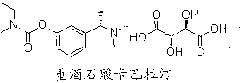Rivastigmine orally disintegration tablet and preparation method thereof
A technology of rivastigmine bitartrate and orally disintegrating tablets, which can be applied to pharmaceutical formulas, medical preparations of non-active ingredients, drug combinations, etc., and can solve problems such as research reports on rivastigmine bitartrate orally disintegrating tablets. , to achieve the effects of stable and easy-to-control preparation quality, good medication compliance, and appropriate hardness
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0048] prescription
[0049]
[0050] method All the components in the prescription are crushed into fine powder with a fineness of more than 100 mesh; the prescription amount of rivastigmine bitartrate, lactose, mannitol, microcrystalline cellulose, citric acid, orange essence and menthol are mixed with 4g Mix cross-linked povidone, pass through a 100-mesh sieve three times to make the mixture uniform, then add a soft material made of povidone K30 aqueous solution with a concentration of 2% by mass, pass through a 24-mesh sieve to granulate, dry at 50°C, and dry granules Pass through a 24-mesh sieve for granulation; then add 2 g of crospovidone, sodium bicarbonate and magnesium stearate in the prescribed amount to the granulated dry granules, mix evenly, and compress into tablets to make rivastigmine bitartrate 1000 orally disintegrating tablets, each containing 1.5 mg rivastigmine bitartrate.
[0051] quality The obtained orally disintegrating tablet has a smooth ...
Embodiment 2
[0053] prescription
[0054]
[0055] method All the components in the prescription were crushed into fine powder with a fineness above 100 meshes; the prescription amount of rivastigmine bitartrate, lactose, mannitol, microcrystalline cellulose, tartaric acid, orange essence and menthol were mixed with 6.7g Mix polyvidone, pass through a 100-mesh sieve three times to make the mixture uniform, then add 2% povidone K30 aqueous solution to make a soft material, pass through a 24-mesh sieve to granulate, dry at 50°C, and dry granules Pass through a 24-mesh sieve for granulation; then add 3.3g of crospovidone, sodium bicarbonate and magnesium stearate in the prescribed amount to the granulated dry granules, mix evenly, and compress into tablets to make Kabbalah tartrate 1000 orally disintegrating tablets, each containing rivastigmine bitartrate 1.5mg.
[0056] quality The obtained orally disintegrating tablet has a smooth and beautiful surface; a hardness of 25-40 Newto...
Embodiment 3
[0058] prescription
[0059]
[0060] method All the components in the prescription are crushed into fine powders with a fineness of more than 100 mesh; the prescription amount of rivastigmine bitartrate, mannitol, microcrystalline cellulose, citric acid, aspartame and menthol are mixed with 10g Mix cross-linked povidone, pass through a 100-mesh sieve three times to make the mixture uniform, then add a soft material made of povidone K30 aqueous solution with a concentration of 2% by mass, pass through a 24-mesh sieve to granulate, dry at 50°C, and dry granules Sieve through a 24-mesh sieve for granulation; then add 5g of crospovidone, sodium bicarbonate and magnesium stearate in the prescribed amount to the granulated dry granules, mix evenly, and compress into tablets to make rivastigmine bitartrate altogether 1000 orally disintegrating tablets, each containing 1.5 mg rivastigmine bitartrate.
[0061] quality The obtained orally disintegrating tablet has a smooth a...
PUM
| Property | Measurement | Unit |
|---|---|---|
| hardness | aaaaa | aaaaa |
| hardness | aaaaa | aaaaa |
| hardness | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap