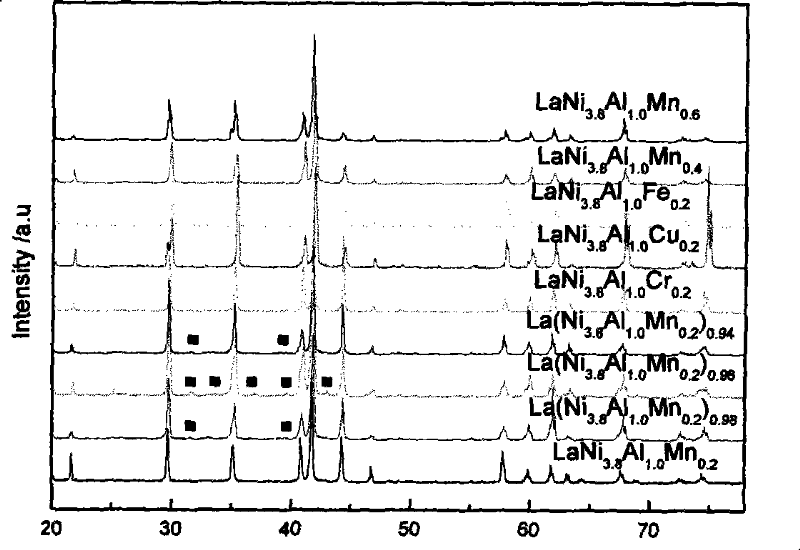
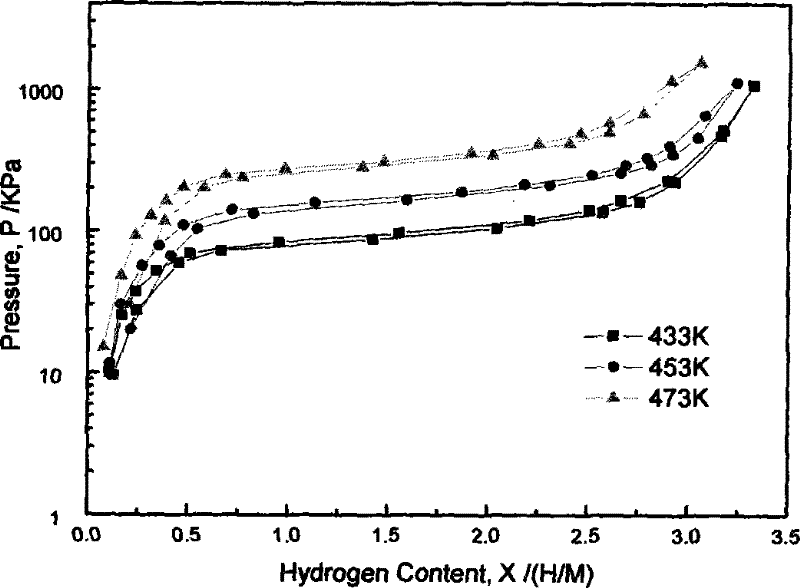
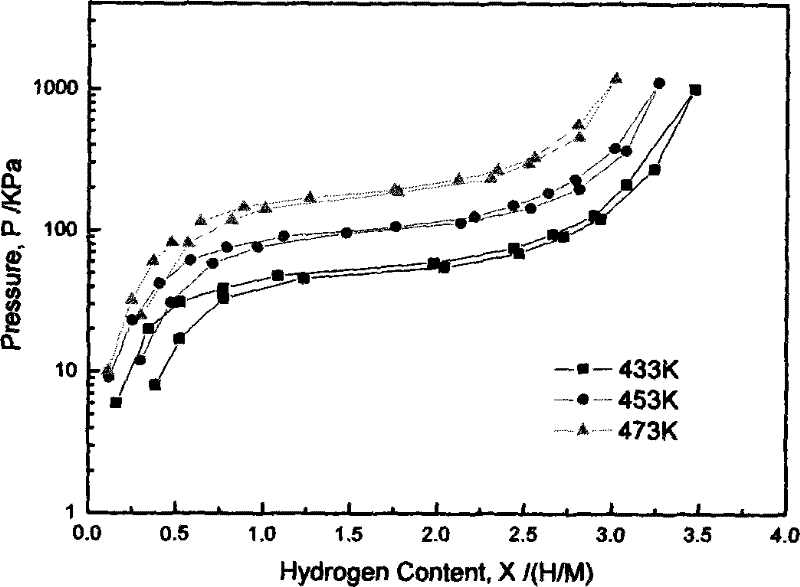
A kind of rare earth hydrogen storage alloy for metal hydride heat pump and preparation method thereof
A hydrogen storage alloy and hydride technology, which is applied in heat pumps, lighting and heating equipment, refrigerators, etc., can solve the problems of large hysteresis in hydrogen absorption and desorption, small alloy capacity, and difficulty in activation, achieving small hysteresis and good resistance to pulverization Good performance and dynamic performance, comprehensive performance
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0032] Preparation and testing of hydrogen storage alloy LaNi 3.8 Al 1.0 Cu 0.2 alloy. The preparation method, heat treatment conditions and activation method are as described above.
