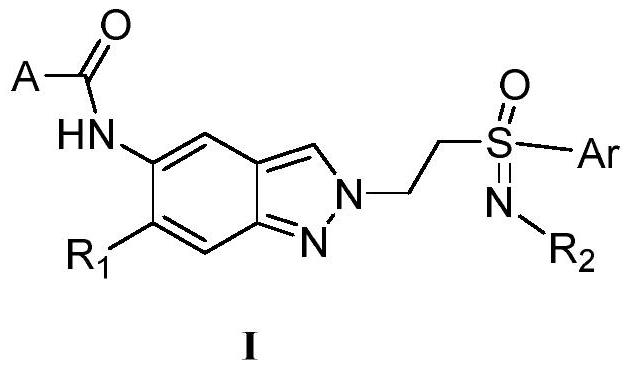
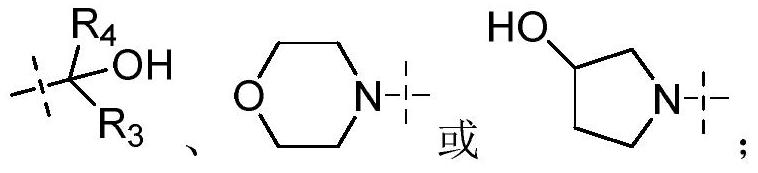
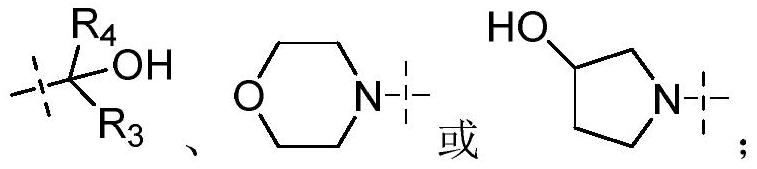
Sulfoximide substituted indazole IRAK4 kinase inhibitor as well as preparation method and application thereof
A technology of sulfinimides and indazoles, applied in the field of biomedicine, can solve the problems of unsatisfactory efficacy, safety, pharmacokinetics, low pharmacokinetics and bioavailability, and animal safety risks To achieve good pharmacokinetic properties, reduce the risk of hERG inhibition, and good IRAK4 inhibitory activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0076] Example 1: Synthesis of I-1
[0077] synthetic route:
[0078]
[0079] Steps:
[0080] step 1:
[0081] Compound IA-1 (1.63 g, 0.01 mol), IB-1 (1.91 g, 0.01 mol) were added to dichloromethane (DCM, 30 mL), followed by N,N-diisopropylethylamine (DIPEA, 1.94 g, 0.015mol), 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide (EDCI) (2.3g, 0.015mol), the reaction system was stirred at 30°C for 12h. The reaction mixture was extracted with water (20 mL), the organic layer was concentrated to dryness under reduced pressure, and recrystallized by adding absolute ethanol (10 mL) to obtain a pale yellow solid IC-1 (2.67 g, yield 79.5%). 1 H NMR (400MHz, DMSO-d 6 ): δ=13.10(br,1H),11.55(br,1H),8.35(d,J=8.0Hz,1H),8.14(m,1H),7.98(d,J=8.0Hz,1H),7.72 (s, 1H), 7.47 (s, 1H), 7.04 (s, 1H), 3.92 (s, 3H). LCMS: MS Calcd.: 336.3, MS Found: 337.2 [M+1].
[0082] Step 2:
[0083] Compound IC-1 (400 mg, 1.2 mmol), ID-1 (327 mg, 1.32 mmol), K 2 CO 3 (332 mg, 2.4 mmol), KI (17 mg, 0.1 mmo...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More