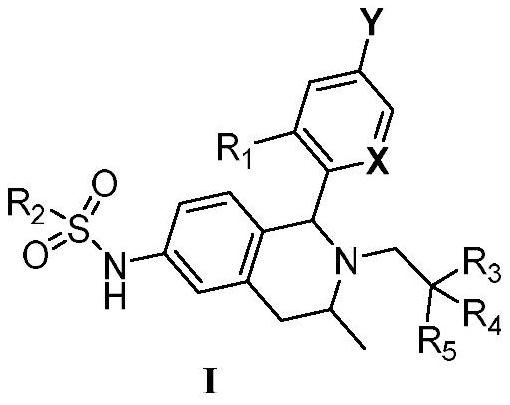
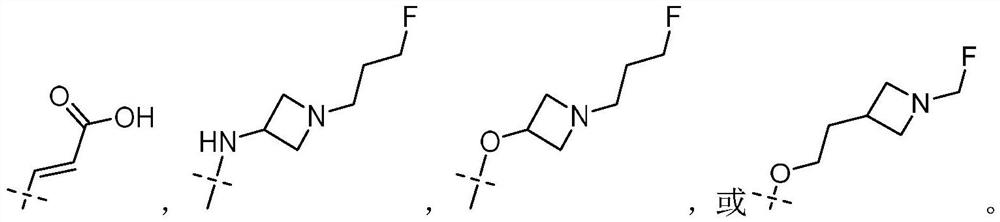
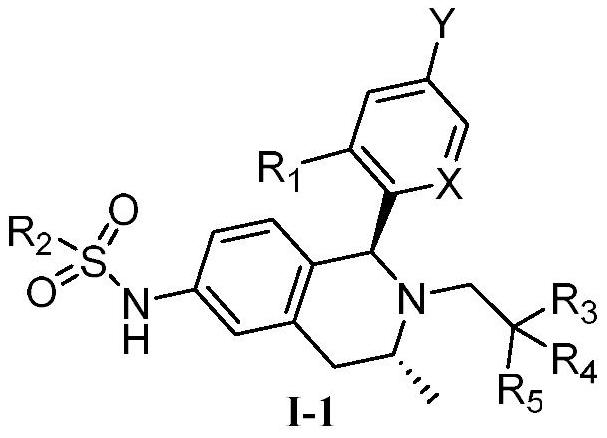
Tetrahydroisoquinoline compound used as selective estrogen receptor down-regulation agent, synthetic method and application
A technology of estrogen receptors and compounds, which is applied in the field of medicine, can solve the problems of poor drug properties and restrictions on the effective degradation of ER
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1~16
[0065] The synthesis of embodiment 1~16:I-1
[0066] synthetic route:
[0067]
[0068] step 1:
[0069] Compound IA-1 (2.28g, 0.01mol), IB-1 (2.24g, 0.01mol) was added to 1,4-dioxane (40mL), and then N,N-diisopropylethylamine (DIPEA , 3.87g, 0.03mol), the reaction system was stirred at 80°C for 12h. The reaction solution was concentrated to dryness under reduced pressure, and the residue was subjected to silica gel column chromatography (eluent: ethyl acetate / petroleum ether 1 / 1) to obtain the target product IC-1 (1.36 g, yield 45%). LCMS: MS Calcd.: 302.4, MS Found: 303.2 [M+1].
[0070] Step 2:
[0071] Compound IC-1 (500mg, 1.65mmol), ID-1 (411mg, 1.82mmol) and acetic acid (500mg, 8.3mmol) were added to toluene (10mL), under the protection of nitrogen, the reaction system was heated to 80°C and stirred for 12h. The reaction solution was concentrated to dryness under reduced pressure, and the residue was subjected to silica gel column chromatography (eluent: ethyl a...
Embodiment 17-40
[0081] synthetic route:
[0082]
[0083] step 1:
[0084] Compound IA-1 (2.28g, 0.01mol), IB-1 (2.24g, 0.01mol) was added to 1,4-dioxane (40mL), and then N,N-diisopropylethylamine (DIPEA , 3.87g, 0.03mol), the reaction system was stirred at 80°C for 12h. The reaction solution was concentrated to dryness under reduced pressure, and the residue was subjected to silica gel column chromatography (eluent: ethyl acetate / petroleum ether 1 / 1) to obtain the target product IC-1 (1.36 g, yield 45%). LCMS: MS Calcd.: 302.4, MS Found: 303.2 [M+1].
[0085] Step 2:
[0086] Compound IC-1 (500mg, 1.65mmol), IF-1 (383mg, 1.82mmol) and acetic acid (500mg, 8.3mmol) were added to toluene (10mL), under the protection of nitrogen, the reaction system was heated to 80°C and stirred for 12h. The reaction solution was concentrated to dryness under reduced pressure, and the residue was subjected to silica gel column chromatography (eluent: ethyl acetate / petroleum ether 1 / 3) to obtain the targe...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More