Pharmaceutical composition of oil of zedoary turmeric for injection
A technology for zedoary turmeric oil and injection, which is applied in the field of medicine, can solve the problems of unqualified inspection items for visible foreign bodies, no better solution, easy precipitation of small white spots, etc., so as to facilitate clinical drug use and promotion, and achieve stable pH value. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
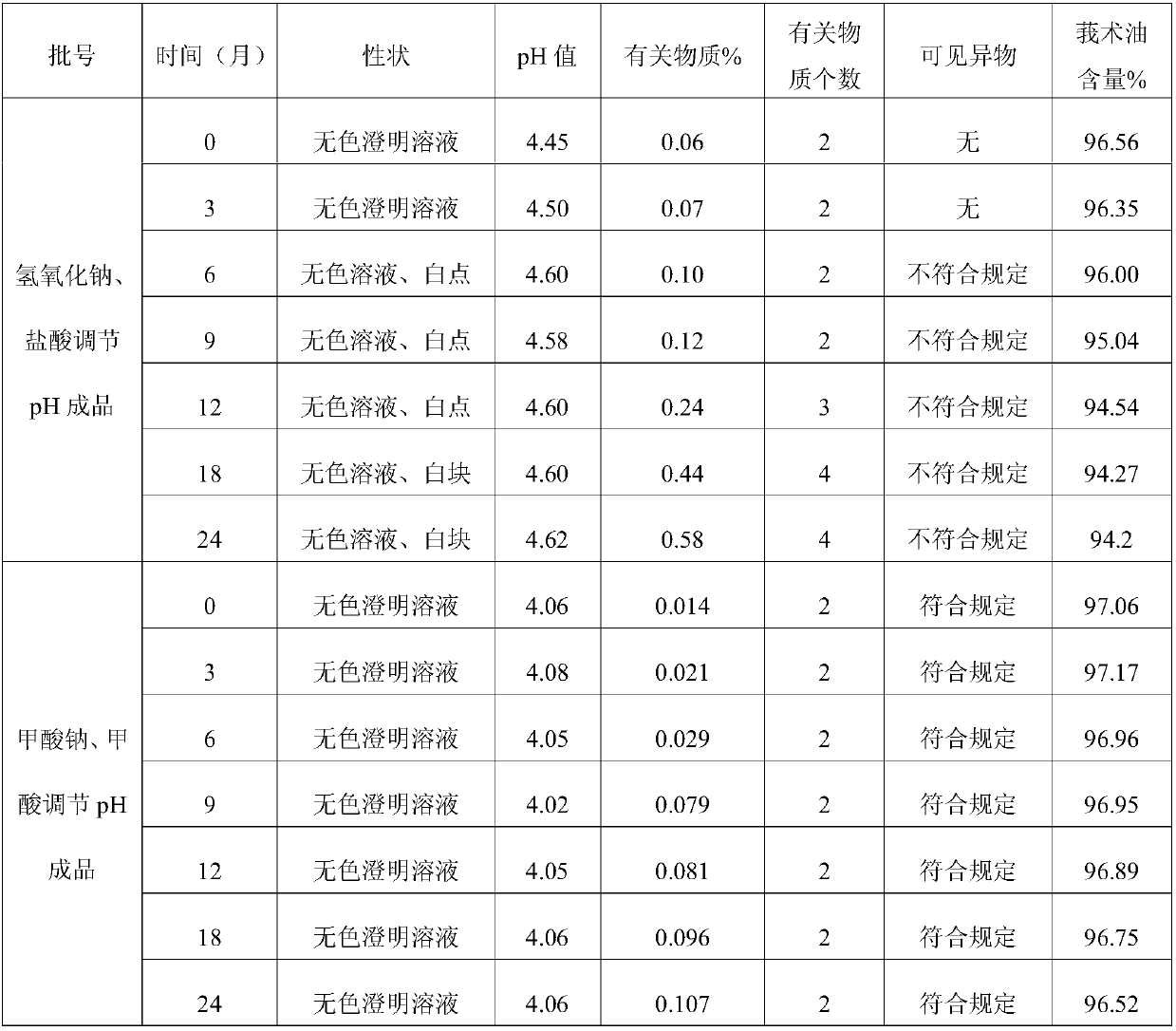
[0026] A method for preparing a pharmaceutical composition for injection that improves the stability of zedoary oil drug injection preparations, comprising the following steps: (1) weighing 0.1 g to 100 g of raw materials calculated as zedoary oil, 9.0 g of sodium pyrosulfite, and 1 mg to 1 mg of formic acid. 2.0g, sodium formate 1mg ~ 2.0g; (2) Formic acid and sodium formate were prepared into 10% ~ 20% solutions respectively, and set aside. (3) Add to 500ml of water for injection below 40°C, stir until completely dissolved, then add 0.02% (g / ml) activated carbon, stir for 15 minutes, filter and decarburize. (4) Use formic acid or sodium formate solution to adjust the pH value of the filtrate to 3.0-7.0, add water for injection below 40°C to 1000ml; (5) filter the medicinal solution until it is clear, fill it, and sterilize it.
[0027] The specific components and contents thereof of the present embodiment are as follows:
[0028]
[0029] Formic acid and sodium formate w...
Embodiment 2
[0031] Alternatively, the above-mentioned pharmaceutical composition for injection that improves the stability of the zedoary oil pharmaceutical injection preparation is prepared according to the following steps:
[0032] (1) Weigh 0.1g-100g of raw materials calculated as zedoary oil, 1mg-2.0g of formic acid, and 1mg-2.0g of sodium formate; (2) prepare 10%-20% solutions of formic acid and sodium formate respectively, and set aside. (3) Add to 500ml of water for injection below 40°C, stir until completely dissolved, then add 0.02% (g / ml) activated carbon, stir for 15 minutes, filter and decarburize. (4) Use formic acid or sodium formate solution to adjust the pH value of the filtrate to 3.0-7.0, add water for injection below 40°C to 1000ml; (5) filter the medicinal solution until it is clear, fill it, and sterilize it.
[0033] The specific components and contents thereof of the present embodiment are as follows:
[0034] Curcuma oil hydrochloride 20g
[0035] Formic acid 1.0...
Embodiment 3
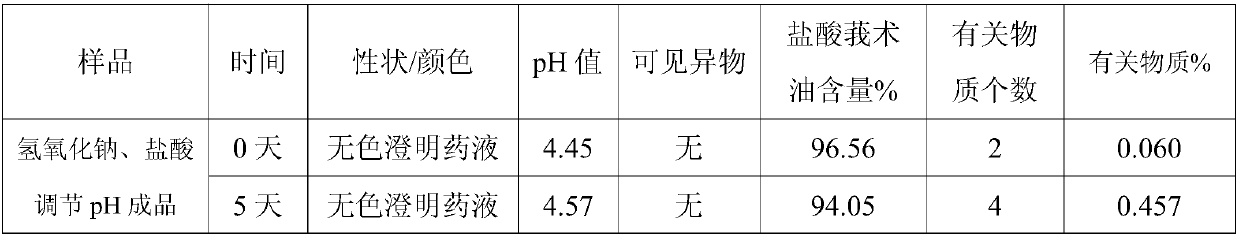
[0039] Comparative Test of Stability of Zedoary Oil Sodium Pyrosulfite Injection
[0040] The detection of visible foreign matter of zedoary oil sodium pyrosulfite injection prepared by the present invention complies with the provisions of the drug quality standard, and the solution stability is very good. Under the condition of avoiding the use of other co-solvents that increase the risk of clinical application, the zedoary oil coke is solved. Sodium sulfite injection is prone to problems such as small white spots, white lumps, and cloudy solution during storage. Utilize the curcuma oil sodium pyrosulfite injection that the present invention makes according to the relevant requirements of the Chinese Pharmacopoeia 2005 edition two appendix XIXC drug preparation stability test guiding principle, respectively investigated and placed 24 months at 25 ℃, 6 months at 40 ℃ , Placed at 60°C for 10 days, and placed at 0-5°C for 20 days for drug stability. As a result, the product qual...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap