Thiadiazole amide compounds and their applications
A technology of thiadiazole amides and compounds, which is applied in the field of preparation of drugs for the treatment of prostate cancer, can solve the problems of reducing drug efficacy, multi-drug resistance, limiting clinical treatment options and benefits, etc., and achieve good safety and high Antagonize the activity of androgen receptor transcription, the effect of good biological activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
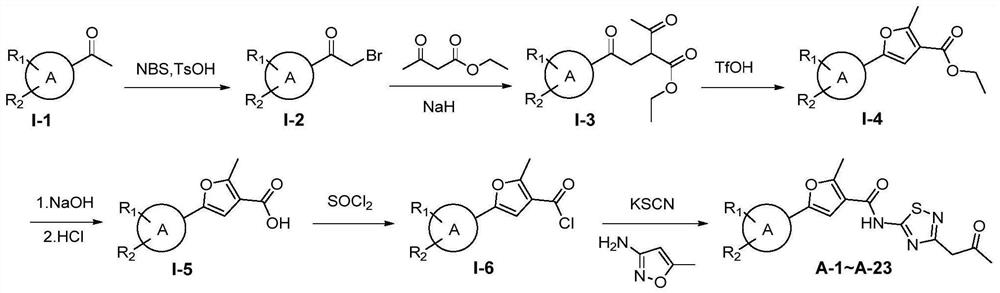
[0121] Example 1: Synthesis of target molecules A-1 to A-23
[0122] The synthetic route of compound A-1~A-23 is as follows figure 1 shown.
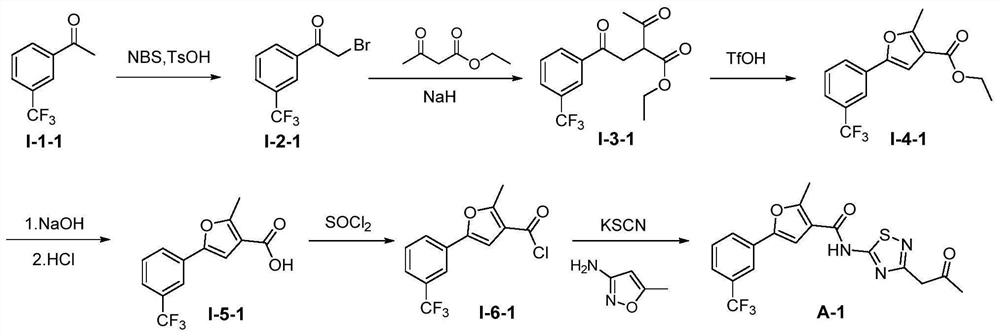
[0123] 2-Methyl-5-(3-(trifluoromethyl)phenyl)-N-(3-(2-oxopropyl)-1,2,4-thiadiazol-5-yl)furan- 3-Carboxamide (Compound A-1)
[0124] The specific synthetic route is as figure 2 shown.
[0125] (a) Synthesis of 2-bromo-1-(3-trifluoromethylphenyl)ethan-1-one (Compound I-2-1)
[0126] NBS (1.04 g, 5.9 mmol) and p-toluenesulfonic acid (458 mg, 2.7 mmol) were dissolved in acetonitrile (20 mL), and m-trifluoromethylacetophenone (1-1-1, 1.00 g, 5.3 mmol) was added , 55 ℃ stirring reaction 1h. An appropriate amount of ethyl acetate was added to the system, washed with water and saturated brine successively, and the organic layer was washed with anhydrous Na 2 SO 4 Dry and concentrated. The residue was obtained by column chromatography to obtain light yellow oily liquid, yield 88%; ESA-MS: m / z=267.0 [M+H] + .
[0127] (b) Synthesis of ...
Embodiment 2
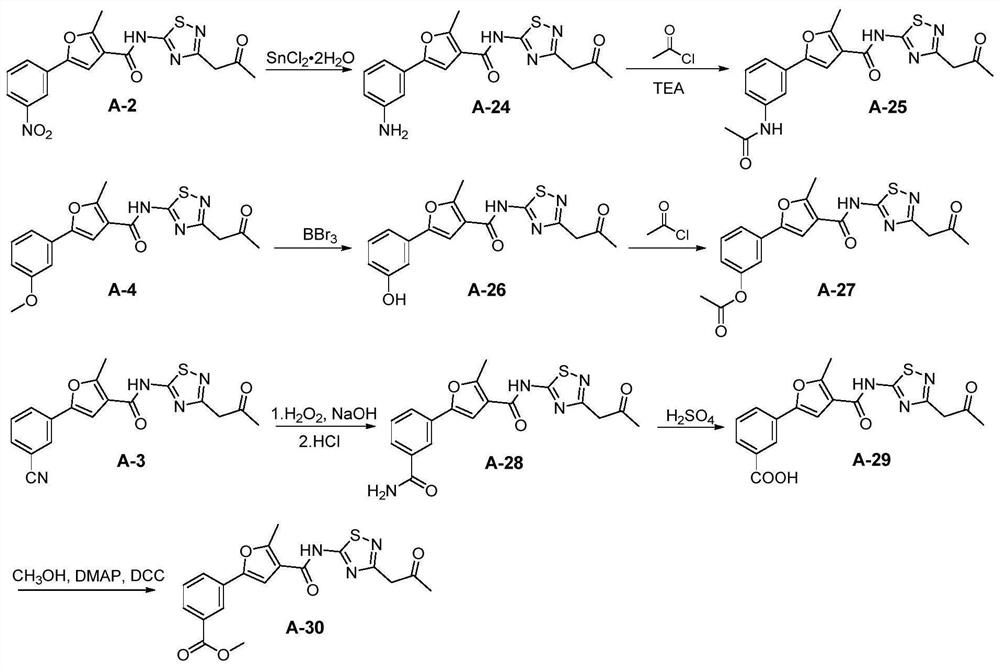
[0181] Example 2: Synthesis of Compounds A-24 to A-30
[0182] The synthetic routes of compounds A-24~A-30 are as follows image 3 shown.
[0183] 2-Methyl-5-(3-aminophenyl)-N-(3-(2-oxopropyl)-1,2,4-thiadiazol-5-yl)furan-3-carboxamide ( Compound A-24)
[0184] Compound A-2 (215 mg, 0.56 mmol) was dissolved in ethanol and water (3:1, 12 mL), iron powder (225 mg, 4.0 mmol) and ammonium chloride (89 mg, 1.7 mmol) were added, and the reaction was carried out at 75° C. for 6 h. Saturated sodium bicarbonate solution was added to the system to adjust the pH to weakly alkaline, then an appropriate amount of ethyl acetate was added, washed with water and saturated brine in turn, and the organic layer was washed with anhydrous Na 2 SO 4 After drying and concentration, the residue was subjected to column chromatography to obtain a pale yellow solid with a yield of 72%; 1 H NMR (500MHz, DMSO-d 6 ): δ13.12(s, 1H), 7.54(s, 1H), 7.10(t, J=8.0Hz, 1H), 6.87(t, J=2.0Hz, 1H), 6.80-6.76(m, ...
Embodiment 3
[0197] Example 3: Synthesis of Compounds A-31 to A-33
[0198] 2-Methyl-5-(pyridin-3-yl)-N-(3-(2-oxopropyl)-1,2,4-thiadiazol-5-yl)furan-3-carboxamide ( Compound A-31)
[0199] Synthetic routes such as Figure 4 shown.
[0200] (a) Synthesis of methyl 2-methyl-5-(pyridin-3-yl)furan-3-carboxylate (Compound II-2-1)
[0201] 3-Bromopyridine (II-1-1, 122 μL, 1.27 mmol), methyl 2-methylfuran-3-carboxylate (317 μL, 2.53 mmol), potassium acetate (248 mg, 2.53 mmol) and palladium acetate (0.1%) equiv.) was added to DMA (5 mL) and stirred overnight at 150°C. An appropriate amount of ethyl acetate was added to the system, washed with water and saturated brine successively, and the organic layer was washed with anhydrous Na 2 SO 4 After drying and concentration, the residue was subjected to column chromatography to obtain a yellow solid with a yield of 88%; ESA-MS: m / z=218.1 [M+H] + .
[0202] (b) Synthesis of 2-methyl-5-(pyridin-3-yl)furan-3-carboxylic acid (Compound II-3-1)
[...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap