A pan-type inert carrier Escherichia coli and its potential application
An inert carrier, Escherichia coli technology, applied in the field of biomedical technology detection, can solve problems such as non-agglutination, and achieve the effects of perfecting poor specificity, improving poor specificity, huge application value and market prospect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
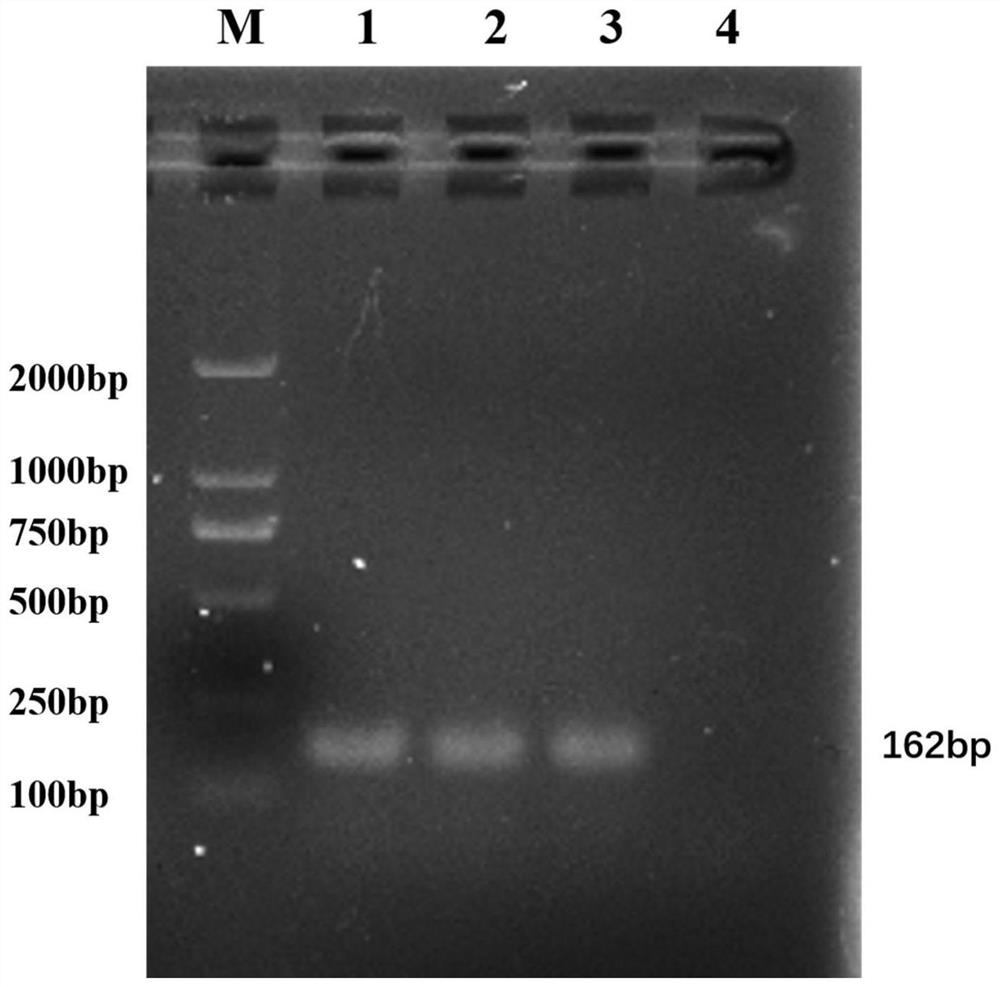
[0039] Example 1 Acquisition and verification of pan-type inert carrier E. coli SE1H
[0040] Inoculate the inert carrier Escherichia coli SE1 (preservation number is CGMCC No.17339) in the LB liquid medium, shake at 37°C for 12 hours, draw 30 μL of the bacterial liquid into the LB solid medium for streak culture, and culture at 37°C for 16-18 hours Obtain the second-generation colony, pick the second-generation single colony and inoculate it in LB liquid medium, and use LB liquid and solid medium as a cycle to cultivate alternately. Starting from the 40th generation, the single colony obtained is a pan-type inert carrier bacterium SE1H, in fact, through the 41st to 60th generations of continuous subculture, any generation of them also has the characteristics of the pan-type inert carrier bacteria SE1H.
[0041] Take 1 mL of the SE1H cultured overnight to prepare DNA template by boiling method. The Escherichia coli β-glucuronidase gene uidA was amplified by PCR, identified ...
Embodiment 2
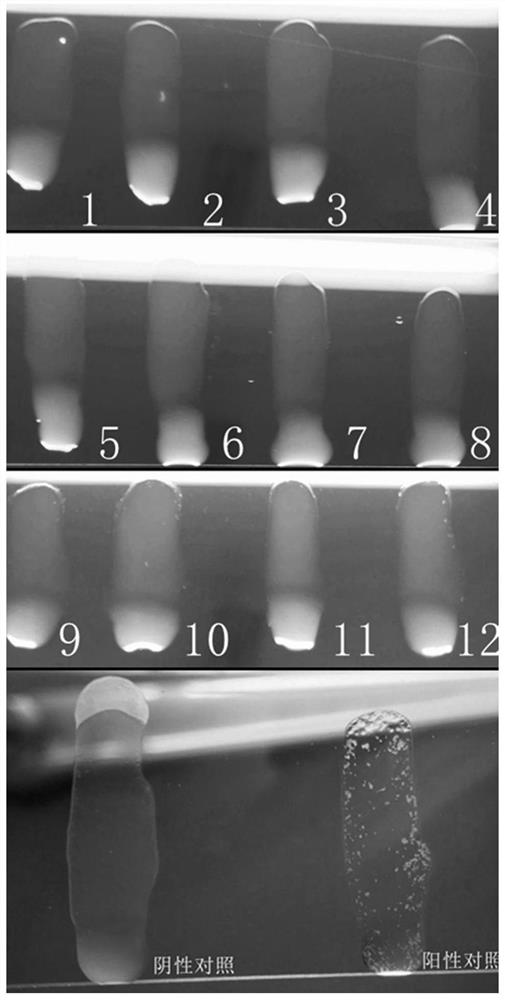
[0049] Example 2 Test of no non-specific agglutination phenomenon of pan-type inert carrier Escherichia coli SE1H and human and animal source serum and whole blood of different backgrounds
[0050] Centrifuge the Escherichia coli SE1H bacterial solution obtained from the overnight culture in Example 1 at 4° C. at 4000 rpm for 10 min, discard the supernatant, resuspend the bacteria sludge with sterile saline, wash by centrifugation three times, and then resuspend to a concentration gradient of different concentrations of bacteria. Before the test, the bacterial liquid was mixed with a vortex instrument, and the agglutination test was performed with sterile saline and SPF chicken serum to ensure that the test bacterial liquid had no self-agglutination and no non-specific agglutination. In the ultra-clean bench (20 ° C ~ 25 ° C), take several pieces of ordinary glass plates with clean surfaces, and use sterile pre-cooled PBS to 4 ° C to resuspend and wash the carrier bacteria for ...
Embodiment 3
[0056] Example 3 Test and verification of the surface expression of pan-type inert carrier bacteria Escherichia coli SE1H and the ability to carry the avian-derived Salmonella antigenic factor P
[0057] (1) Amplification of gene p encoding antigenic factor P expressed by avian Salmonella
[0058] According to the whole genome sequence of Salmonella pullorum ATCC 9120 strain in NCBI GenBank (NCBI accession number: CP012347.1), the whole genome sequence of Salmonella pullorum S44987_1 strain (NCBI accession number: LK931482.1), the whole genome sequence of Salmonella pullorum S06004 strain ( NCBI accession number: CP006575.1), whole genome sequence of Salmonella pullorum QJ-2D-Sal strain (NCBI accession number: CP022963.1), whole genome sequence of Salmonella gallinarum typhi 287 / 91 strain (NCBI accession number: AM933173.1) , Salmonella gallinarum typhi 9184 strain genome sequence (NCBI accession number: CP019035.1) published full-length genome sequence, search and compare the...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap