Recombinant Anti-cd30 antibodies and uses thereof
a technology of cd30 and anti-cd30, which is applied in the field of recombinant anti-cd30 antibodies, can solve the problems of limiting the ability to deliver curative doses of these agents, no patient experienced tumor regression, and known anti-cd30 antibodies have not been shown to inhibit the proliferation of hd cells in culture, so as to reduce the incorporation of 3h-thymidin
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
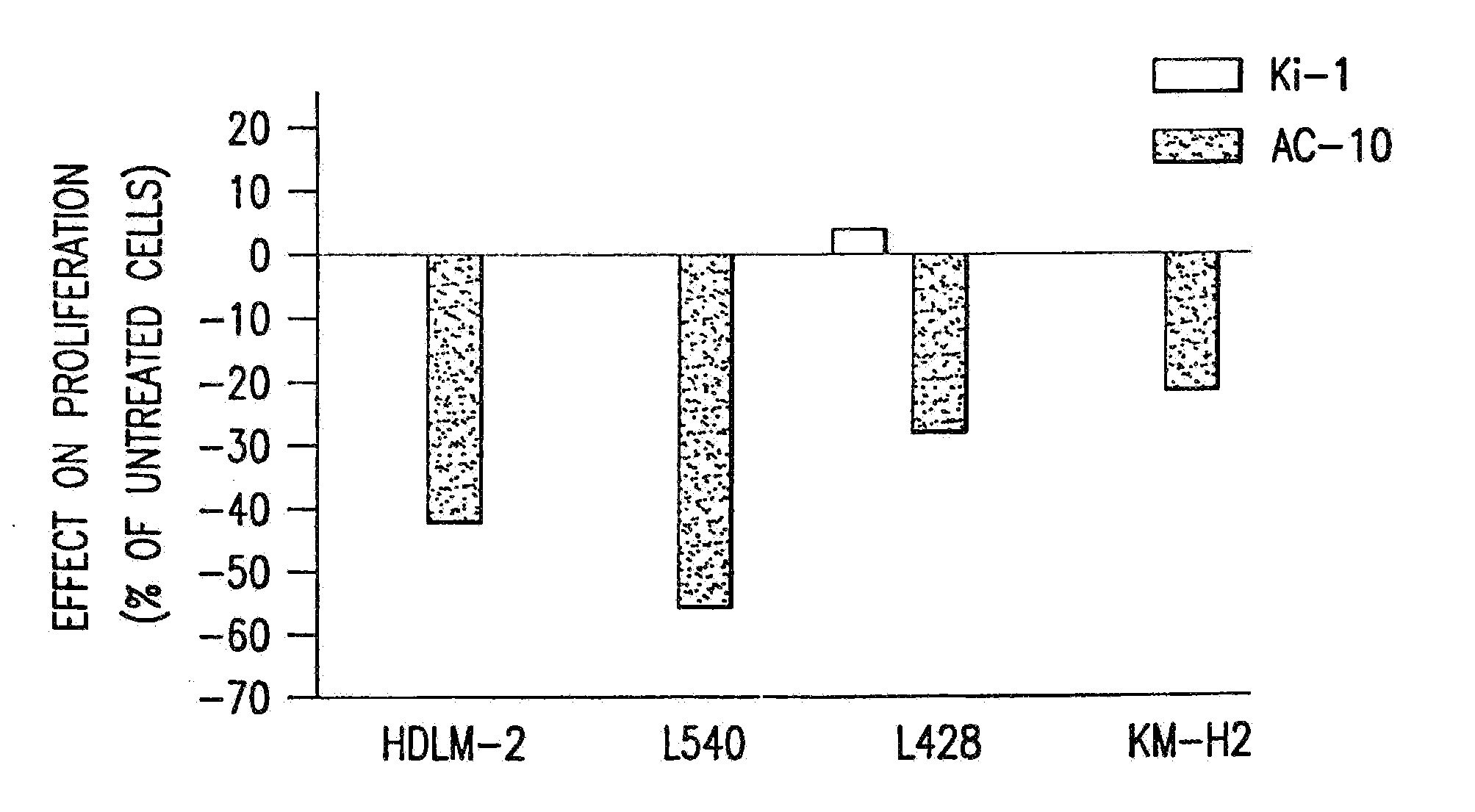
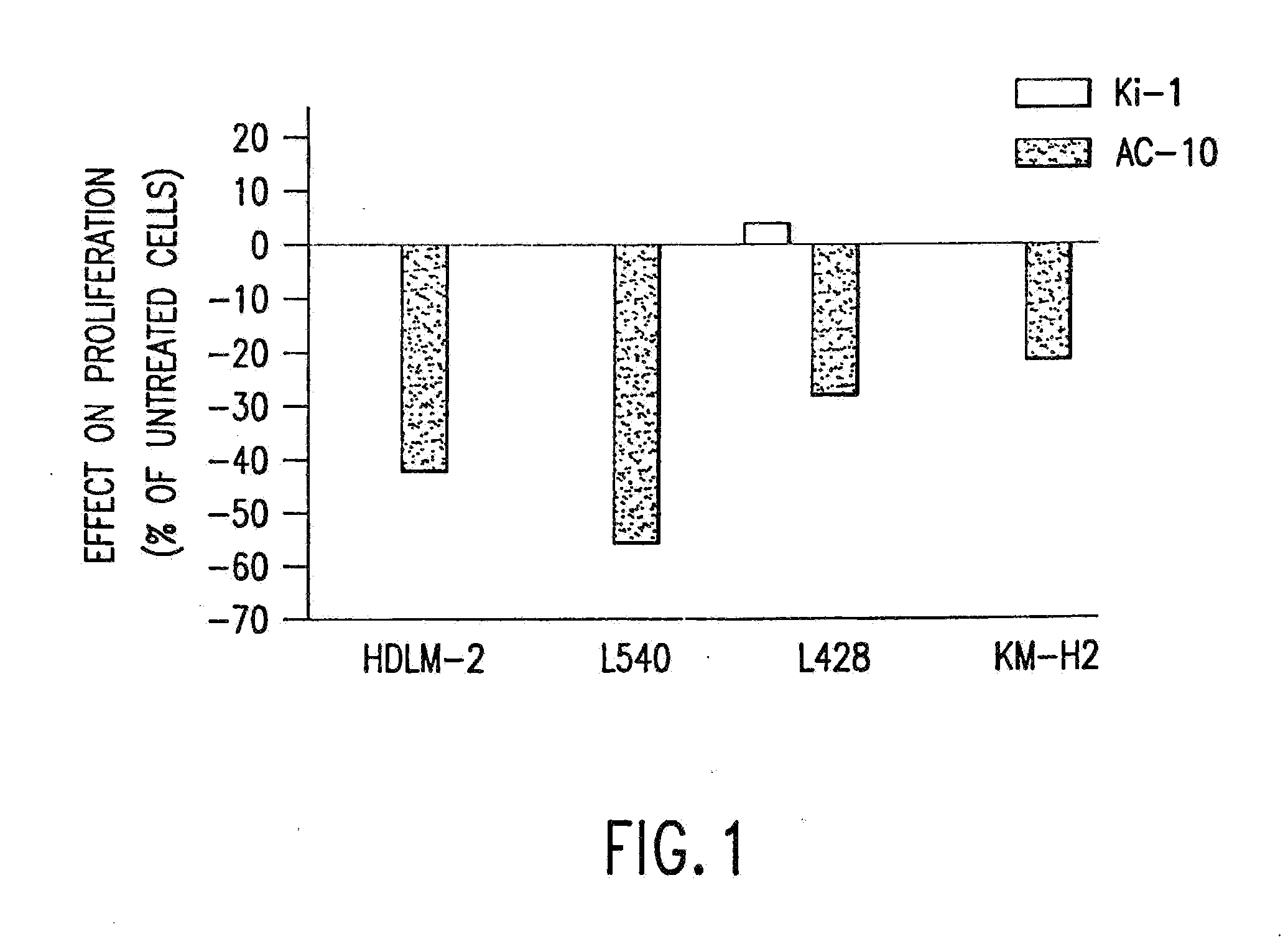
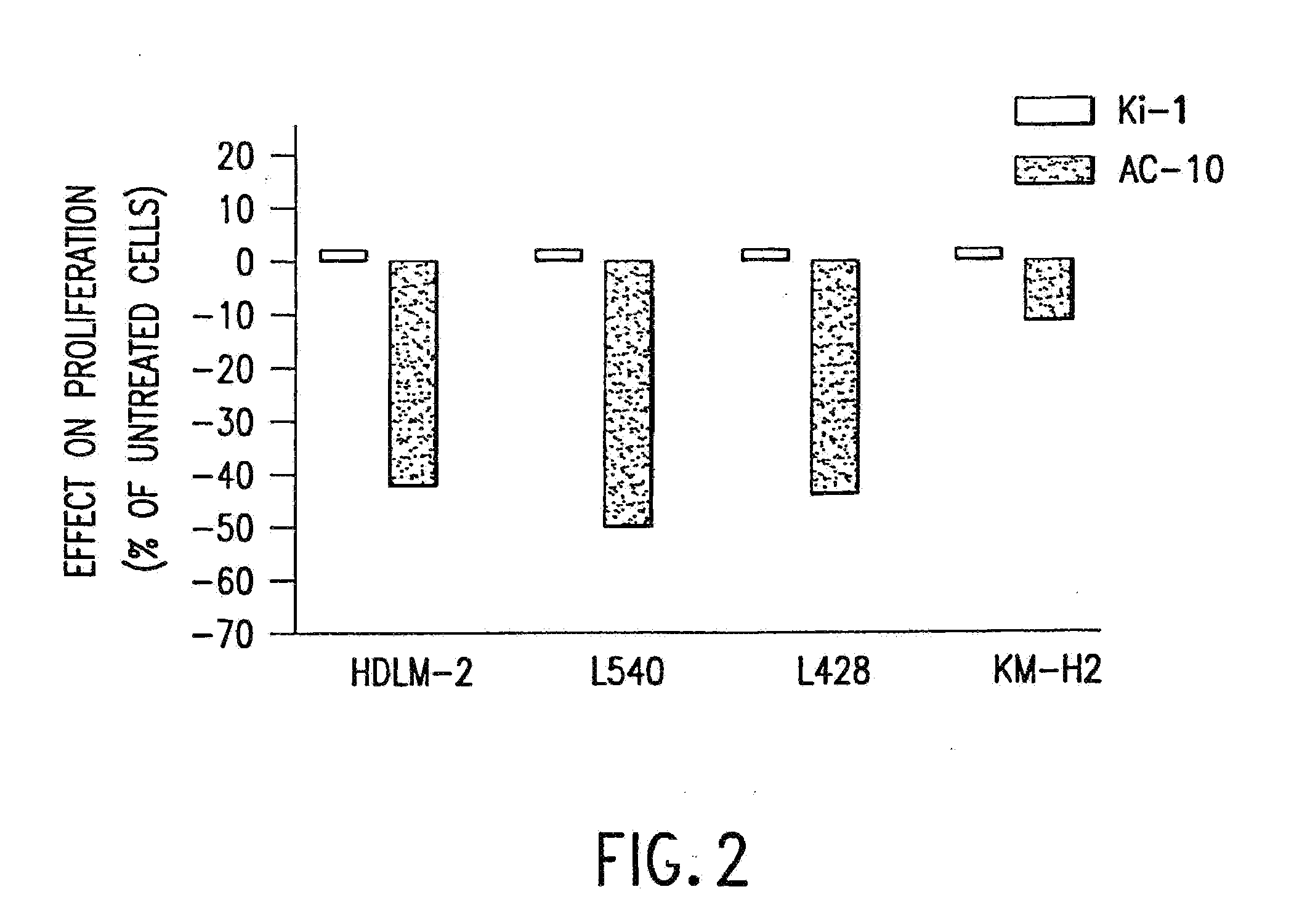
[0094]The present invention relates to proteins that bind to CD30 and exert a cytostatic or cytotoxic effect on HD cells. The invention further relates to proteins that compete with AC10 or HeFi-1 for binding to CD30 and exert a cytostatic or cytotoxic effect on HD cells. In one embodiment, the protein is an antibody. In a preferred mode of the embodiment, the antibody is AC10 or HeFi-1, most preferably a humanized or chimeric AC10 or HeFi-1.
[0095]The invention further relates to proteins encoded by and nucleotide sequences of AC10 and HeFi-1 genes. The invention further relates to fragments and other derivatives and analogs of such AC10 and HeFi-1 proteins. Nucleic acids encoding such fragments or derivatives are also within the scope of the invention. Production of the foregoing proteins, e.g., by recombinant methods, is provided.
[0096]The invention also relates to AC10 and HeFi-1 proteins and derivatives including fusion / chimeric proteins which are functionally active, i.e., whic...
PUM
| Property | Measurement | Unit |
|---|---|---|
| area | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More