[0033] Test result: LaNi 3.8 Al 1.0 Cu 0.2 Under the above activation conditions, the alloy can begin to absorb hydrogen quickly after only about 1100s of incubation period, and the activation process can be completed in one time. The hydrogen storage capacity of the hydride formed at 286K is 4.70. The equilibrium pressure of hydrogen absorption at 433K is 100kPa, the equilibrium pressure of hydrogen desorption is 93kPa, the hysteresis coefficient is very small, and the kinetic speed of hydrogen absorption and desorption is very fast. The maximum hydrogen absorption capacity is reached when the initial hydrogen pressure is 1.2MPa and the temperature is 433K. 90% only takes 40s. The enthalpy changes of hydrogen absorption and desorption are -49.45KJ / Mol and 49.87KJ / Mol respectively. The enthalpy ...
Embodiment 2
[0035] The difference from Example 1 lies in: preparing and testing hydrogen storage alloy LaNi 3.8 Al 1.0 Cr 0.2 .
[0036] Test result: LaNi 3.8 Al 1.0 Cr 0.2 Under the above activation conditions, the alloy can begin to absorb hydrogen quickly after only about 120s of incubation period, and the activation process can be completed in one time. The hydrogen storage of the hydride formed at 286K is 4.86. The equilibrium pressure of hydrogen absorption at 433K is 56kPa, the equilibrium pressure of hydrogen desorption is 53kPa, the hysteresis coefficient is small, and the kinetic speed of hydrogen absorption and desorption is very fast. The maximum hydrogen absorption capacity is reached when the initial hydrogen pressure is 1.2MPa and the temperature is 433K. 90% only takes 30s. The hydrogen absorption and desorption enthalpy changes are -52.69KJ / Mol and 53.21KJ / Mol respectively. The enthalpy of reaction, the platform pressure of hydrogen absorption and desorption, and the kinet...
Embodiment 3
[0038] The difference from Example 1 lies in: preparing and testing hydrogen storage alloy LaNi 3.8 Al 1.0 Fe 0.2 .
[0039] Test result: LaNi 3.8 Al 1.0 Fe 0.2 Under the above activation conditions, the alloy can begin to absorb hydrogen quickly after only about 750s of incubation period, and the activation process can be completed in one time. The hydrogen storage capacity of the hydride formed at 286K is 4.83. The equilibrium pressure of hydrogen absorption at 433K is 83kPa, the equilibrium pressure of hydrogen desorption is 80kPa, the hysteresis coefficient is very small, and the kinetic speed of hydrogen absorption and desorption is very fast. The maximum hydrogen absorption capacity is reached when the initial hydrogen pressure is 1.2MPa and the temperature is 433K. 90% only needs 35s. The hydrogen absorption and desorption enthalpy changes are -50.02KJ / Mol and 50.43KJ / Mol respectively. The enthalpy of reaction, the platform pressure of hydrogen absorption and desorption,...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap