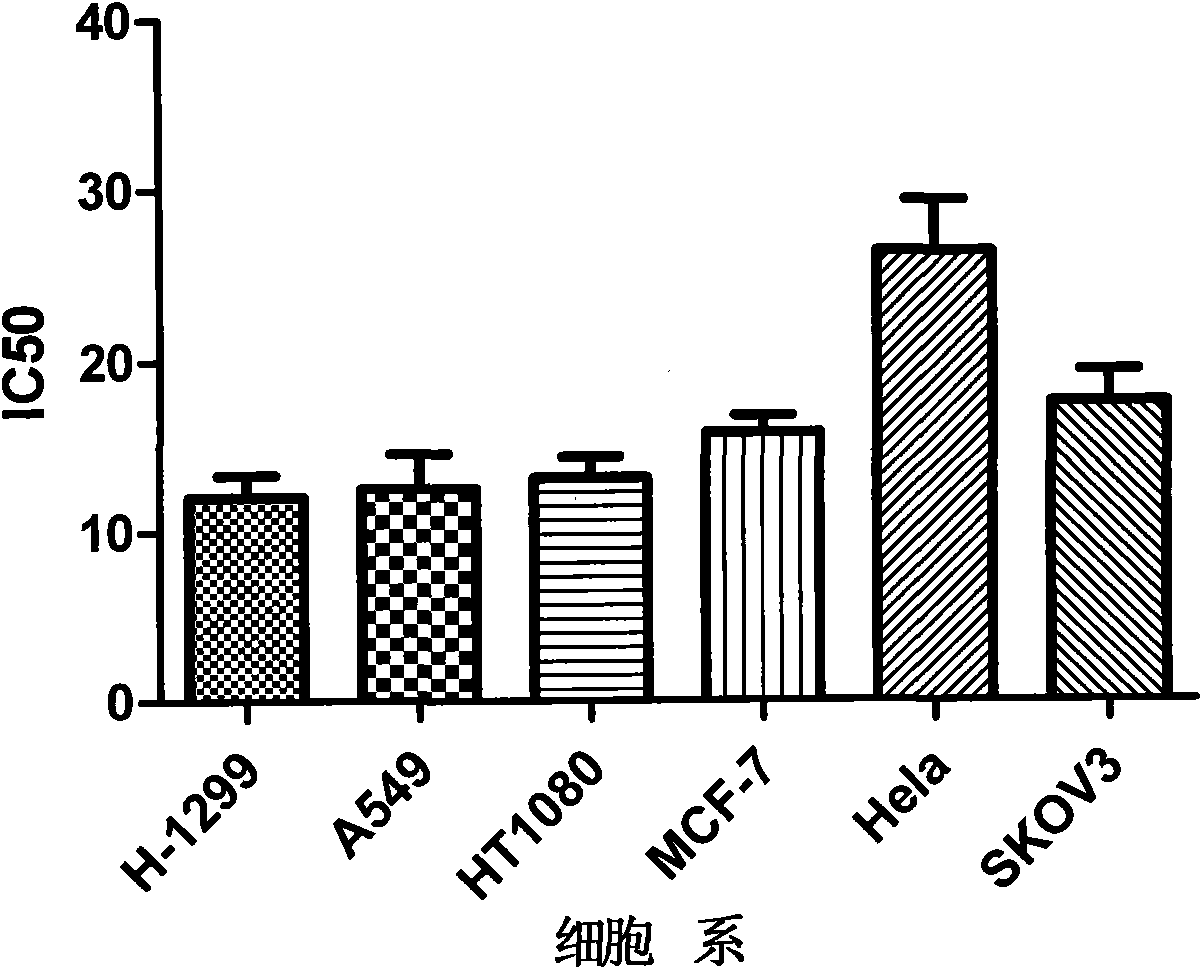
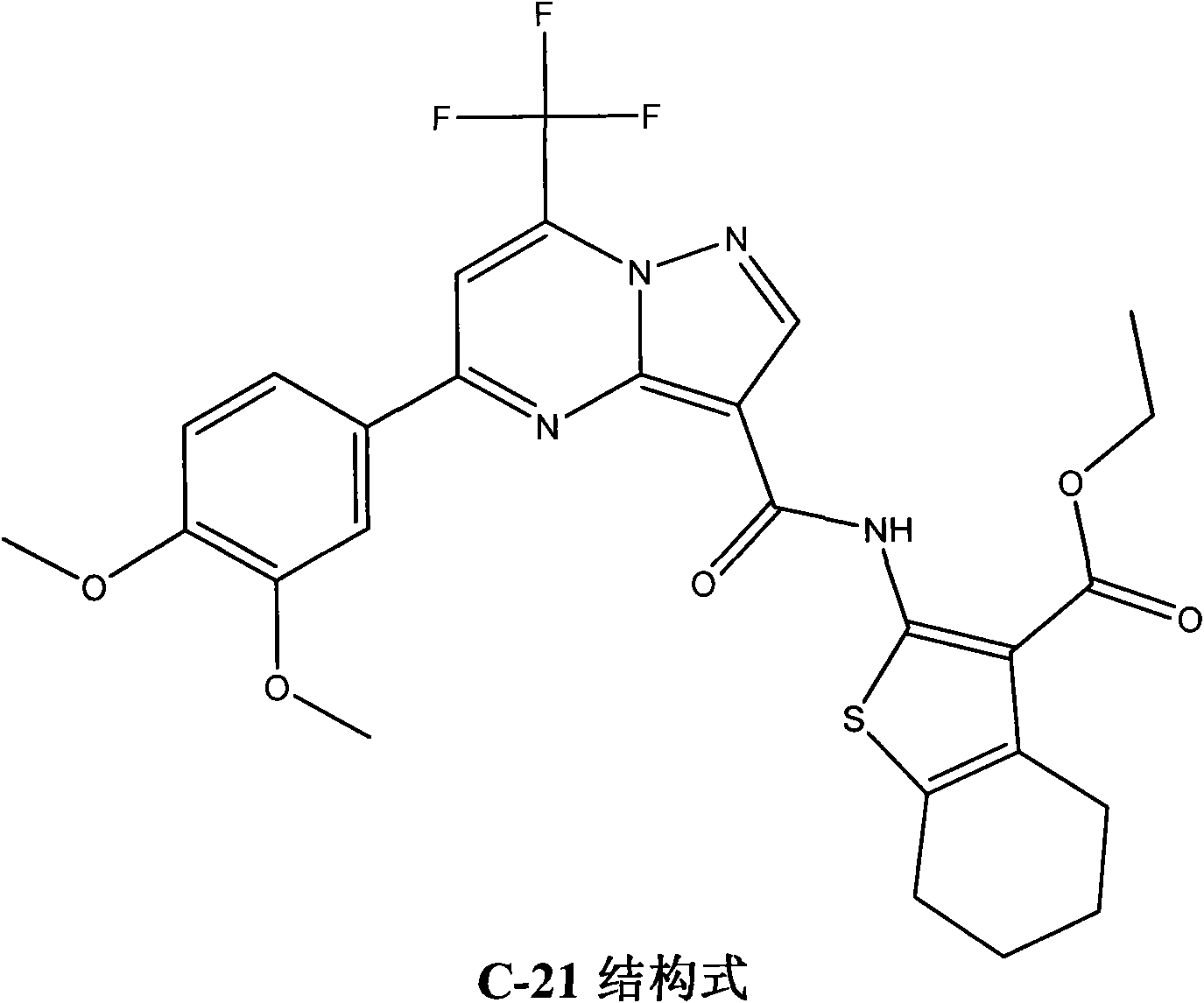
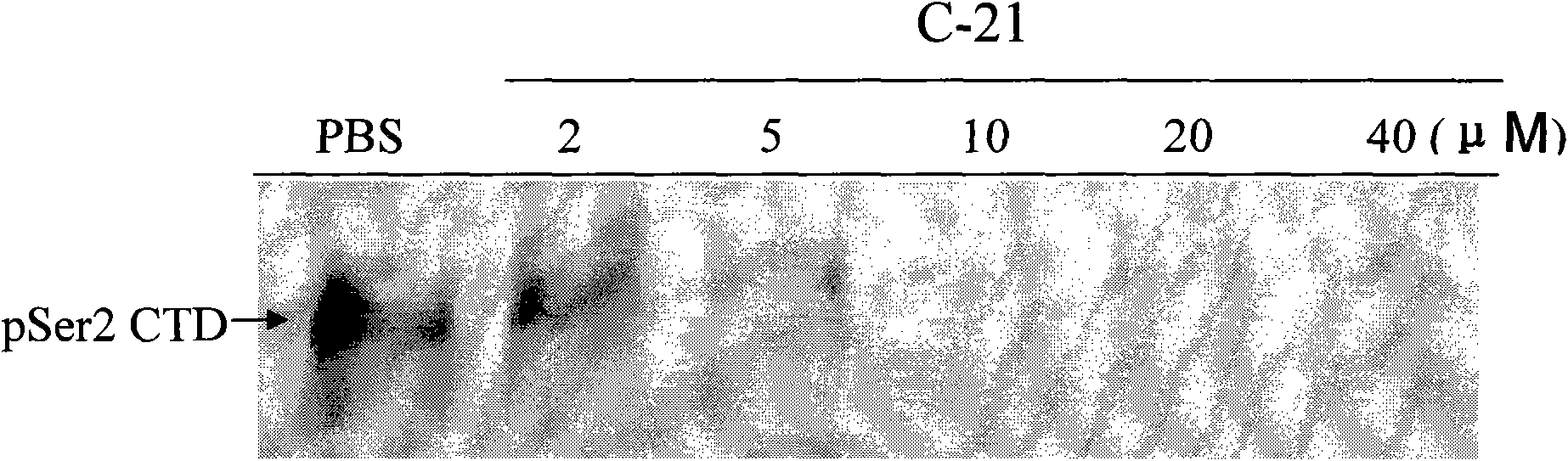
Patents
Literature
2030 results about "Mechanism of action" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
In pharmacology, the term mechanism of action (MOA) refers to the specific biochemical interaction through which a drug substance produces its pharmacological effect. A mechanism of action usually includes mention of the specific molecular targets to which the drug binds, such as an enzyme or receptor. Receptor sites have specific affinities for drugs based on the chemical structure of the drug, as well as the specific action that occurs there. Drugs that do not bind to receptors produce their corresponding therapeutic effect by simply interacting with chemical or physical properties in the body. Common examples of drugs that work in this way are antacids and laxatives.
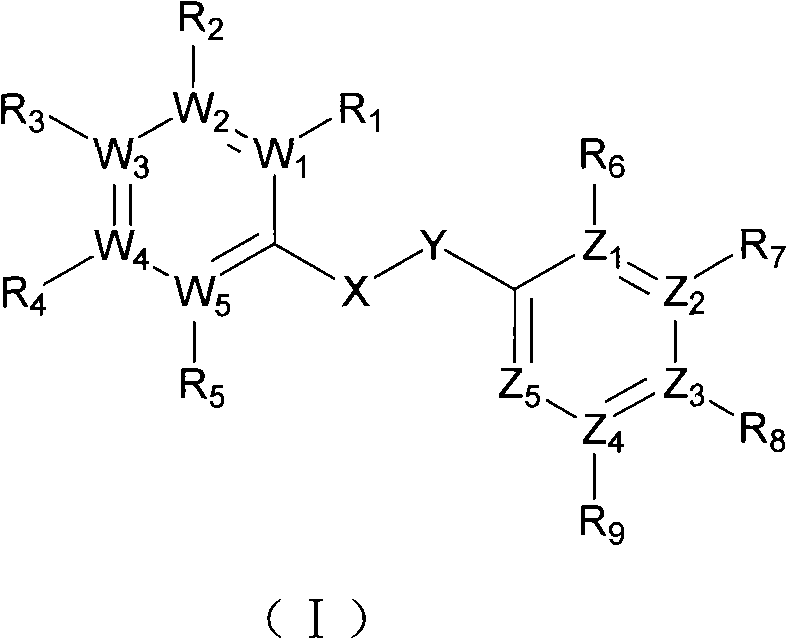
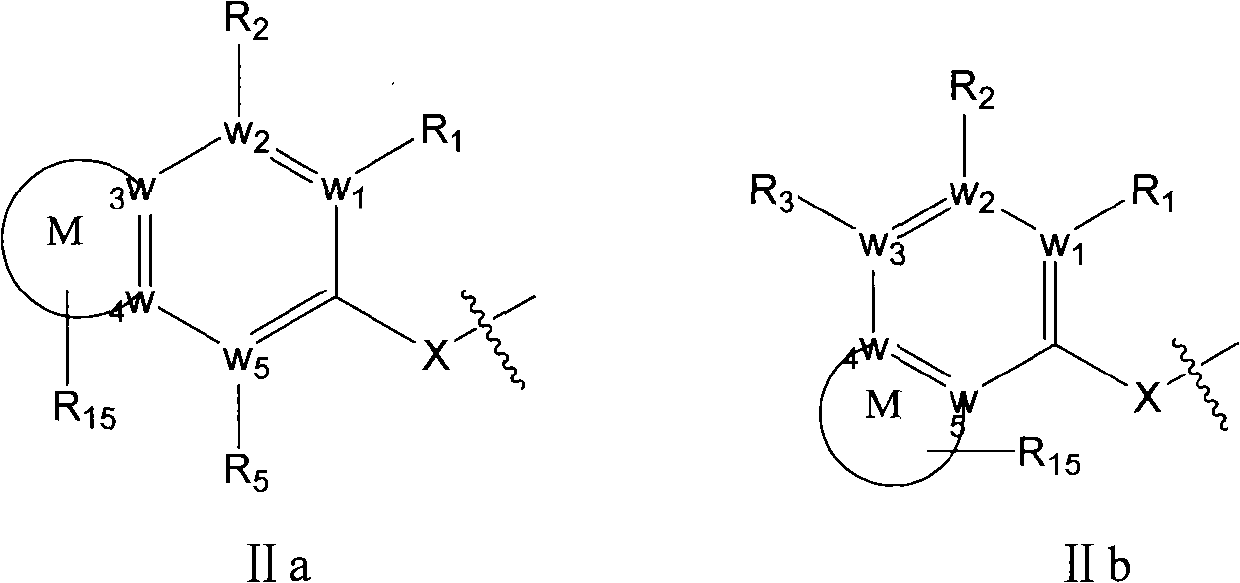
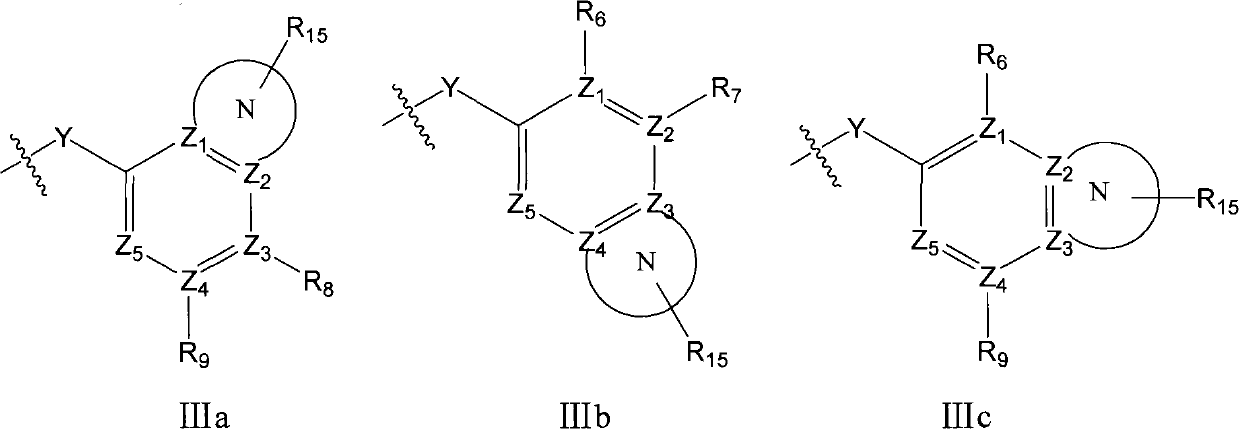
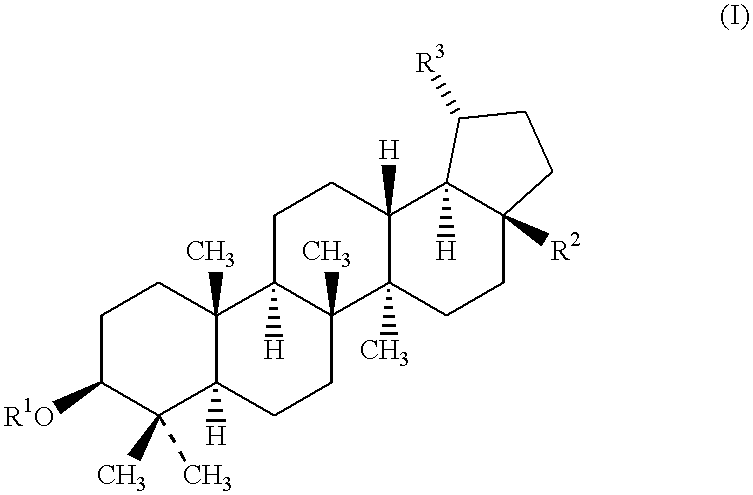
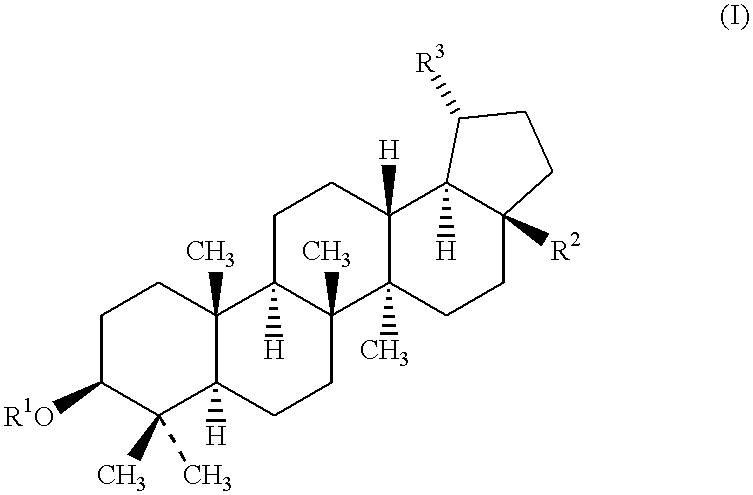
Substituted diaryl compound and preparation method and antiviral application thereof
ActiveCN102206172ALess likely to develop drug resistanceImprove securityCarbamic acid derivatives preparationSulfonic acid esters preparationMechanism of actionStructure–activity relationship
The invention provides substituted diaryl compounds as shown in general formula (I) or their pharmaceutically acceptable salts, and also provides a preparation method; a class of novel broad-spectrum antiviral compounds and pharmaceutical salts targeting cytokines are screened and obtained through studies on structure-activity relationship and action mechanism of active compounds; the compounds not only have significant broad-spectrum antiviral activity, but also have the advantages of low toxicity and good pharmaceutical properties.
Owner:MEDICINE & BIOENG INST OF CHINESE ACAD OF MEDICAL SCI
Compositions & formulations for preventing and treating chronic diseases that cluster in patients such as cardiovascular disease, diabetes, obesity, polycystic ovary syndrome, hyperlipidemia and hypertension, as well as for preventing and treating other diseases and conditions
InactiveUS20140271923A1Good for healthImproving well-beingHeavy metal active ingredientsBiocideSide effectPolycystic ovary
Patients inflicted with various clustering chronic diseases require treatment with multiple drugs having distinct mechanisms of action. Accordingly, patients with multiple conditions suffer from cumulative side effects of multiple drugs as well as drug-drug interactions. Embodiments, agents, compounds or drugs of the present invention, such as sesquiterpenes, e.g., Zerumbone, replace an equal or larger number of approved drugs during patient treatment. Examples of disorders prevented or ameliorated by administration of the formulations of this invention include but are not limited to inflammatory diseases that may be, oncological, genetic, ischemic, infectious, neurological, hematological, ophthalmological, rheumatoid, orthopedic, neurological, hematological, kidney, vascular, dermatological, gynecological, or obstetric. The present invention further relates to a method of identifying agents, compounds or drugs useful in preventing or treating CDCP related diseases and conditions as well as other disorders, diseases and conditions treatable or preventable by the same agents, compounds or drugs.
Owner:REID CHRISTOPHER BRIAN
Recombinant anti-epidermal growth factor receptor antibody compositions
ActiveUS7887805B2Reduce exerciseReduction tendencyImmunoglobulins against cell receptors/antigens/surface-determinantsFermentationHuman cancerCancer cell
Owner:LES LAB SERVIER SA
Betulinic acid and derivatives thereof useful for the treatment of neuroectodermal tumor
The present invention is, generally, directed to the use of betulinic acid and derivatives thereof for the treatment of neuroectodermal tumors. The present invention is based on the discovery that betulinic acid and its derivatives are potent anti-neuroectodermal agents. As disclosed herein, betulinic acid and its derivatives are useful for the treatment of neuroectodermal tumors, including, due to its distinct mechanism of action, neuroectodermal tumors that are resistant to conventional chemotherapeutical agents. In addition to the new use of known compounds, the invention discloses novel compounds and pharmaceutical compositions for the treatment of neuroectodermal tumors.
Owner:DEUTES KREBSFORSCHUNGSZENT STIFTUNG DES OFFENTLICHEN RECHTS
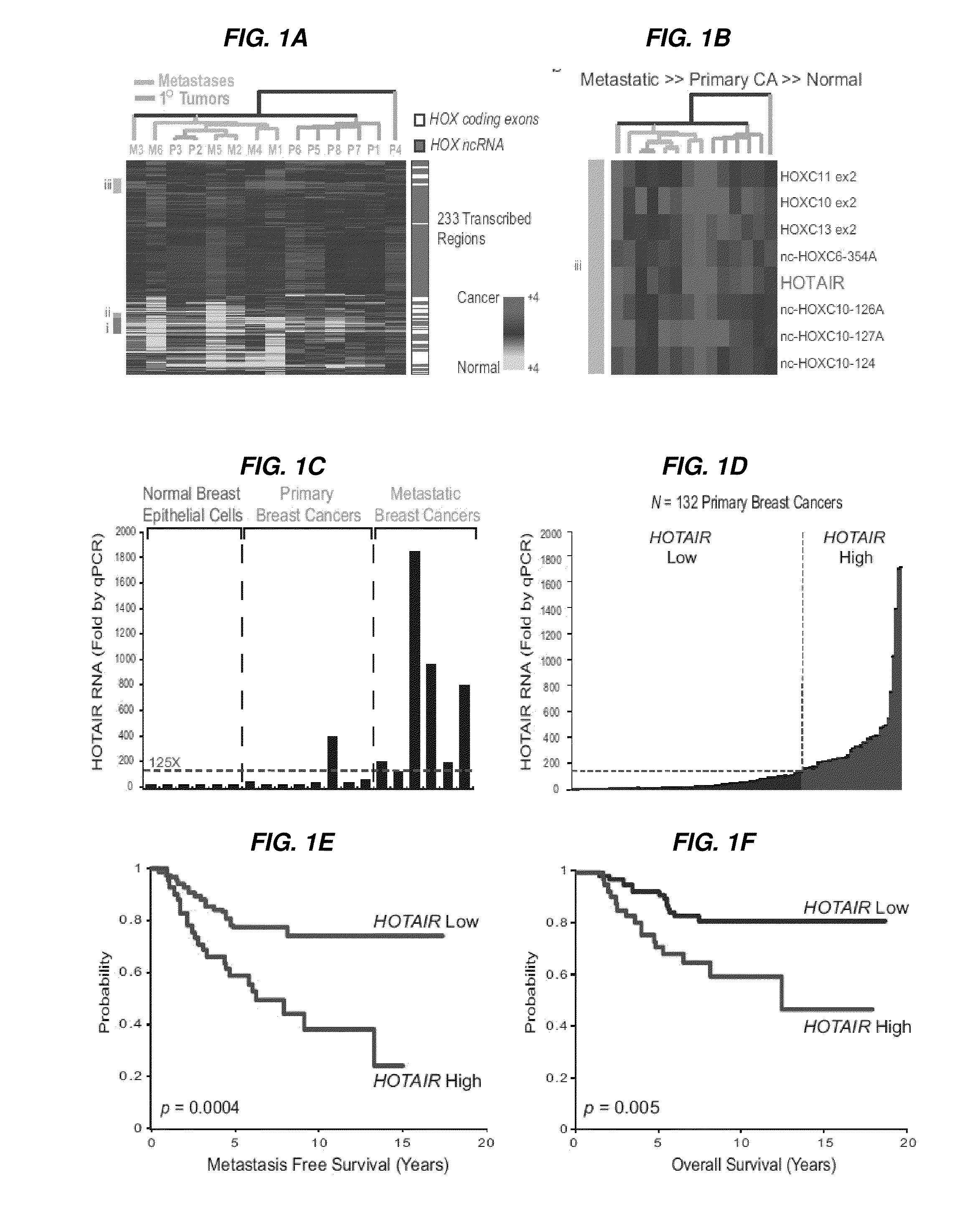
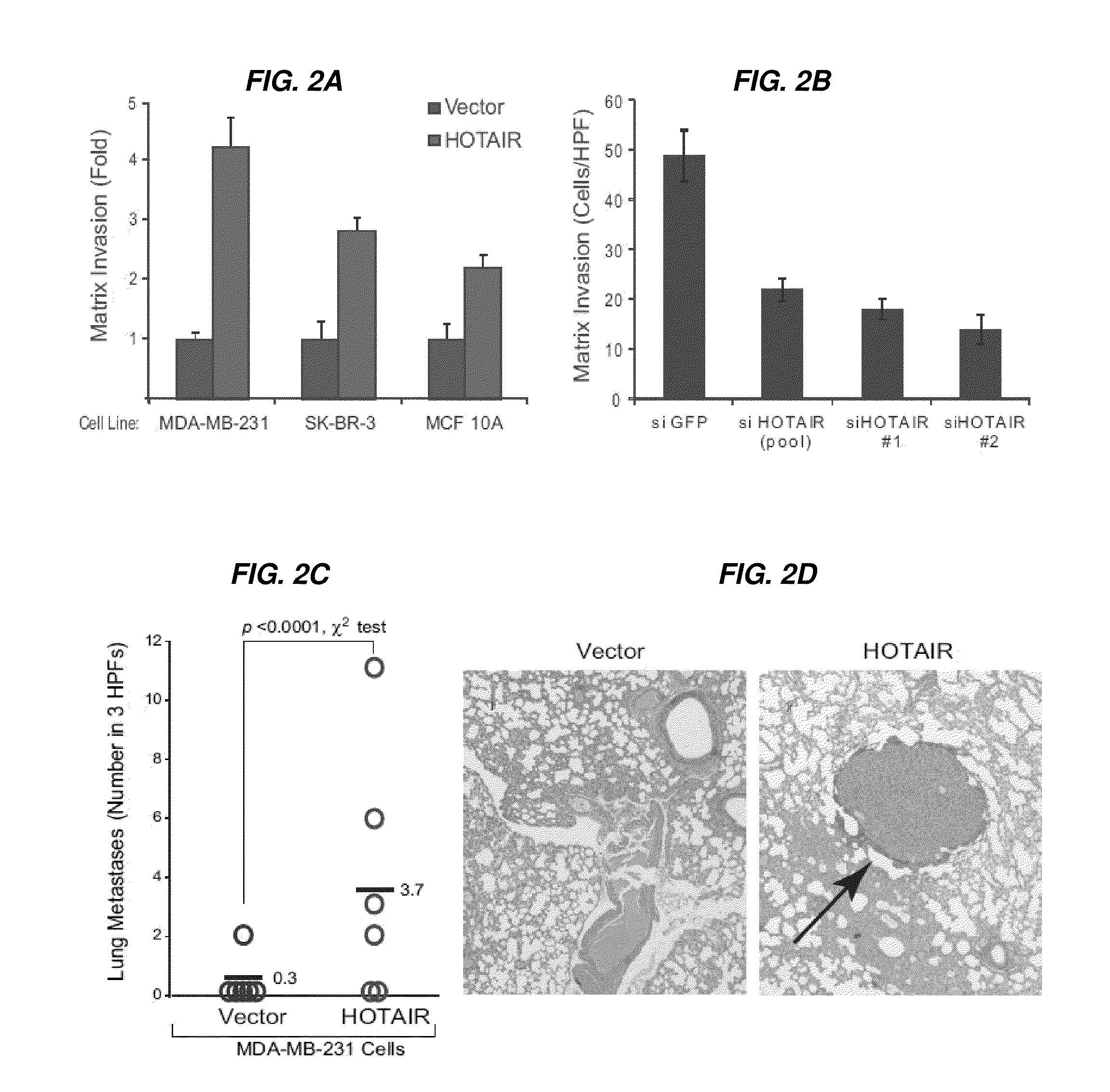
Linc rnas in cancer diagnosis and treatment
InactiveUS20120004278A1Increased and decreased expressionEasy to detectOrganic active ingredientsBioreactor/fermenter combinationsCancers diagnosisMechanism of action
Long non-coding RNAs (lincRNAs), a relatively recently recognized class of widely transcribed genes, are thought to affect chromatin state and epigenetic regulation, but their mechanisms of action and potential roles in human disease are poorly understood. The present invention shows that long non-coding RNAs in the human HOX loci are systematically dysregulated during breast cancer progression, and that expression levels of the lincRNA termed HOTAIR can predict cancer metastasis. Elevated levels of HOTAIR can lead to altered patterns of Polycomb binding to the genome. These findings indicate that lincRNAs have active roles in modulating the cancer epigenome and may be important targets for cancer diagnosis and therapy.
Owner:THE BOARD OF TRUSTEES OF THE LELAND STANFORD JUNIOR UNIV
Modified alpha-MSH peptides and derivatives thereof
InactiveUS20050130901A1High activityReduce productionPeptide/protein ingredientsImmunoglobulinsMicroorganismMechanism of action
Novel peptides with antimicrobial activity are disclosed. The novel peptides are octomeric peptides modified from alpha-MSH. The modified alpha-MSH antimicrobial peptides disclosed herein may have enhanced activity against microbes over alpha-MSH due to modifications in peptide sequence and chirality of amino acids. Due an identified mechanism of action for antimicrobial activity in which cAMP accumulates in the microbial cell, it may be that microbes will not generate resistance to these modified alpha-MSH antimicrobial peptides.
Owner:ZENGEN
Lanthionine synthetase component c-like proteins as molecular targets for preventing and treating diseases and disorders
ActiveUS20110275558A1Organic active ingredientsPeptide/protein ingredientsThiazolidinedioneAutoimmune disease
The present invention relates to the field of medical treatments for diseases and disorders. More specifically, the present invention relates to the use of the lanthionine synthetase component C-like (LANCL) proteins as therapeutic targets for novel classes of anti-inflammatory, immune regulatory and antidiabetic drugs. This includes but it is not limited to abscisic acid (ABA), ABA analogs, benzimidazophenyls, repurposed drugs or drug combinations, including thiazolidinediones (TZDs); naturally occurring compounds such as conjugated diene fatty acids, conjugated triene fatty acids, isoprenoids, and natural and synthetic agonists of peroxisome proliferator-activated receptors that activate this receptor through an alternative mechanism of action involving LANCL2 or other membrane proteins to treat or prevent the common inflammatory pathogenesis underlying type 2 diabetes, atherosclerosis, cancer, some inflammatory infectious diseases such as influenza and autoimmune diseases including but not limited to inflammatory bowel disease (Crohn's disease and Ulcerative colitis), rheumatoid arthritis, multiple sclerosis and type 1 diabetes and other chronic inflammatory conditions.
Owner:VIRGINIA TECH INTPROP INC
Complex probiotics for environmental remediation, preparation method and application thereof
ActiveCN101544959ARealize the efficiency of processingAchieve benefitsFungiBacteriaBacillus aryabhattaiBiology
The invention discloses a complex probiotics for environmental remediation, a preparation method and application thereof. The complex probiotics is prepared from the following strains in weight proportion: 1 to 5 portions of photosynthetic bacteria, 1 to 3 portions of lactic acid bacteria, 0.5 to 1.5 portions of bacillus, 1.0 to 1.4 portions of actinomycete, 0.6 to 1.0 portion of microzyme, and 1 to 5 portions of filamentous fungus, wherein the contents of active bacteria in the strains are all between1,800 and 2,600 million per milliliter. The mechanism of action of the complex probiotics comprises that: the probiotics is used as a leading raw material and combined with other useful microbes to generate an oxidation resisting material, and the oxidation resisting material decomposes and oxidizes organic matters by oxidization, reduction, fermentation and the like, so that the harmful and poisonous materials are turned into unharmful and nontoxic materials, and the idea of changing wastes into valuables is realized. The complex probiotics is widely applied in environmental protection field of deodorizing in a washroom, reutilizing domestic garbage, treating odorous lakes and stagnant water, purifying tap water and a swimming pool and the like, and has low cost.
Owner:ZHEJIANG HONGDIAN ENVIRONMENTAL PROTECTION & TECH CO LTD
Response Predictors for Erbb Pathway-Specific Drugs
InactiveUS20080254497A1Microbiological testing/measurementDisease diagnosisPhosphorylationMechanism of action
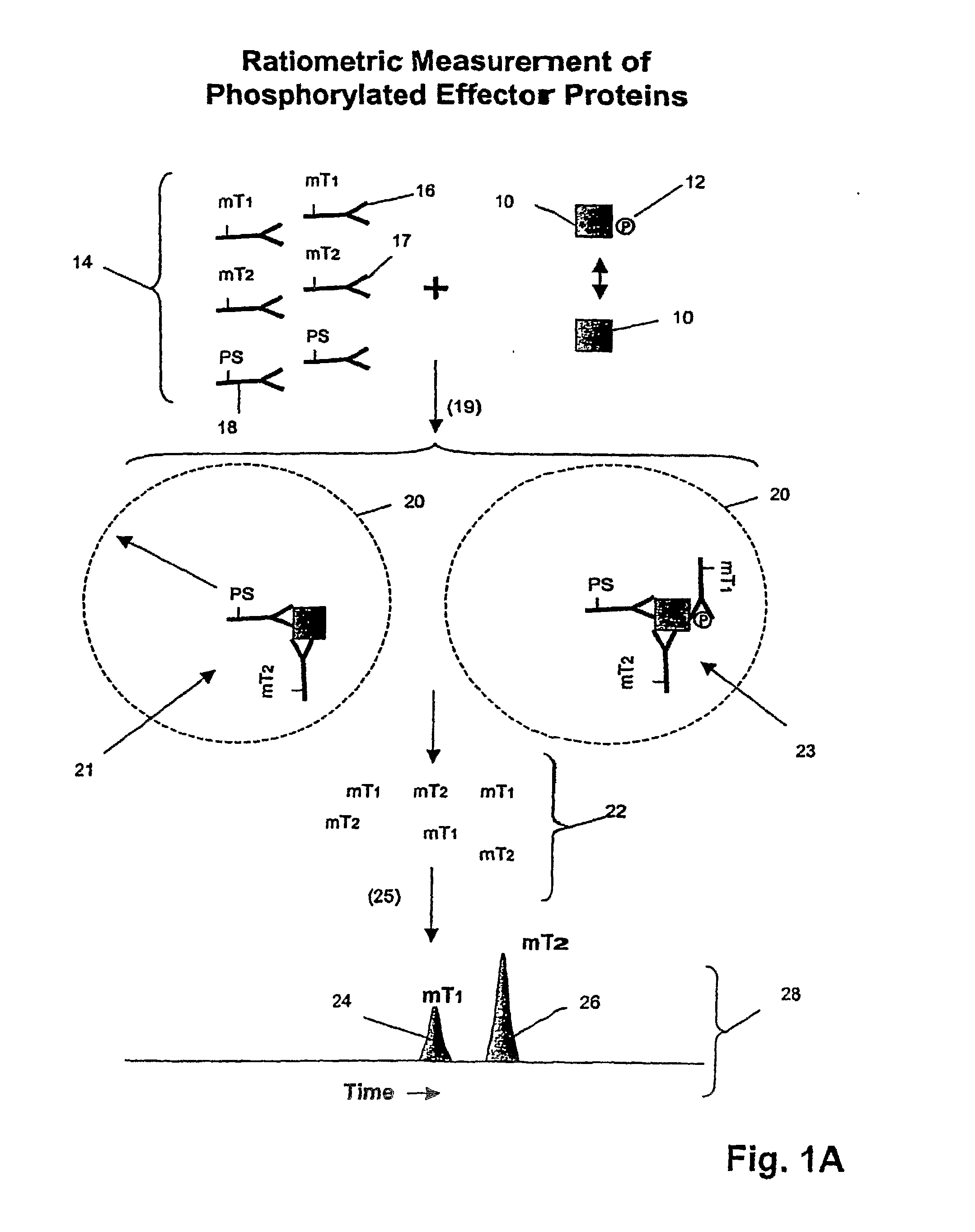
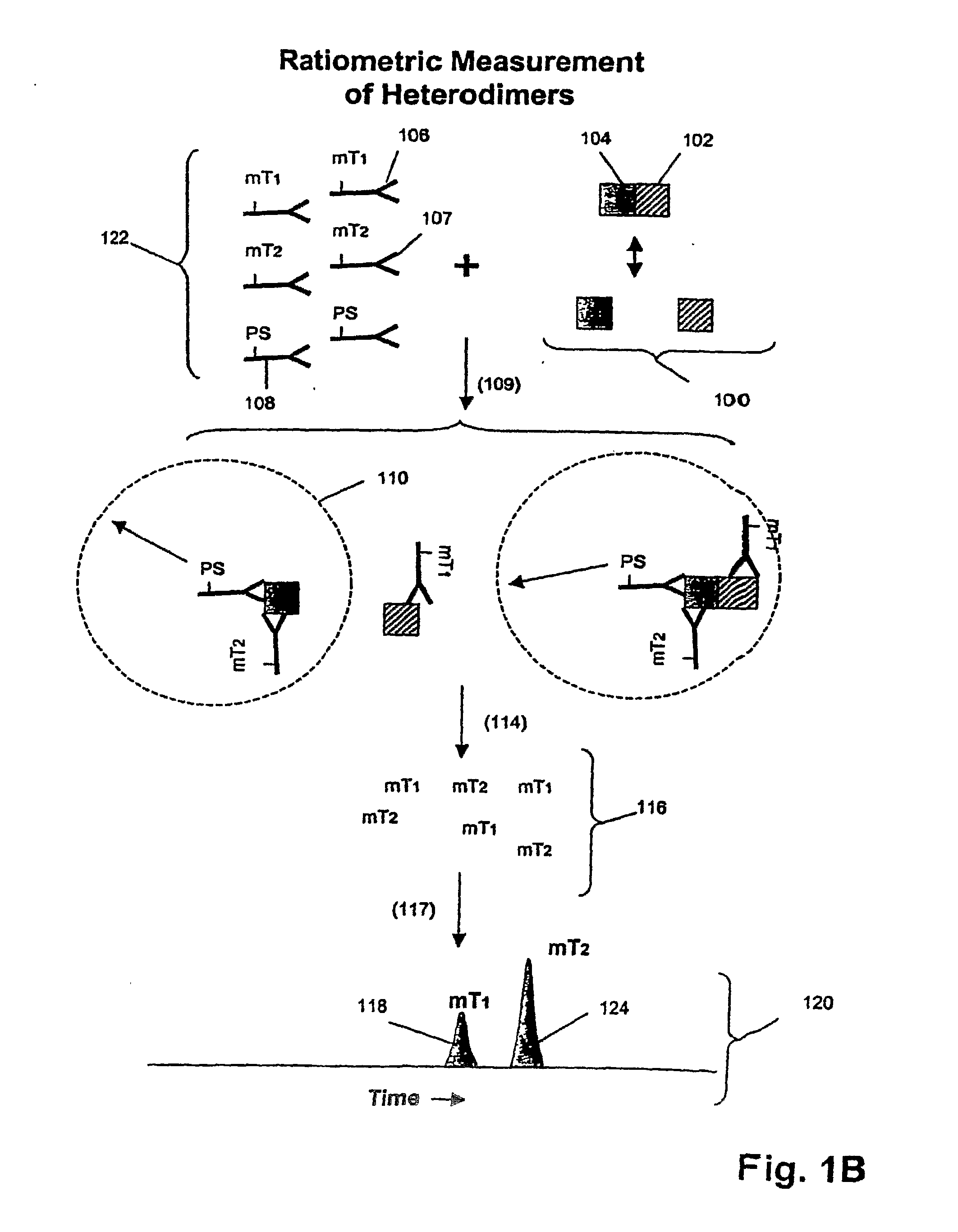
The invention provides a method of determining whether tumor cells or tissue is responsive to treatment with an ErbB pathway-specific drug. In accordance with the invention, measurements are made on such cells or tissues to determine values for total ErbB receptors of one or more types, ErbB receptor dimers of one or more types and their phosphorylation states, and / or one or more ErbB signaling pathway effector proteins and their phosphorylation states. These quantities, or a response index based on them, are positively or negatively correlated with cell or tissue responsiveness to treatment with an ErbB pathway-specific drug. In one aspect, such correlations are determined from a model of the mechanism of action of a ErbB pathway-specific drug on an ErbB pathway. Preferably, methods of the invention are implemented by using sets of binding compounds having releasable molecular tags that are specific for multiple components of one or more complexes formed in ErbB pathway activation. After binding, molecular tags are released and separated from the assay mixture for analysis.
Owner:MONOGRAM BIOSCIENCES
Application of active ingredients of juncus effuses in preparation of medicaments for resisting tumors or inhibiting angiogenesis, health-care food or cosmetics
ActiveCN102178796AStrong specificityInnovation and courageCosmetic preparationsSenses disorderDiseaseTumor chemotherapy
The invention discloses application of active ingredients of juncus effuses in the preparation of medicaments for resisting tumors or inhibiting angiogenesis, health-care food or cosmetics. In the invention, the active ingredients of juncus effuses has the activity of inhibiting the tumors and tumor angiogenesis, and the prepared medicaments for inhibiting the tumors and the tumor angiogenesis can be used for treating or preventing malignancy and diseases relevant to the tumor angiogenesis and also can be used for treatment in tumor chemotherapy and / or auxiliary chemotherapy. In the application, the medicinal curative effect and effect mechanism of the active ingredients of juncus effuses are studied and clarified by taking tumor stem cells, tumor cells and tumor neoangiogenesis as targetspots for treating diseases so as to establish a foundation for the research and development of the active ingredients of juncus effuses and the innovative anti-tumor traditional Chinese medicines ofthe active ingredients and provide scientific basis and important information for the Chinese medicinal treatment of the malignancy.
Owner:JIANGSU SUDA INVESTMENT CO LTD
Application of N-(thiofuran-2) pyrazolo (1, 5-a) pyridine-3-formanides compounds for preparing antineoplastic
The invention searches the novel micromolecule inhibitor pyrazolo (1, 5-a) miazines compounds of cyclin-dependent kinase CDK9 (cyclin-dependent kinase) through the virtual screening of a computer, biometrically measures activity thereof, and validates interaction mechanism. The invention specifically comprises the following steps: the three-dimensional crystal conformation of the cyclin-dependent kinase family member CDK9 is obtained in a way of homology modeling; and micromolecule three-dimensional database is screened with DOCK (molecular docking). The invention uses a MTT tumor cell growth inhibition test to biometrically measures the activity of the selected compounds, researches the selected compounds pyrazolo (1, 5-a) miazines with high activety in a way of molecular mechanism, validates the inhibiting effect of the compounds to the activity of CDK9 kinase, and clarifies the interaction mechanism of the compounds for inhibiting the external activity and the molecule of various malignancies such as lung cancer, osteosarcoma, oophoroma, cervical carcinoma, breast cancer, etc.
Owner:INST OF HEMATOLOGY & BLOOD DISEASES HOSPITAL CHINESE ACADEMY OF MEDICAL SCI & PEKING UNION MEDICAL COLLEGE
Preparing method for MC3R gene knockout pigs
InactiveCN106191113AKnockout is fast and efficientPeptidesVector-based foreign material introductionLarge fragmentMechanism of action
The invention discloses a preparing method for MC3R gene knockout pigs. The preparing method for MC3R gene knockout pigs includes the steps that a CRISPR / Cas9 system is adopted, the CRISPR / Cas9 system comprises an encoding gene of gRNA1 and an encoding gene of gRNA2, and the target sequence of a MC3R gene identified by the gRNA1 is a sequence 1 in a sequence table; the target sequence of the MC3R gene identified by the gRNA2 is a sequence 2 in a sequence table. An experiment shows that the MC3R gene knockout efficiency is 29.16% with the preparing method for MC3R gene knockout pigs; a target gene can be rapidly and efficiently knocked out in a large-fragment mode, exogenous gene fragments are not left, and the preparing method can be used for researching the action mode and mechanism of the MC3R gene, and can be further used for animal breeding.
Owner:CHINA AGRI UNIV
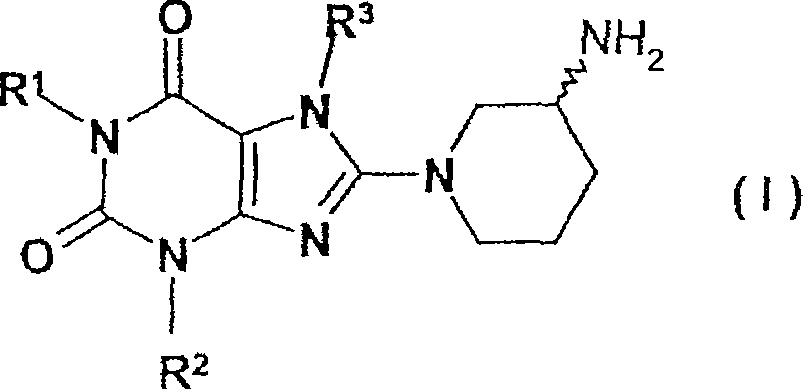
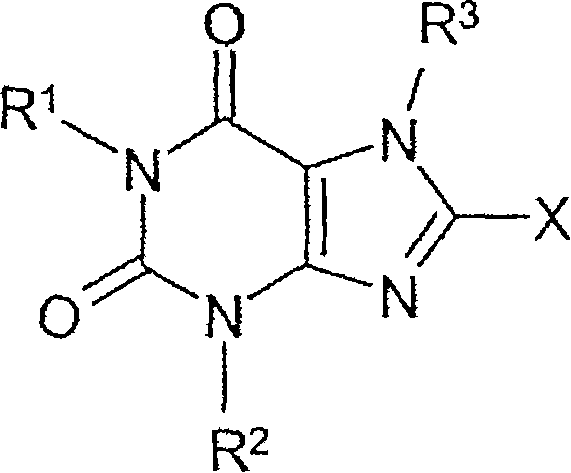
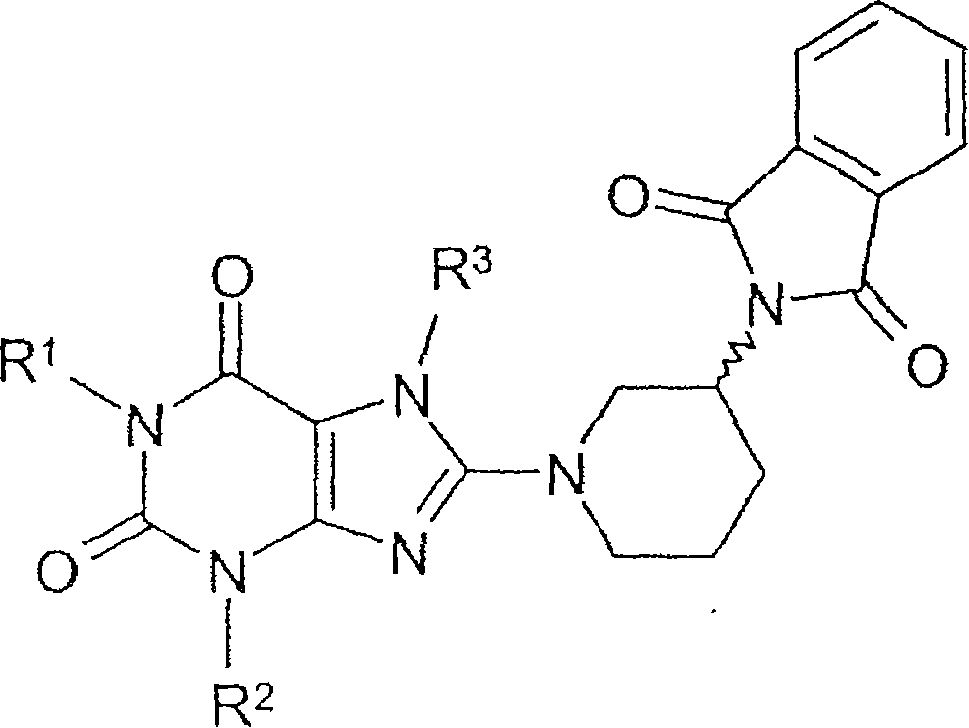
Method for producing chiral 8-(3-amino-piperidin-1-yl)-xanthines
Method for preparing 8-(3-aminopiperidin-1-yl)-xanthine derivatives (I) and their enantiomers and salts. Method for preparing 8-(3-aminopiperidin-1-yl)-xanthine derivatives of formula (I) and their enantiomers and salts by: (a) reacting an 8-X-xanthine (III) with 3-phthalimidopiperidine (A), or its enantiomer; (b) deprotecting the product (II); and (c) optionally conversion to salt. X : halo or sulfonate ester, especially bromo; R 1>phenylcarbonylmethyl, benzyl, naphthylmethyl, pyridinylmethyl, pyrimidinylmethyl, (iso)quinolinylmethyl, quinazolinylmethyl, quinoxalinylmethyl, naphthyridinylmethyl or phenanthridinylmethyl, all optionally substituted by one or two, same or different, Ra; R 2>1-3C alkyl, cyclopropyl or phenyl; R 3>2-buten-1-yl, 3-methyl-2-buten-1-yl, 2-butyn-1-yl, or 2-(fluoro, chloro, bromo, iodo, methyl, trifluoromethyl or cyano)-benzyl; Ra : hydrogen, fluoro, chloro, bromo, cyano, methyl, trifluoromethyl, ethyl, phenyl, methoxy, di- or tri-fluoromethoxy, or two Ra on adjacent C atoms complete OCH 2O or OCH 2CH 2O Independent claims are also included for the following: (1) (R) and (S)-3-phthalimidopiperidine as new compounds; and (2) methods for preparing the compounds of (1). [Image] [Image] - ACTIVITY : Antidiabetic; Antiarthritic; Anorectic; Osteopathic. No details of tests for these activities are given. - MECHANISM OF ACTION : Inhibition of dipeptidylpeptidase IV.
Owner:BOEHRINGER INGELHEIM INT GMBH
Reelin deficiency or dysfunction and methods related thereto
InactiveUS20090215896A1High activityImprove the level ofBiocideSenses disorderNervous systemSupplement use
A method of measuring Reelin as a biomarker, to non-destructively assess or predict DHA levels in the brain and in other, currently inaccessible or difficult-to-access, key components of the central nervous system (CNS) is described. Also described is a method to prevent, delay the onset of, or treat Reelin deficiency or dysfunction and / or a disease or condition associated with Reelin deficiency or dysfunction, comprising administering to a patient diagnosed with or suspected of having a Reelin deficiency or dysfunction an amount of a PUFA, and particularly an omega-3 PUFA, and more particularly, docosahexaenoic acid (DHA) or a precursor or source thereof, to compensate for the effects of Reelin deficiency or dysfunction in the patient. Also described is a method to prevent or reduce developmental defects or disorders associated with Reelin dysfunction or deficiency through the supplemental use of polyunsaturated fatty acids (PUFAs—unsaturated fatty acids having two or more double bonds), and particularly highly unsaturated fatty acids (HUFAs—unsaturated fatty acids having three or more double bonds), and more particularly a HUFA selected from arachidonic acid (ARA), eicosapentacnoic acid (EPA), docosahexaenoic acid (DHA) and docosapentaenoic acid (DPA), and even more particularly omega-3 HUFAs, and more particularly DHA, to: compensate for reduced fatty acid binding protein or function thereof in the patient; compensate for reduced brain lipid binding protein or function thereof in the patient; improve the activity of fatty acid binding proteins in the patient; increase the expression of brain lipid binding proteins (BLBPs) in the patient; improve at least one parameter of the mechanism of action of brain lipid binding proteins in the patient; overcome a deficiency of DHA in central nervous system (CNS) structures and improve the resulting function thereof; increase the incorporation of functional DHA and other PUFAs into the phospholipid membranes of glial cells and neurons in the patient; increase the level of Reelin and / or improve the activity of Reelin in the patient; and / or improve at least one symptom of a disease or condition associated with Reelin deficiency or dysfunction.
Owner:MARTEK BIOSCIENCES CORP
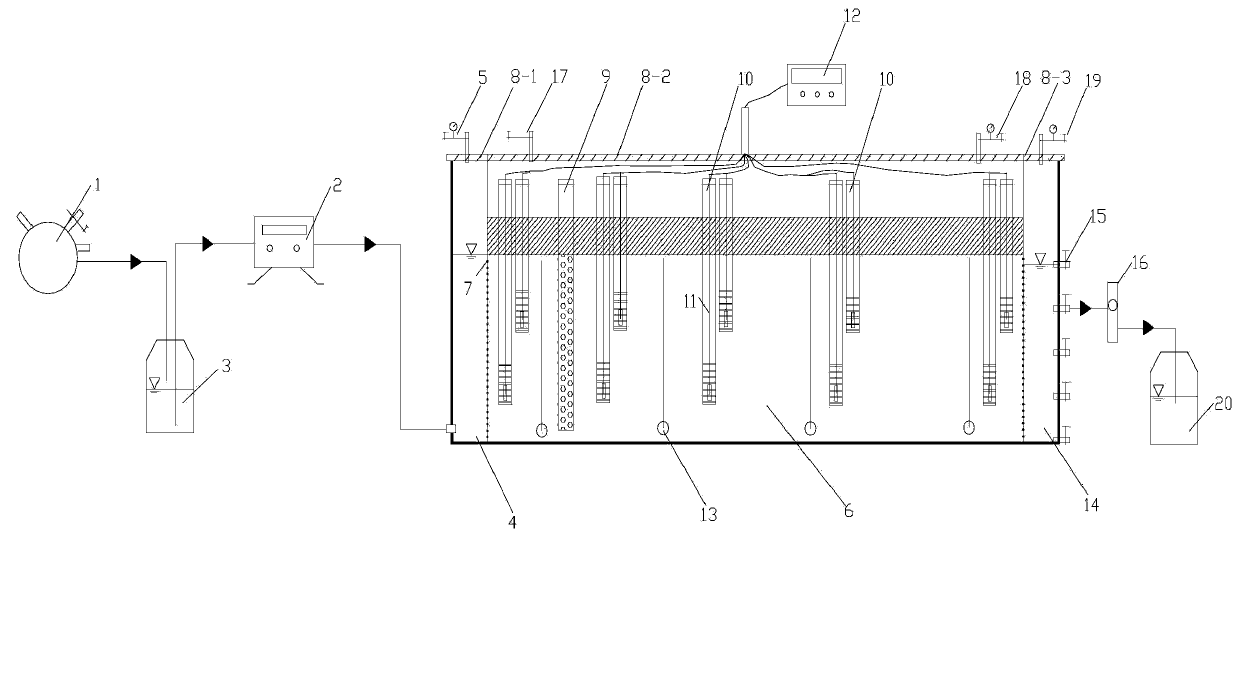
Simulation test device and simulation test method for in-situ chemical and biological remediation of underground water
ActiveCN103630659APromote degradationFlexible and easy to designGeneral water supply conservationTesting waterUltrasound attenuationMechanism of action
The invention discloses a simulation test device and a simulation test method for in-situ chemical and biological remediation of underground water. The simulation test device comprises a seepage slot, an inlet water buffer slot, an outlet water buffer slot, a water supply device and a liquid recovering bottle; the seepage slot is respectively communicated with the inlet water buffer slot and the outlet water buffer slot through porous water distribution baffles; the outer side of the outlet water buffer slot is provided with a drain hole with a valve; the seepage slot is provided with a row of injection wells at the upstream and a plurality of monitoring wells inside, and the monitoring wells are internally provided with water quality analysis probes; the tops of the seepage slot and the inlet water buffer slot are provided with a top sealed plate in which air inlets and air outlets are arranged; and the side wall of the seepage slot is provided with a pressure measuring pipe. The method comprises: filling with a medium, saturating with water, introducing polluted underground water by pumping, and adding a reagent for simulating the in-situ chemical and biological remediation process. The simulation test device is flexible, simple and convenient in design, low in test cost and strong in process controllability, can be used for researching the dynamic effect and the action mechanism for chemical and biological enhanced treating on organically-polluted underground water under an in-situ condition, and also can be used for observing the attenuation process of specific pollutants in underground water.
Owner:SHANGHAI ACADEMY OF ENVIRONMENTAL SCIENCES
Targeted-FTO-gene-knockout sgRNA (small guide ribonucleic acid) and CRISP (clustered regularly interspaced short palindromic repeats)/Cas9 slow virus system and application thereof
InactiveCN106047877AConvenient researchNucleic acid vectorOxidoreductasesMechanism of actionCell strain
The invention discloses a targeted-FTO-gene-knockout sgRNA (small guide ribonucleic acid) and CRISP (clustered regularly interspaced short palindromic repeats) / Cas9 slow virus system and application thereof. The sgRNA is selected from FTOsgRNAsp2 or FTOsgRNAsp3 with the following DNA sequence. The sgRNA has high cutting efficiency for the FTO gene. When the sgRNA-containing CRISPR / Cas9 slow virus system is used for transfecting SV-HUC-1, the FTO protein expression level of the obtained cell strain is obviously lowered. Therefore, the sgRNA disclosed by the invention can effectively implement targeted FTO gene knockout; and after the sgRNA is established into the CRISPR / Cas9 slow virus system, the system can knock out the FTO gene to obtain the FTO-gene-knockout cell strain, thereby being beneficial to researching the action mechanism of FTO in the cell strain.
Owner:THE FIRST AFFILIATED HOSPITAL OF SUN YAT SEN UNIV
Recombinant anti-epidermal growth factor receptor antibody compositions
ActiveUS20090004192A1Good effectReduce propensityFermentationAntineoplastic agentsCancer cellEpidermal Growth Factor Receptor Antibody
The invention relates to the field of recombinant antibodies for use in human cancer therapy. More specifically the invention provides compositions or mixtures of antibodies capable of binding human EGFR. Antibody compositions with 3 or more antibodies shown synergy in reduction of proliferation of representative cancer cell lines. Advantageous results have also been obtained with a composition comprising two different chimeric anti-hEGFR antibodies which show a new mechanism of action based on rapid and efficient receptor internalisation, induction of terminal differentiation and subsequent tumour eradication in an animal model. The antibodies of the invention can be manufactured in one bioreactor as a polyclonal antibody.
Owner:LES LAB SERVIER
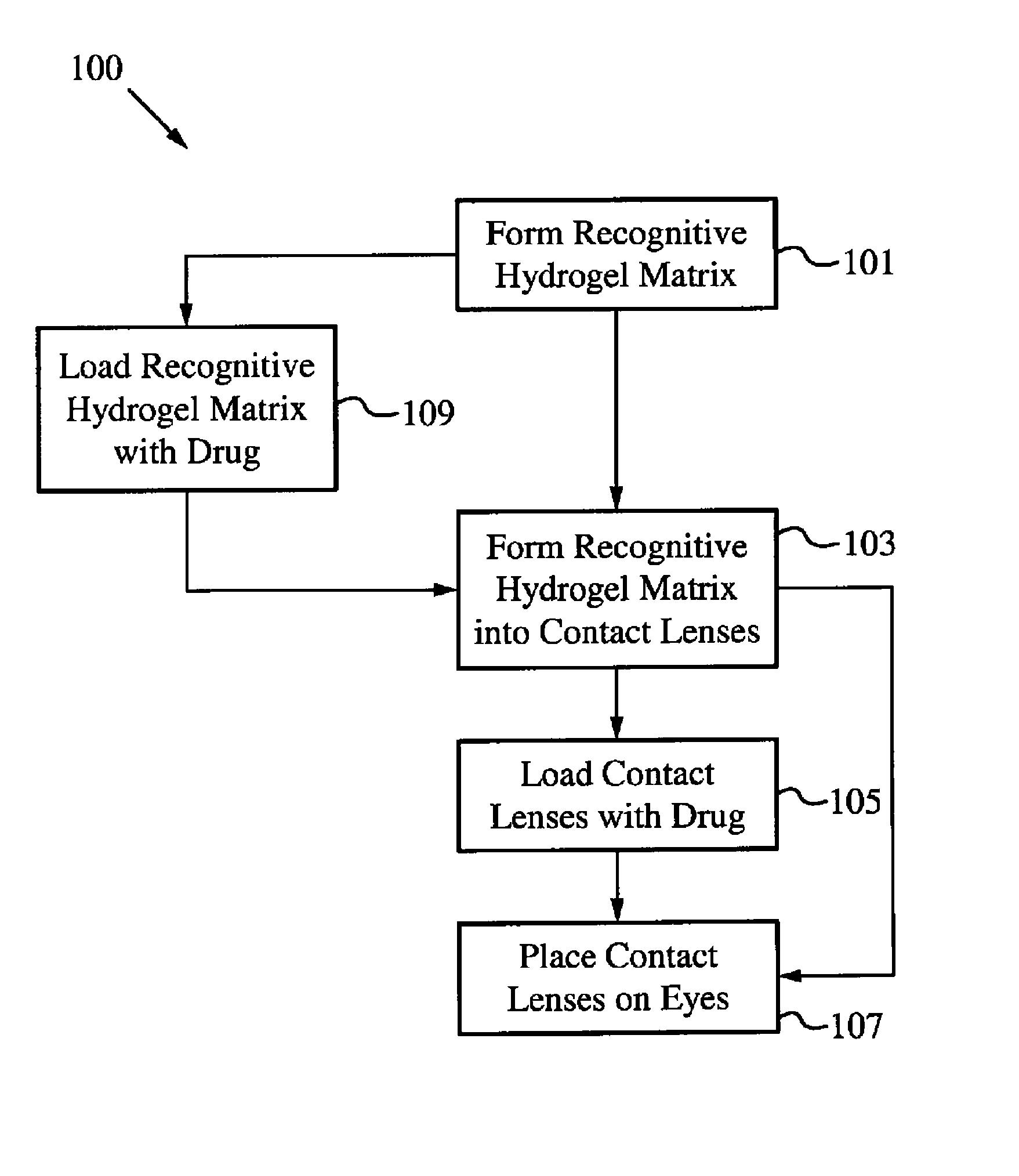
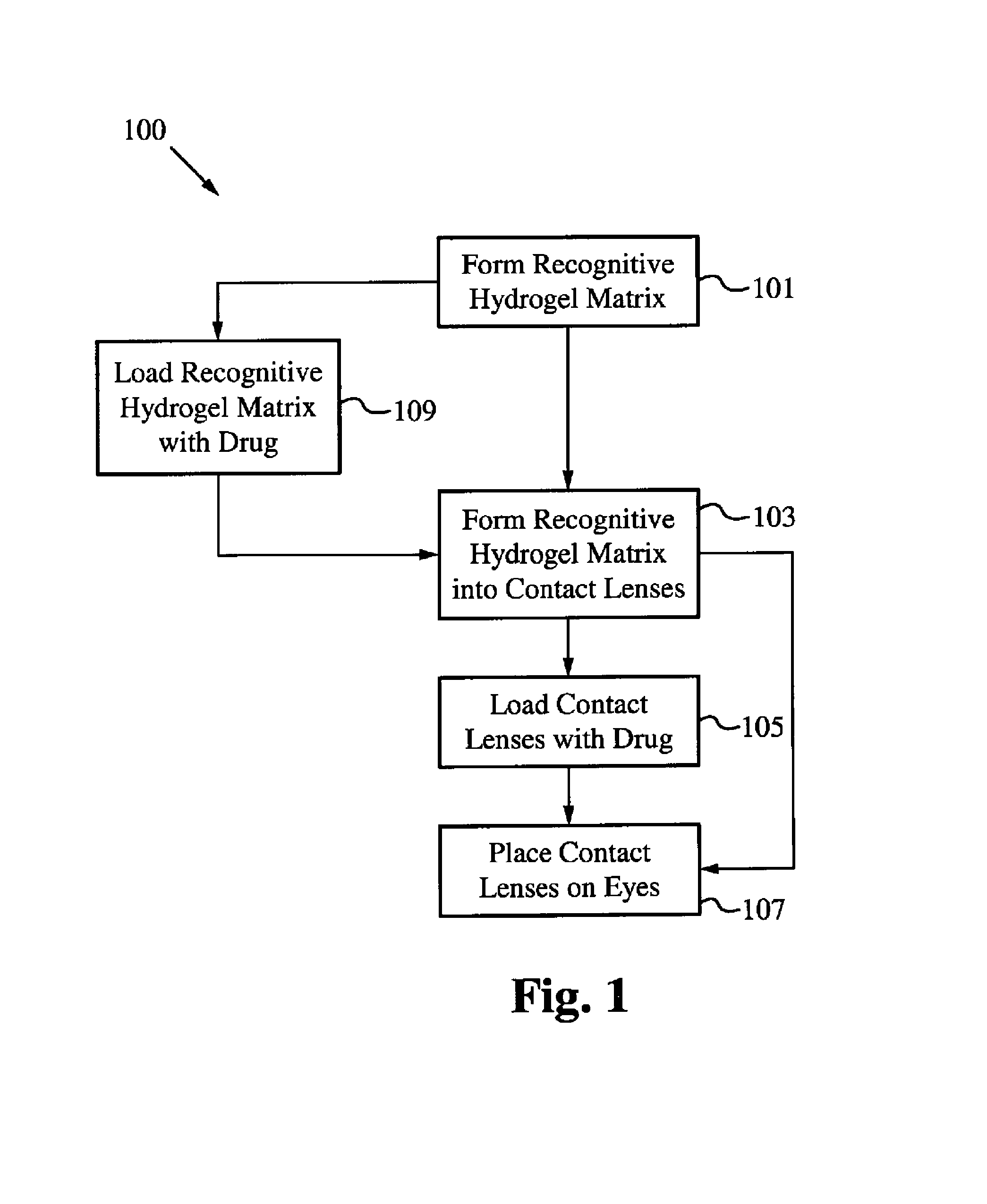
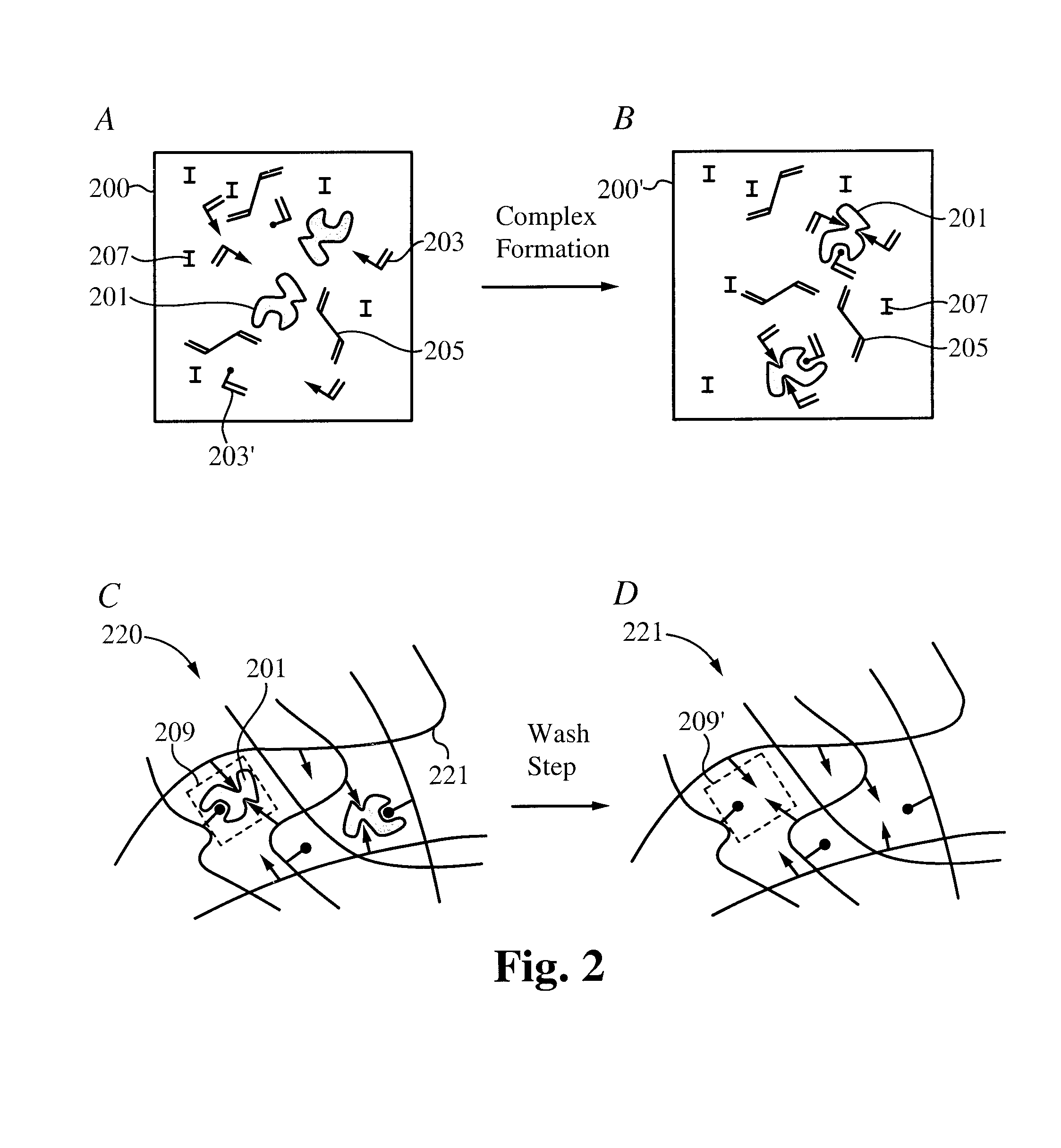
Contact drug delivery system
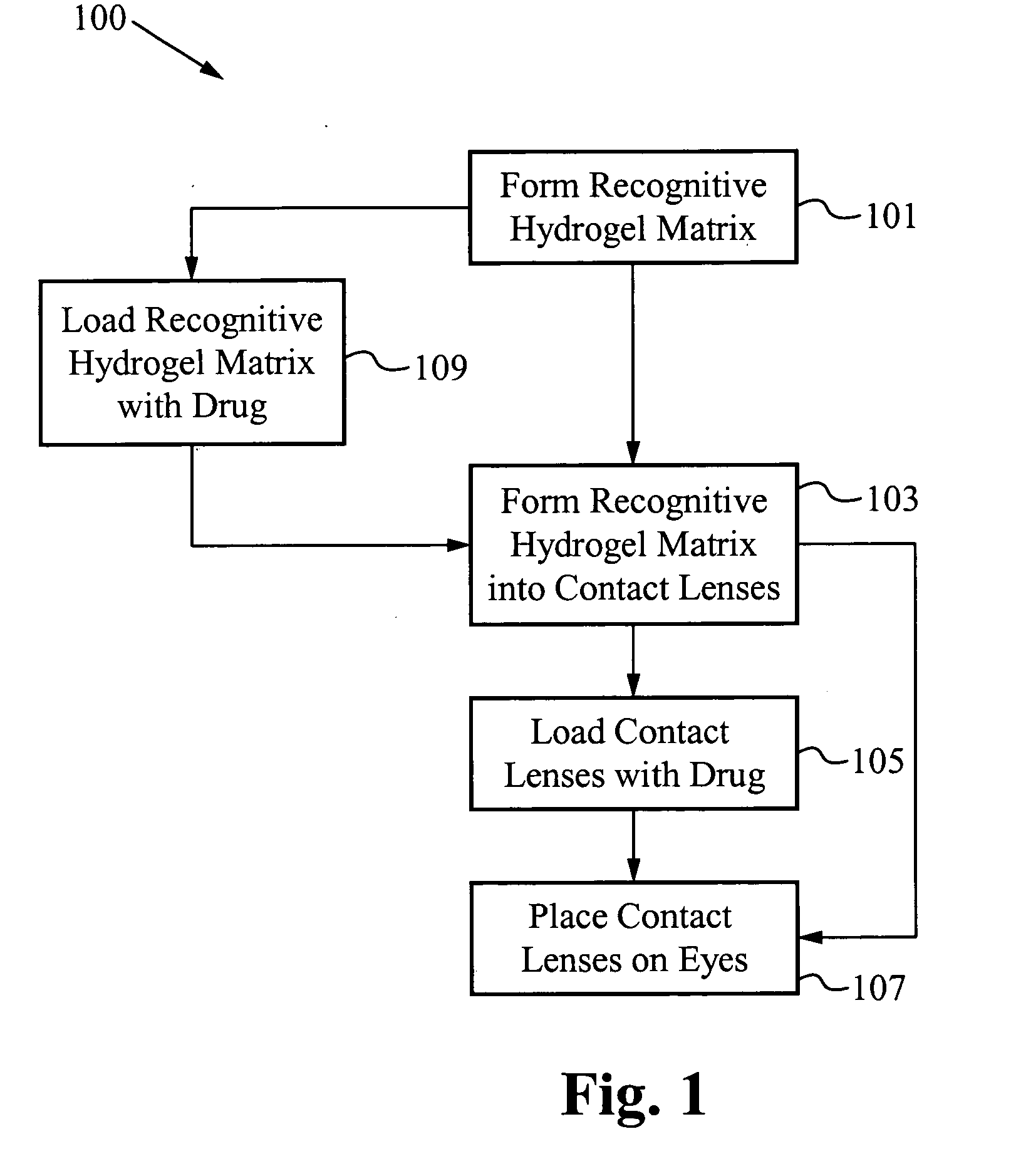
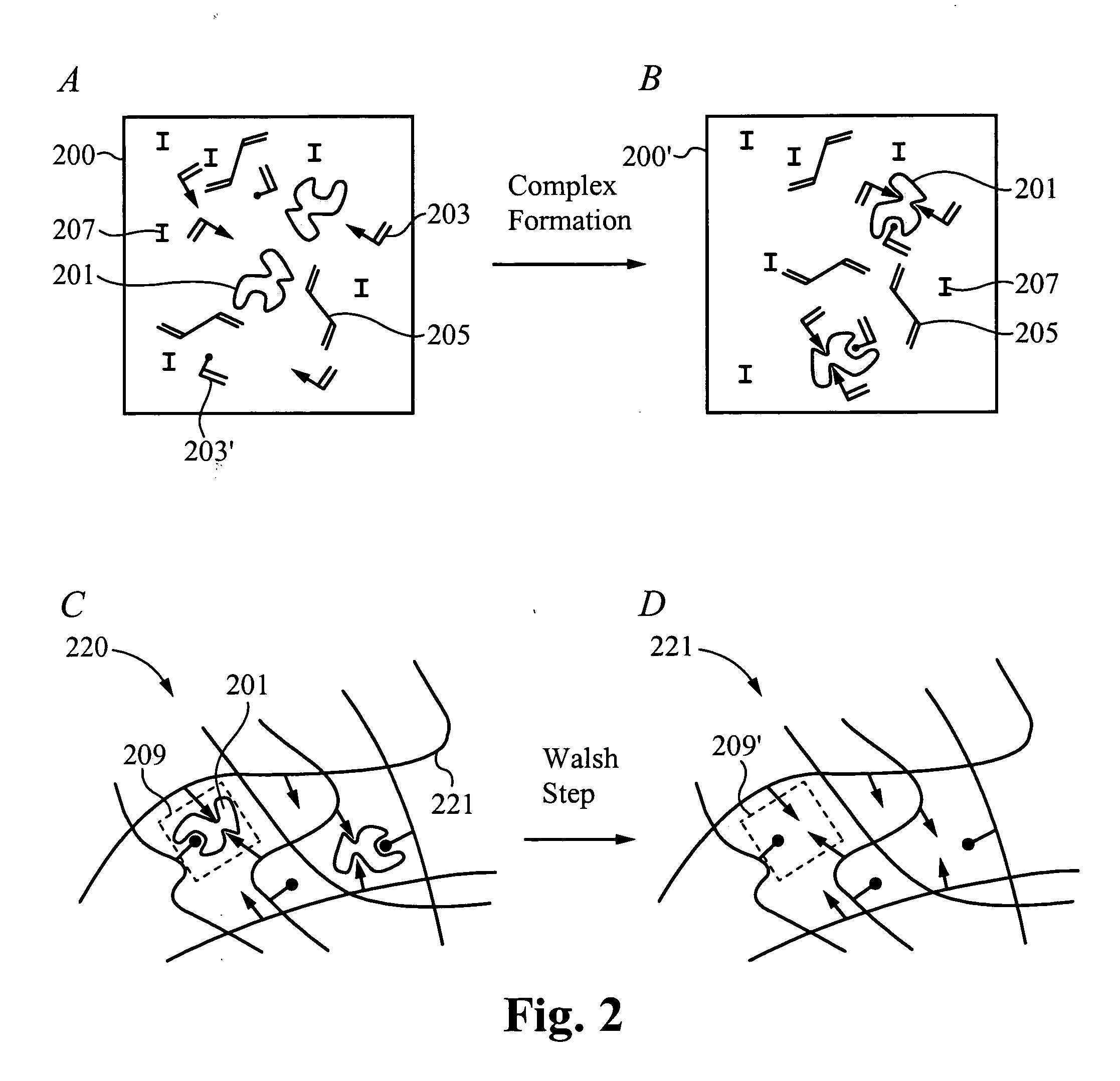
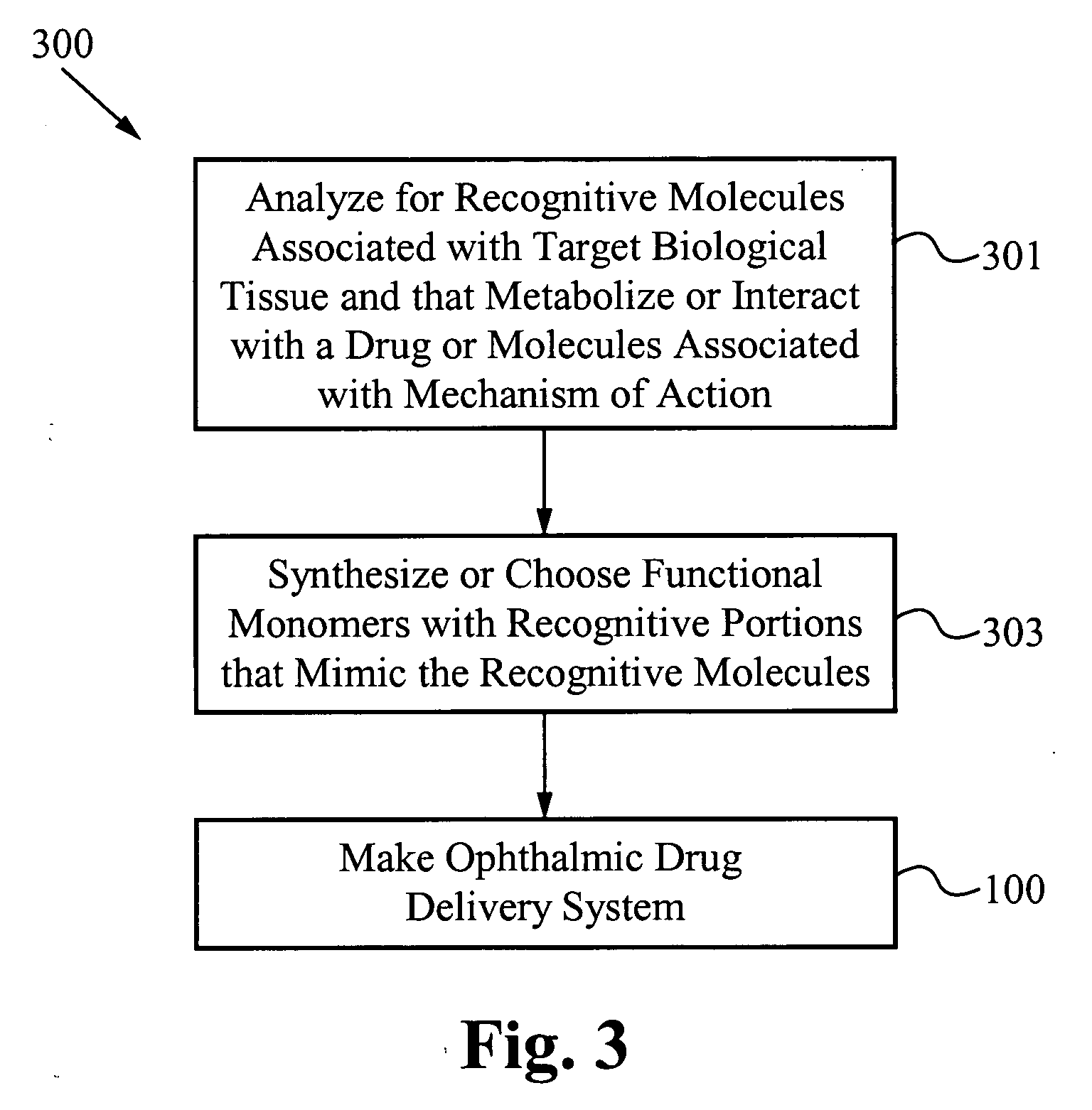
A drug delivery system is disclosed. The drug delivery system includes a recognitive polymeric hydrogel through which a drug is delivered by contacting biological tissue. The recognitive polymeric hydrogel is formed using a bio-template, which is a drug or is structurally similar to the drug, functionalized monomers, preferably having coamplexing sites, and cross-linking monomers, which are copolymerized using a suitable initiator. The complexing sites of the recognitive polymeric hydrogel that is formed preferably mimics receptor sites of a target biological tissue, biological recognition, or biological mechanism of action. The system in accordance with an embodiment of the intention is a contact lens for delivering a drug through contact with an eye.
Owner:AUBURN UNIV
Harnessing network biology to improve drug discovery
InactiveUS20060160109A1Enable optimizationMaterial nanotechnologyPeptide librariesToxicantOn pathway
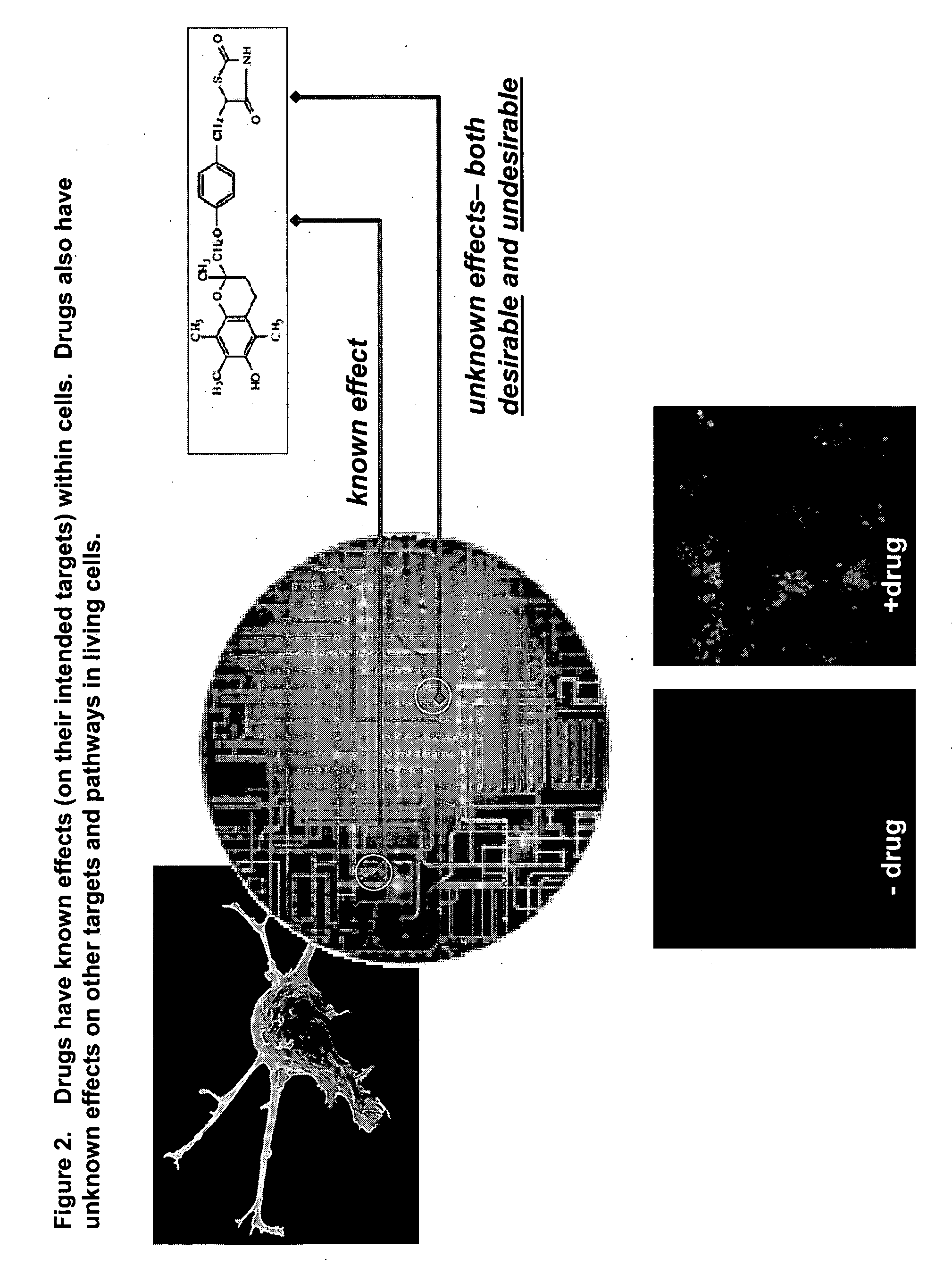
This invention provides principles, methods and compositions for ascertaining the mechanism of action of pharmacologically important compounds in the context of network biology, across the entire scope of the complex pathways of living cells. Importantly, the principles, methods and compositions provided allow a rapid assessment of the on-pathway and off-pathway effects of lead compounds and drug candidates in living cells, and comparisons of lead compounds with well-characterized drugs and toxicants to identify patterns associated with efficacy and toxicity. The invention will be useful in improving the drug discovery process, in particular by identifying drug leads with desired safety and efficacy and in effecting early attrition of compounds with potential adverse effects in man.
Owner:ODYSSEY THERA INC
Antimicrobial compositions and fibres incorporating the same
InactiveUS20120082711A1Reduces difficulty and disadvantagePreventing the growth of a broad spectrum of bacteriaBiocideInorganic active ingredientsMasterbatchMechanism of action
The present application defines an antimicrobial composition comprising (a) at least two antimicrobial agents having different antimicrobial mechanisms of action and being present in amounts that together provide a synergistic antimicrobial effect or (b) an antimicrobial agent and a surface modifying agent, an antimicrobial masterbatch comprising antimicrobial composition (a) or (b) and a polymer carrier, an antimicrobial fibre composition comprising the antimicrobial masterbatch and a fibre substrate, an antimicrobial fibre comprising a fibre body or a fibre surface having the antimicrobial fibre composition, and a process for producing antimicrobial fibres.
Owner:NOVEKO
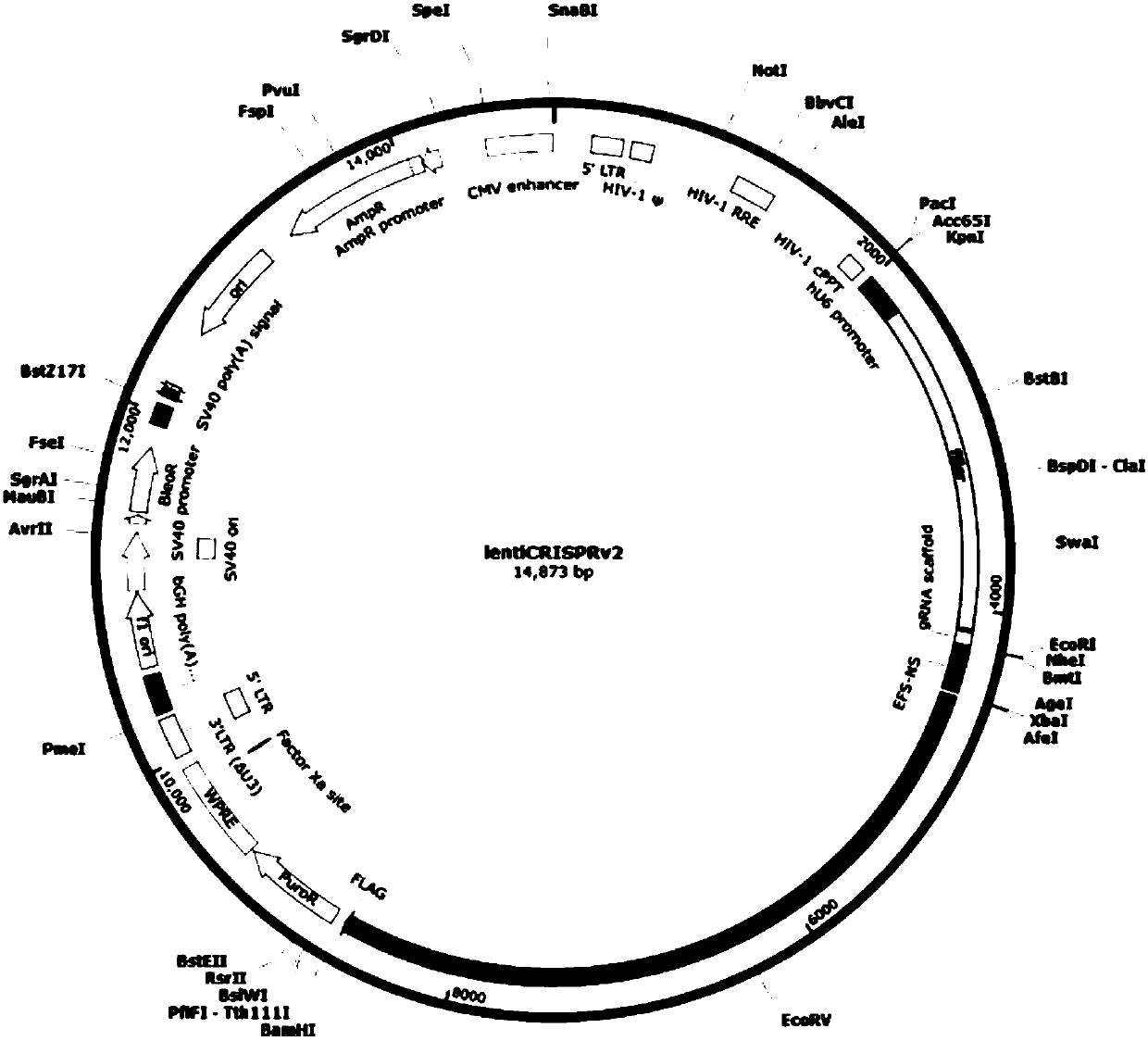
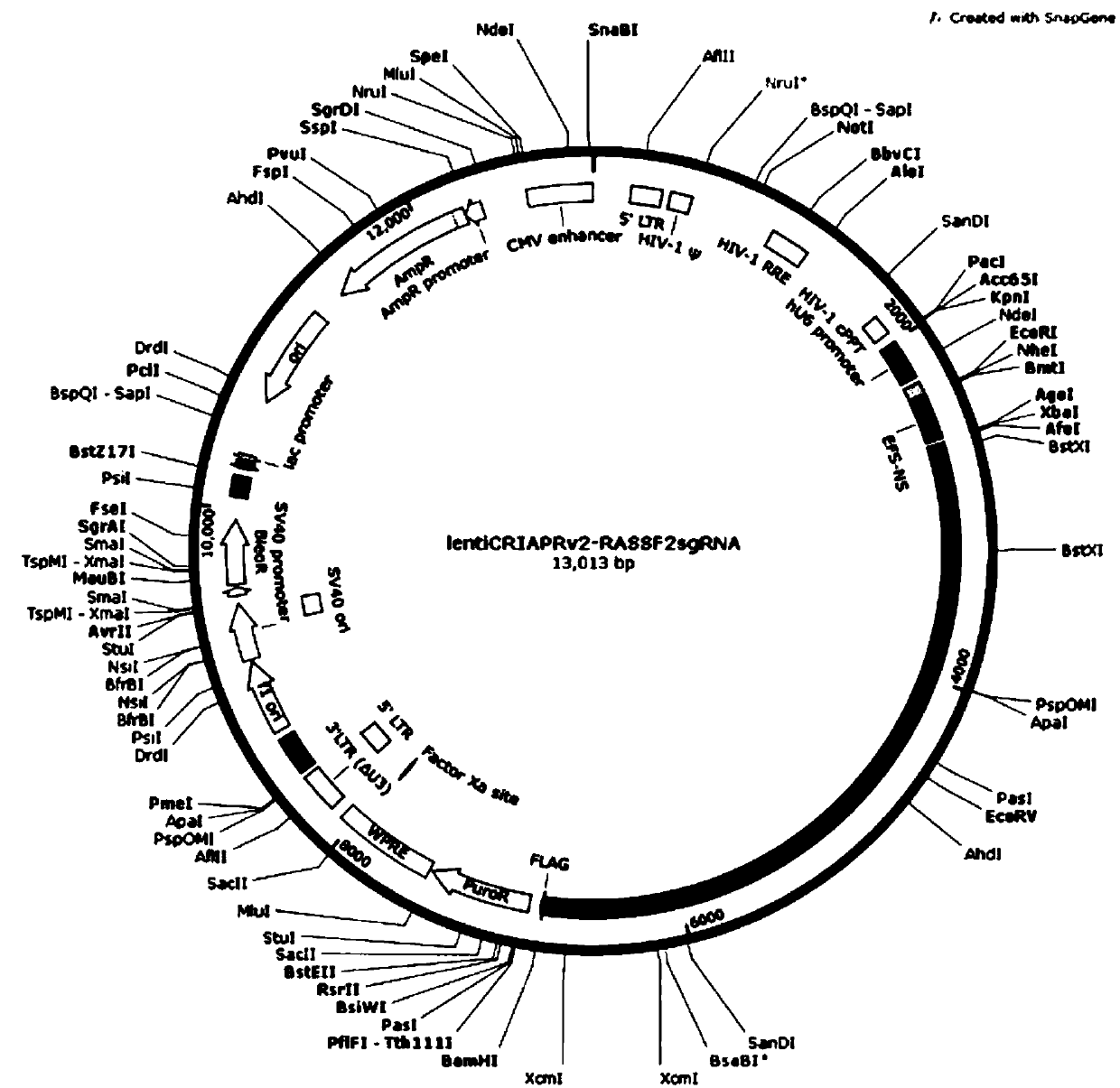
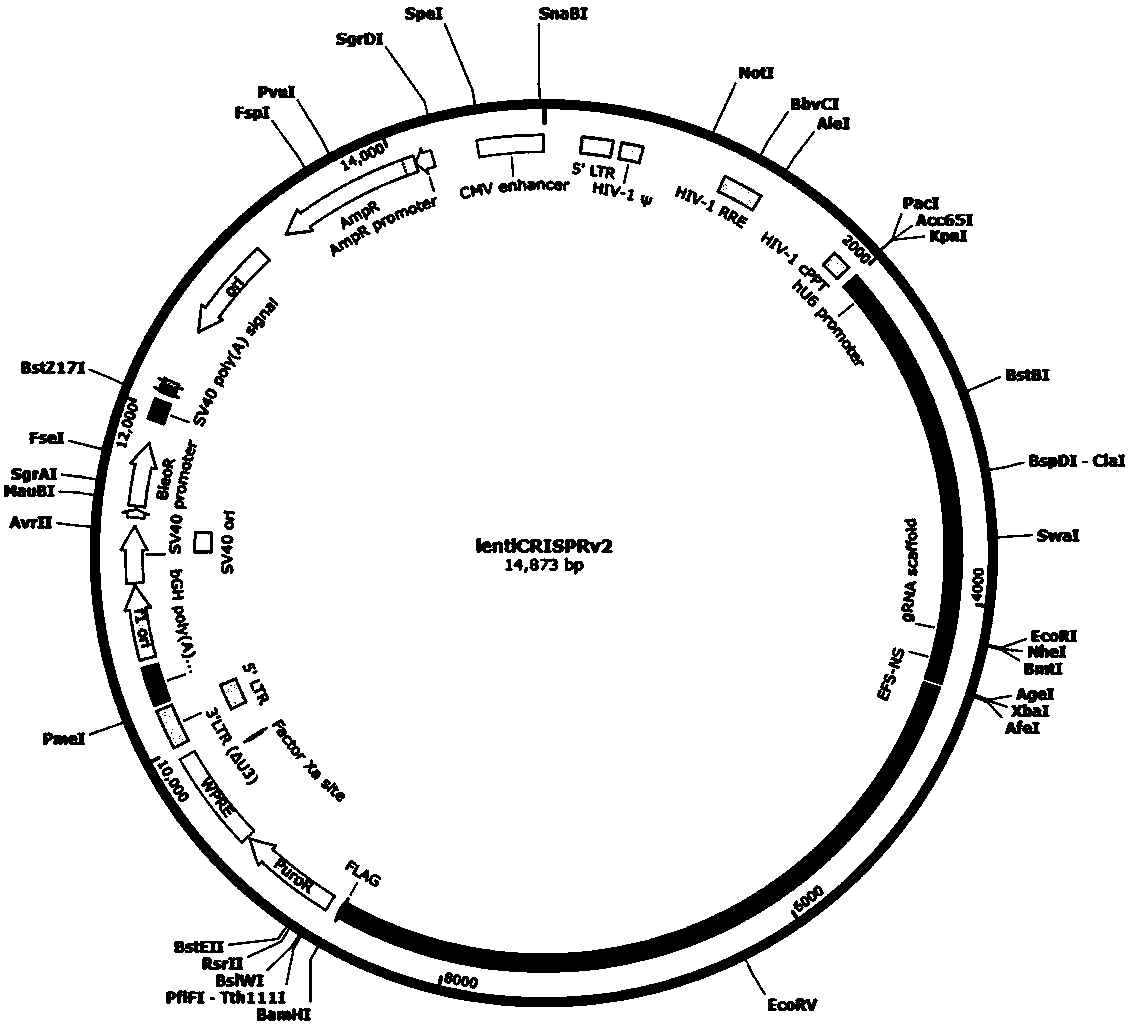
CRISPR-Cas9 targeting knockout of human breast cancer cell RASSF2 gene and specific sgRNA of RASSF2 gene
The invention discloses CRISPR-Cas9 targeting knockout of human breast cancer cell RASSF2 gene and specific sgRNA of RASSF2 gene. According to the method, the sgRNA of specific targeting RASSF2 gene is obtained, wherein the base sequence of the sgRNA is represented by SEQ ID NO.1; construction of the sgRNA of RASSF2 gene into a lentiviral vector system is carried out, wherein the lentiviral vectorsystem contains Cas9 protein; and at last, human breast cancer cell MDA-MB-231 is infected with the CRISPR-Cas9 lentivirus containing the sgRNA so as to obtain a cell strain with obviously reduced RASSF2 protein expression level. The operation and the steps are simple; the sgRNA targeting performance is excellent; cutting efficiency on RASSF2 gene is high; the constructed CRISPR-Cas9 lentiviral vector system is high in knockout efficiency, and is capable of realizing specific knockout of RASSF2 gene to obtain human breast cancer cells without RASSF2 gene, so that powerful instrument is provided for study on the action mechanisms of RASSF2 in breast cancer celles.
Owner:OBIO TECH SHANGHAI CORP LTD
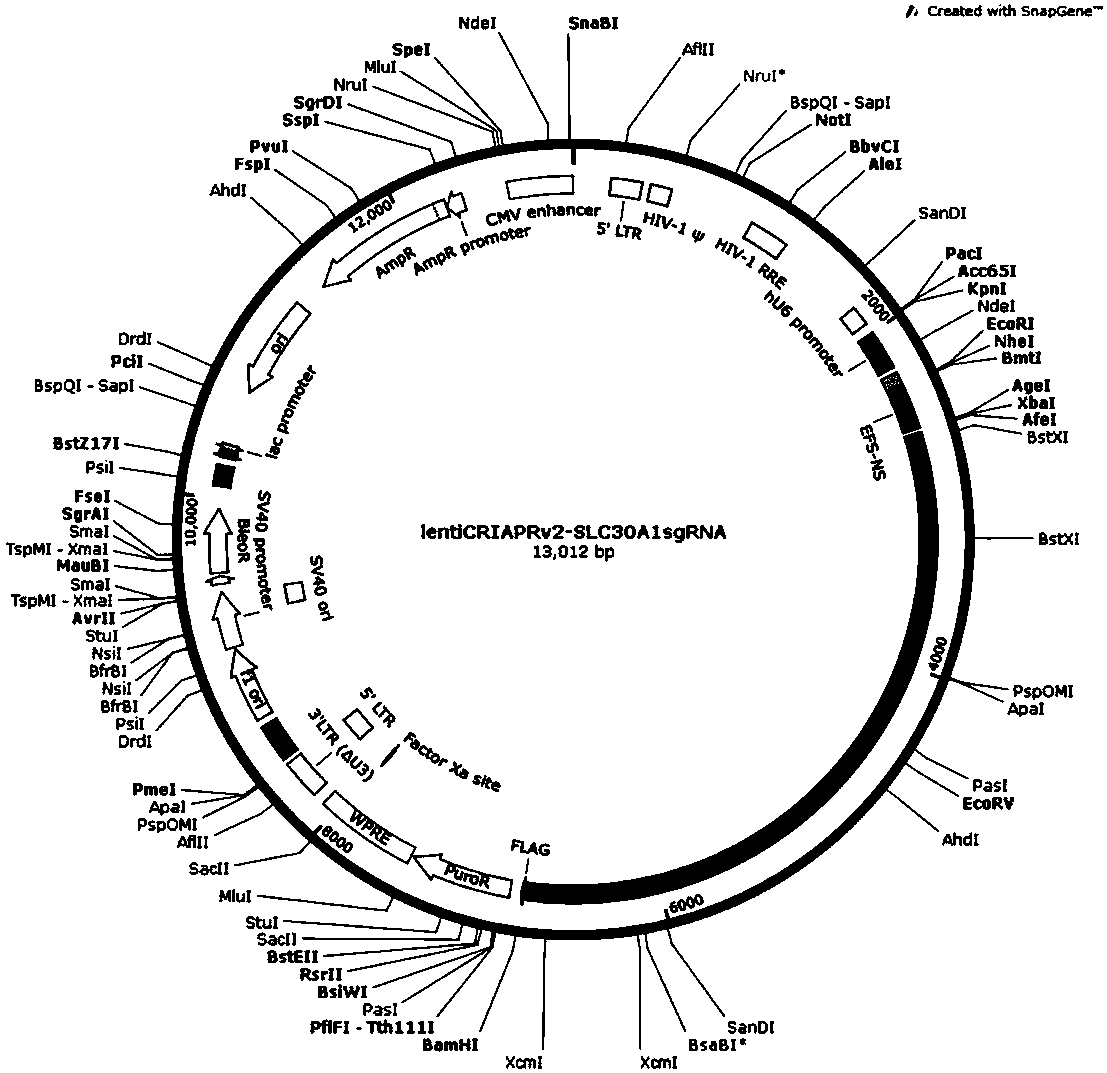
CRISPR-Cas9 targeted and knockout SLC30A1 gene and specific sgRNA of gene
The invention discloses a CRISPR-Cas9 targeted and knockout human breast cancer cell SLC30A1 gene and the specific sgRNA of the gene. Firstly, sgRNA for specific targeting of the SLC30A1 gene is obtained, and a base sequence of the sgRNA is shown in SEQ ID NO.1; secondly, the sgRNA of the SLC30A1 gene is constructed to a lentivirus vector system containing Cas9 proteins; finally, CRISPR / Cas9 lentivirus containing the sgRNA is infected with a human breast cancer cell MDA-MB-231 to obtain a cell strain of which the SLC30A1 protein expression level is obviously reduced. The CRISPR-Cas9 targeted and knockout human breast cancer cell SLC30A1 gene and the specific sgRNA of the gene disclosed by the invention have the advantages of simple operation steps, good sgRNA targetability and high cuttingefficiency on the SLC30A1 gene; in addition, the constructed CRISPR / Cas9 lentivirus system has the advantage of high knockout efficiency and can specifically knock out the SLC30A1 gene so as to obtain the human breast cancer cell having the SLC30A1 gene knocked out; therefore, a powerful tool is provided for further researching an action mechanism of the SLC30A1 in breast cancer cells.
Owner:OBIO TECH SHANGHAI CORP LTD
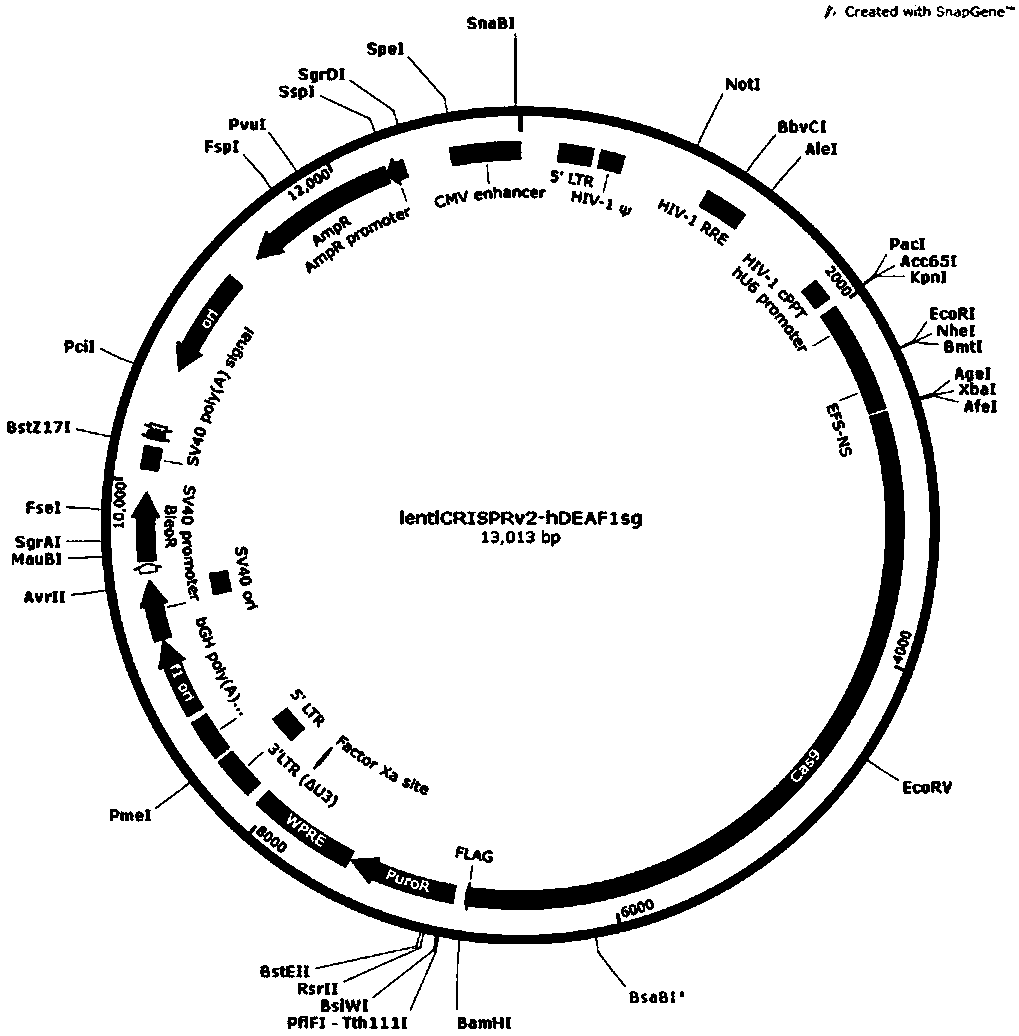
CRISPR-Cas9 targeted knockout human colorectal carcinoma cell DEAF1 gene and specific sgRNA thereof
InactiveCN108396027AConvenient researchGenetically modified cellsStable introduction of DNAVector systemLentivirus
The invention discloses a CRISPR / Cas9 targeted knockout human colorectal carcinoma cell DEAF1 gene and specific sgRNA thereof. The CRISPR / Cas9 targeted knockout human colorectal carcinoma cell DEAF1 gene and the specific sgRNA thereof are characterized in that: firstly, sgRNA of a second exon of the specific targeted DEAF1 gene is obtained and the base sequence of the sgRNA is shown as SEQ ID NO.1; secondly, the sgRNA of the DEAF1 gene is constructed into a lentiviral vector system, which contains Cas9 protein; finally, the CRISPR / Cas9 lentivirus containing the sgRNA is infected with human colorectal carcinoma cell HT-29 cell, so that a cell strain of which DEAF1 protein expression level is obviously reduced is obtained. The CRISPR / Cas9 targeted knockout human colorectal carcinoma cell DEAF1 gene disclosed by the invention has the advantages of simple operation steps, good sgRNA target ability and high cutting efficiency for the DEAF1 gene; in addition, the constructed CRISPR / Cas9 lentiviral vector system has the advantage of high knockout efficiency and can specifically knock out the DEAF1 gene to obtain the human colorectal carcinoma cells knocking out the DEAF1 gene, and therebya powerful tool is provided for further studying an action mechanism of DEAF1 in the colorectal carcinoma cells.
Owner:OBIO TECH SHANGHAI CORP LTD
CRISPR/Cas9 targeted knockout human intestinal cancer cell RITA gene and specific sgRNA thereof
InactiveCN107893075AConvenient researchGenetically modified cellsNucleic acid vectorIntestinal CancerLentivirus
The present invention discloses CRISPR / Cas9 targeted knockout human intestinal cancer cell RITA gene and specific sgRNA thereof. First, sgRNA specifically targeted to a second exon of the RITA gene isobtained, and the base sequence of the sgRNA is as shown in SEQ ID NO. 1; secondly, a sgRNA lentiviral vector system of the RITA gene is constructed, and the sgRNA lentiviral vector system contains Cas9 protein; and finally human intestinal cancer HT-29 cells are infected with CRISPR / Cas9 lentivirus containing the sgRNA to obtain a cell line which is significantly reduced in RITA protein expression level. The invention has the advantages of simple operation steps, good sgRNA targeting property, and high RITA gene cutting efficiency; furthermore, the constructed CRISPR / Cas9 lentivirus system has the advantage of high knock-out efficiency and can specifically knock out the RITA gene to obtain RITA gene-knockout human intestinal cancer cells, and a powerful tool for the further study of theaction mechanism of RITA in the intestinal cancer cells is provided.
Owner:OBIO TECH SHANGHAI CORP LTD
Composition of JS399-19 and triazole and application thereof
InactiveCN101595890ALower doseReduce the labor required for pesticide applicationBiocideFungicidesDiseasePesticide residue
The invention provides a pesticide composition and application thereof, wherein the composition comprises the active components, namely JS399-19 and a triazole bactericide, and the composition is suitable for controlling agricultural diseases. The pesticide composition has the following advantages: (1) the composition shows excellent synergistic action in certain proportioning range, the sterilizing effect of the composition is improved obviously compared with a single agent, and simultaneously the dosage, the application labor and the cost of the pesticide are reduced, and harmful effect and pesticide residue to the environment are also reduced; (2) the two active components in the composition have different mechanisms of action, and the application of the composition can retard or overcome the drug resistance of pathogenic bacteria and prolong the service life of the single agent; and (3) the composition has wide control spectrum and can control various crop diseases.
Owner:SHENZHEN NOPOSION AGROCHEM
CRISPR-Cas9 targeted-knockout human colorectal cancer cell PPP1R1C gene and specific sgRNA thereof
The invention discloses a CRISPR-Cas9 targeted-knockout human colorectal cancer cell PPP1R1C gene and a specific sgRNA sequence. The invention comprises the following steps: firstly, acquiring sgRNA of a specific targeting PPP1R1C gene functional area, wherein a base sequence of the sgRNA is shown as SEQ ID NO.1; then, constructing an sgRNA-lentiviral vector system of the PPP1RQC gene, wherein thesystem contains Cas9 protein; and finally, infecting human colorectal cancer HT-29 cells with CRISPR / Cas9 lentivirus containing the sgRNA, so that a cell line that a PPP1R1C protein expression levelis obviously reduced is obtained. The invention is simple in operating steps, good in sgRNA targeting performance and high in cutting efficiency on the PPP1R1C gene; the constructed CRISPR / Cas9 lentiviral system is high in knockout efficiency and is capable of achieving specific knockout of the PPP1R1C gene, so that the human colorectal cancer cells that the PPP1R1C gene is knocked out is obtained; therefore, a powerful tool is provided for further researching an action mechanism of the PPP1R1C in the colorectal cancer cells.
Owner:OBIO TECH SHANGHAI CORP LTD
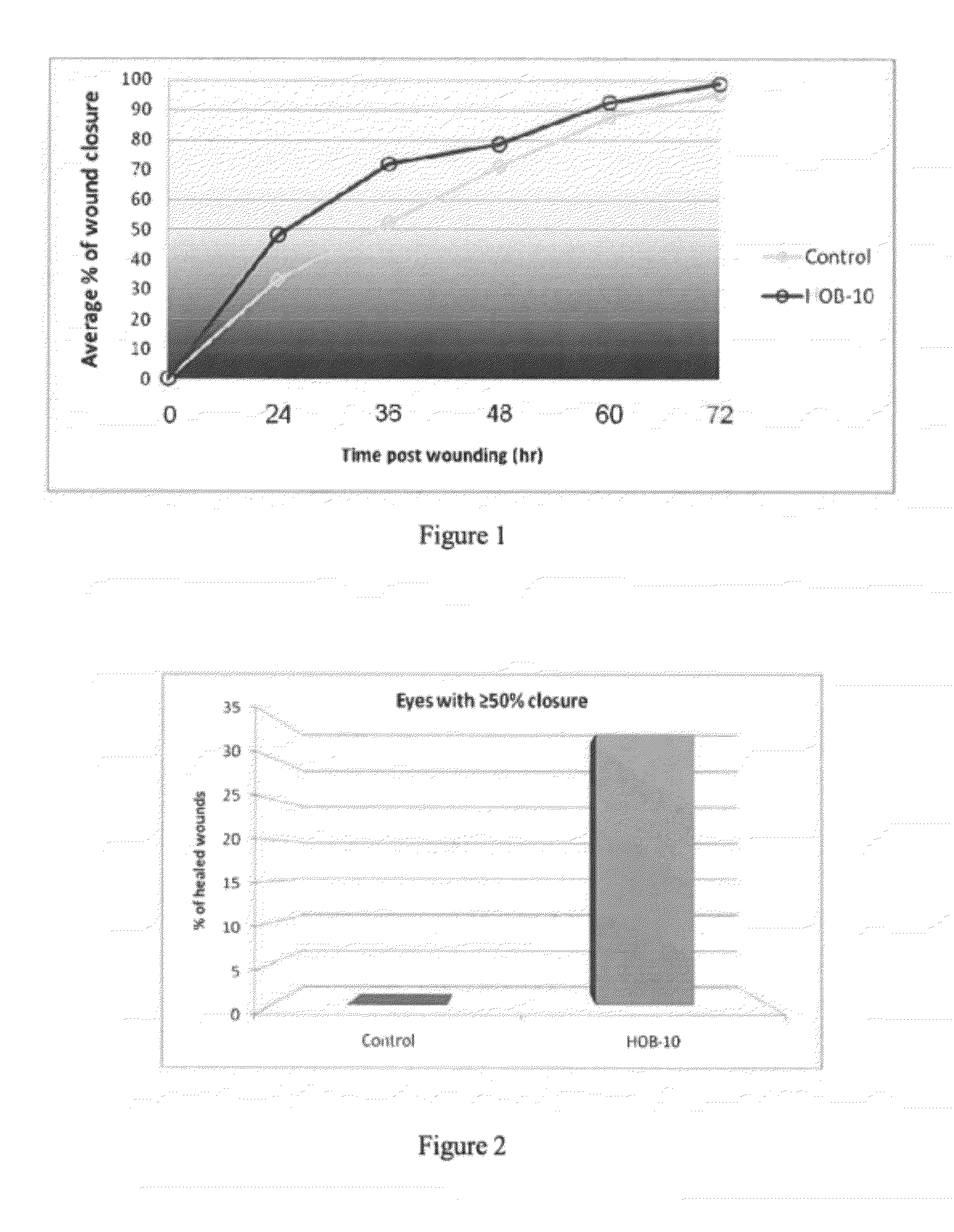
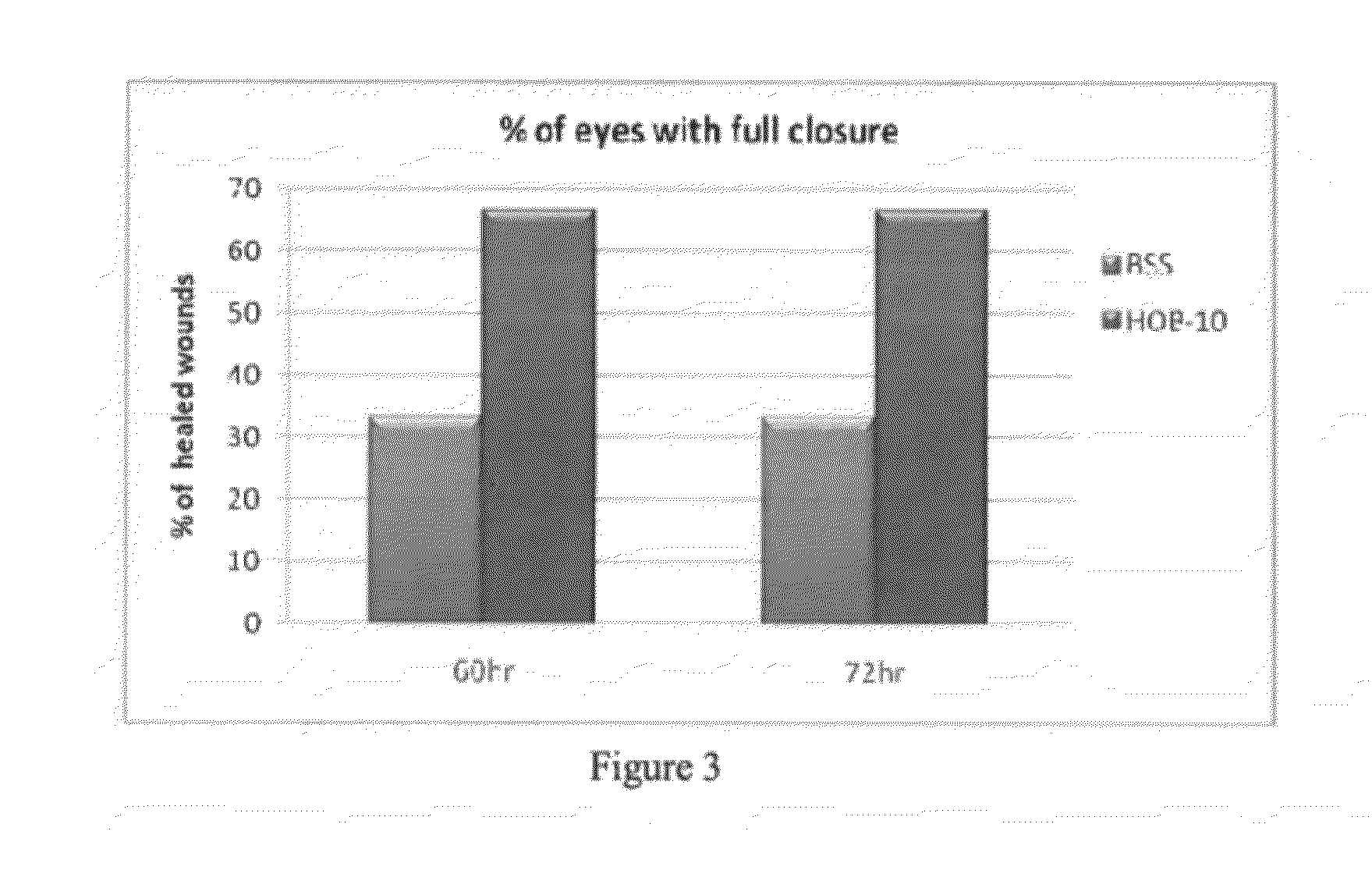
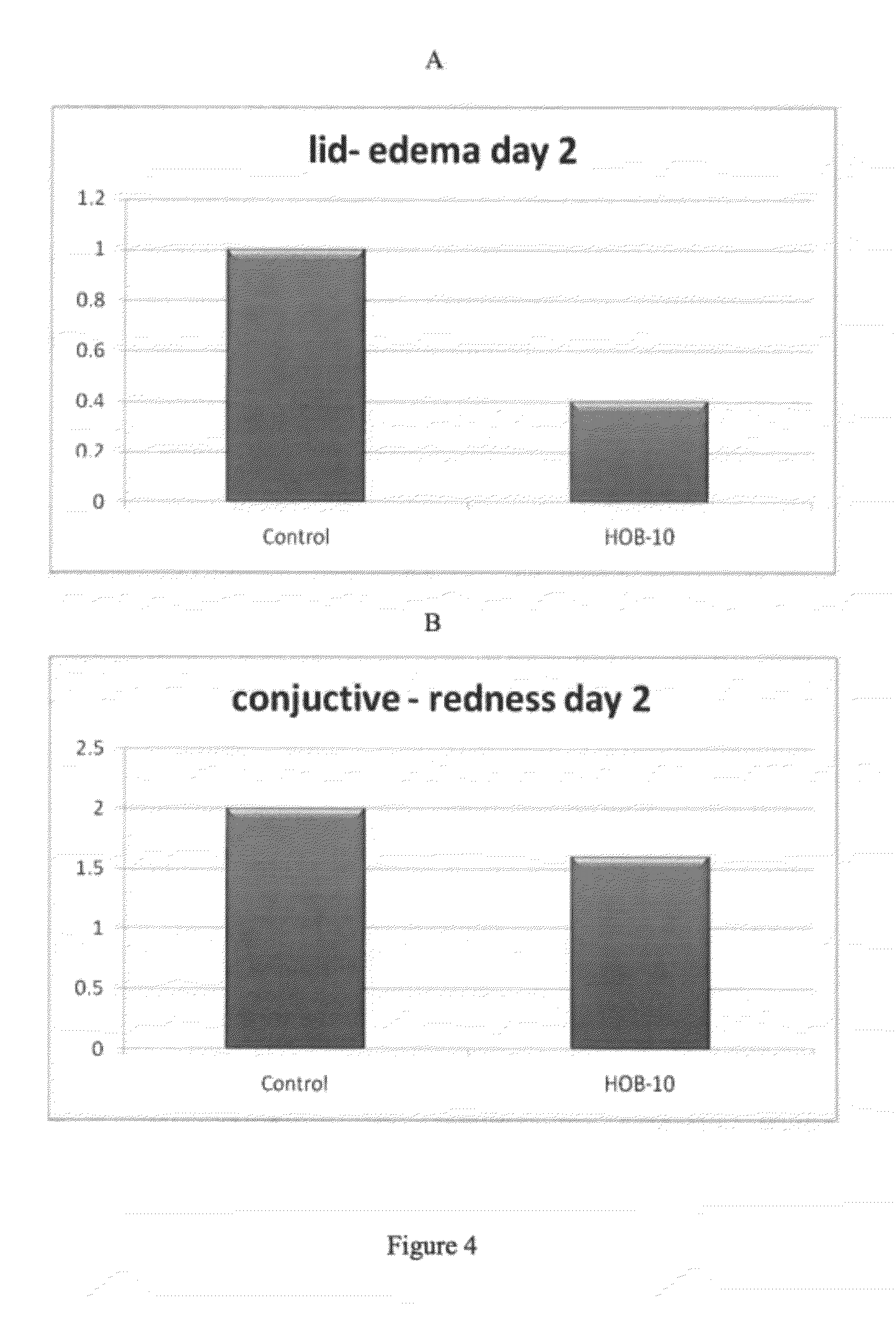
Buffered ophthalmic compositions and methods of use thereof
InactiveUS20120264681A1Accelerating and promoting healingPrevent or inhibit ocular tissue injury or woundingBiocideOrganic active ingredientsDiseaseActive agent
The present disclosure provides a buffered ophthalmic composition for formulation of topically administrable suspensions useful for treating eye disorders by promoting wound healing, delivery of pharmaceutically active agents, and lubricating the eye. In particular the ophthalmic composition includes a buffer solution compatible with application to a mammalian eye, wherein the buffer provides increased mechanism of action of pharmaceutically active agents as well as therapeutic qualities. The ophthalmic composition exhibits dual therapeutic action to alleviate various eye disorders as it concomitantly treats corneal ulcerations and excessive inflammation which results from various eye injuries.
Owner:HEALOR LTD
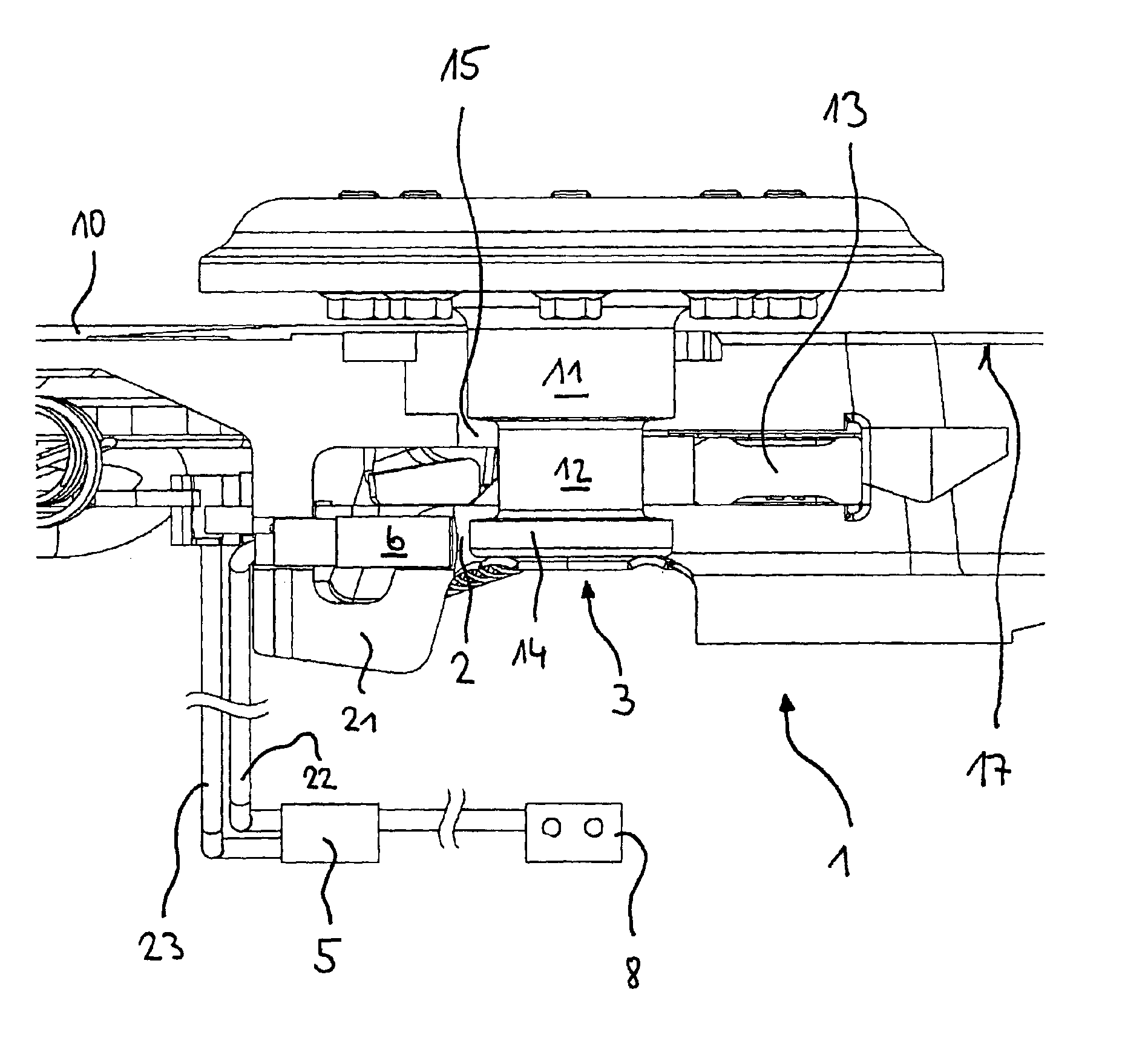
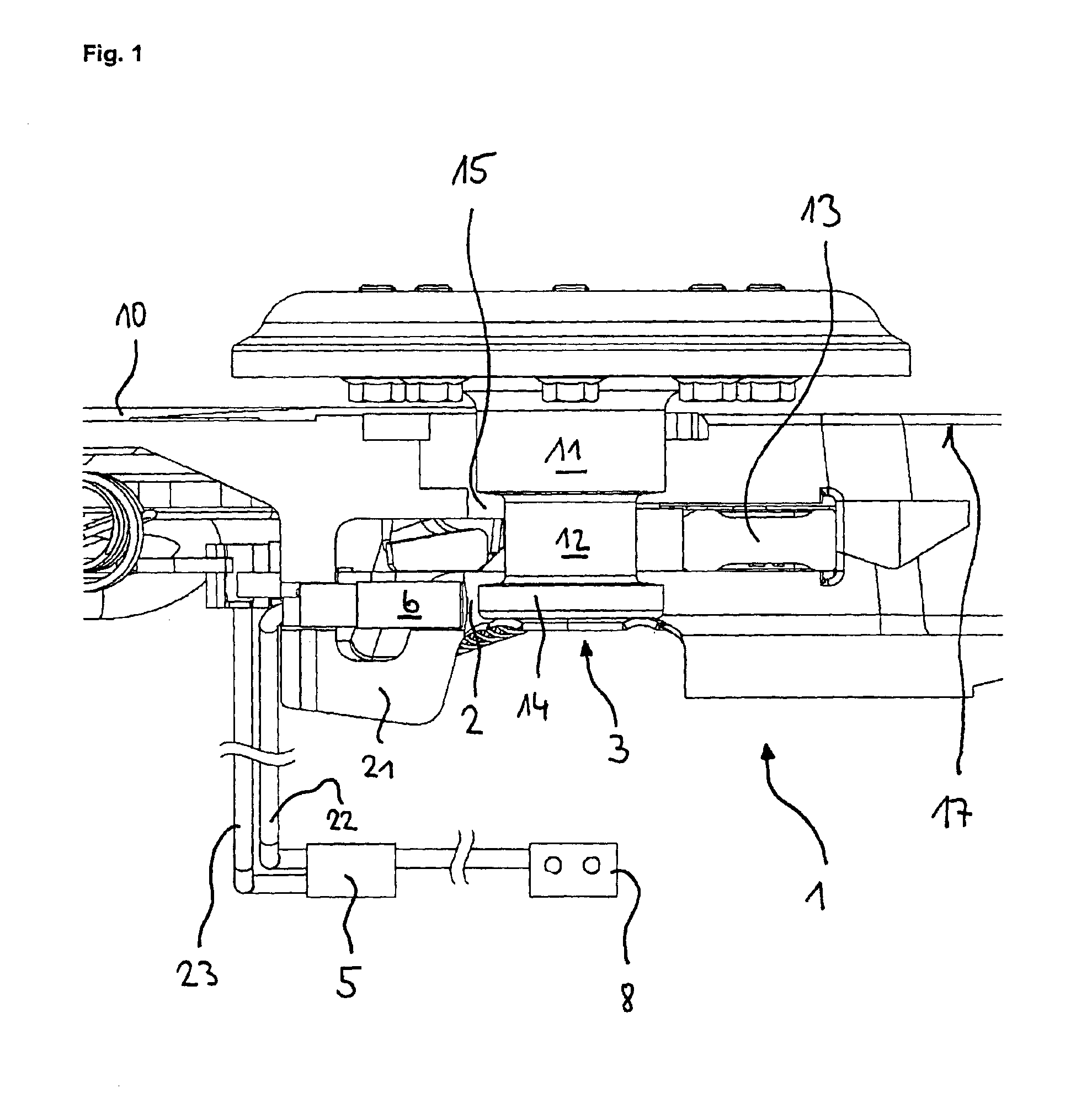
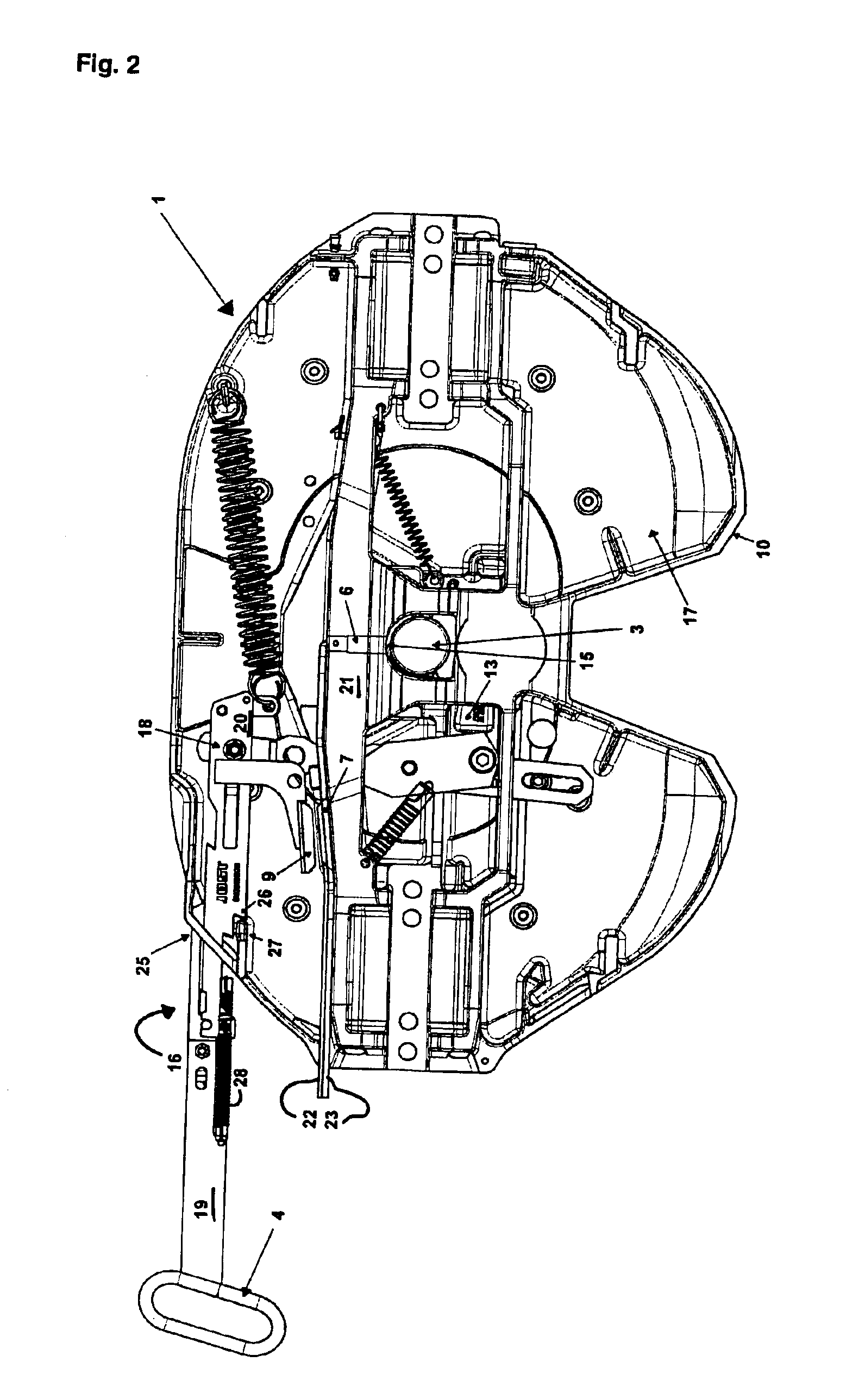
Device for indicating the locking state of a fifth wheel coupling and sensor arrangement
InactiveUS6866283B2Detection of defectLess subject to mechanical loadingAutomatic initiationsTractor-trailer combinationsCouplingMechanism of action
A device is described for indicating the locking state of a fifth wheel coupling and an arrangement of a first and a second sensor. According to the prior art, the first sensor is arranged on the underside of the locking latch and monitors the position of the kingpin in relation to the locking latch. A second sensor that is used is an inductive proximity switch that monitors a safety mechanism against loosening. In practice, this type of positioning of the first sensor has led to damage of the locking latch and the first sensor, while the signals of the second sensor were often false signals. Thus, the object of the invention is to provide a device for indicating the locking state, which maximizes operational availability and minimizes false signals. A further object of the invention is to optimize the arrangement of the first and the second sensor. These objects are attained by arranging the first sensor detecting the kingpin in the area of the locating hole and configuring the second sensor as a magnetically sensitive sensor that interacts with a magnet mounted on the operating lever. The two sensors are based on different mechanisms of action.
Owner:JOST WERKE
Preparation method for cement auxiliary-grinding strengthener of polyethylene glycol amine ester carboxylic acid system
The invention belongs to the technical field of cement auxiliary-grinding agents, and in particular to a preparation method for a cement auxiliary-grinding strengthener of a polyethylene glycol amine ester carboxylic acid system. The preparation method comprises the following steps: (1) performing esterification reaction on alcohol amines and unsaturated carboxylate in a molar mass ratio to obtain esterification monomers which contain polar groups, such as hydroxyl and carboxyl; (2) adding unsaturated large monomers and water into a reaction container, stirring, heating and dissolving, respectively adding an initiator, the esterification monomers and a chain transfer agent dropwise, preserving the heat after the reaction is performed for a certain period of time, and adjusting the pH of a solution to be neutral after the reaction is completed so as to prepare the cement auxiliary-grinding strengthener of the polyethylene glycol amine ester carboxylic acid system. The cement auxiliary-grinding strengthener fully plays the action mechanism of the polar functional groups, such as the hydroxyl and the carboxyl, on cement; when the using amount is 0.01 to 0.1 percent of the mass of the cement, the yield each grinding machine per hour is improved by 12 to 25 percent, the 3-day strength of the cement can be increased by 2 to 5 MPa, the 28-day strength of the cement can be increased by 4 to 8 MPa, and the aims of stable performance and remarkable effects of auxiliary grinding and strengthening are fulfilled.
Owner:KZJ NEW MATERIALS GROUP CO LTD
Controlled and extended delivery of hyaluronic acid and comfort molecules via a contact lens platform
ActiveUS8388995B1Avoid effectivenessImprove bioavailabilityPharmaceutical delivery mechanismPharmaceutical non-active ingredientsCross-linkDiclofenac Sodium
A drug delivery system is disclosed. The drug delivery system includes a recognitive polymeric hydrogel through which a drug is delivered by contacting biological tissue. The recognitive polymeric hydrogel is formed using a bio-template, which is a drug or is structurally similar to the drug, functionalized monomers, preferably having complexing sites, and cross-linking monomers, which are copolymerized using a suitable initiator. The complexing sites of the recognitive polymeric hydrogel that is formed mimic receptor sites of a target biological tissue, biological recognition, or biological mechanism of action. A system in accordance with some embodiments is a contact lens for delivering a drug through contact with an eye. In some embodiments, the drug is an anti-microbial, such as an anti-fungal agent for treatment of large animals. In some embodiments, a comfort molecule hyaluronic acid (HA) is delivered. In some embodiments, ketotifen fumarate (anti-histamine) and / or diclofenac sodium (anti-inflammatory) are delivered.
Owner:AUBURN UNIV
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com