Pharmaceutical composition containing fusion protein capable of inhibiting blood vessel hyperplasia and application thereof
A technology of fusion protein and angiogenesis, applied in the field of medical application, can solve problems such as toxic side effects, poor stability of fusion protein, and decreased protein activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 110
[0044] Stability study of the original prescription of embodiment 110mg / ml FP3 fusion protein in 3ml glass ampoule at 4°C
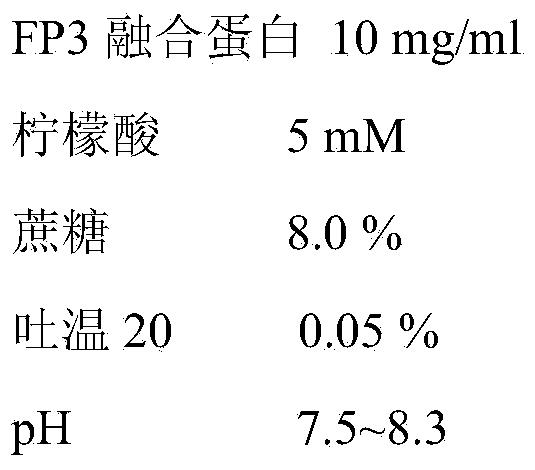
[0045] The prescription is as follows:
[0046]
[0047] Adjust the pH of the system to 6.0-6.5 with hydrochloric acid
[0048] After changing the protein stock solution, aseptically dispense it into 3ml glass ampoules, reserve samples at 4°C, and measure at 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, and December Samples, stability determined by SEC-HPLC. The results showed that the formulation could not effectively inhibit the formation of polymers, resulting in a decrease in product purity and a decrease in affinity with VEGF, which may induce an immune response after entering the body.
[0049] Table 1.10mg / ml FP3 fusion protein stability at 4°C
[0050] time (month)
Embodiment 210
[0051] Stability Study of Example 210mg / ml FP3 Fusion Protein in 3ml Glass Ampoule at 4°C
[0052]
[0053] After changing the protein stock solution, aseptically dispense it into 3ml glass ampoules, reserve samples at 4°C, and measure at 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, and December Samples, stability determined by SEC-HPLC. The results showed that the formulation could not effectively inhibit the formation of polymers, resulting in a decrease in product purity and a decrease in affinity with VEGF, which may induce an immune response after entering the body.
[0054] Table 2.10mg / ml FP3 fusion protein stability at 4°C
[0055] time (month)
[0056] 1
Embodiment 310
[0057] Stability study of embodiment 310mg / ml FP3 fusion protein in 3ml glass ampoule at 4°C
[0058]
[0059] After changing the protein stock solution, aseptically dispense it into 3ml glass ampoules, reserve samples at 4°C, and measure at 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, and December Samples, stability determined by SEC-HPLC. The results showed that the formulation could not effectively inhibit the formation of polymers, resulting in a decrease in product purity and a decrease in affinity with VEGF, which may induce an immune response after entering the body.
[0060] Table 3.10mg / ml FP3 fusion protein stability at 4°C
[0061] time (month)
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More