Ophthalmic formulations of methotrexate
A technology of methotrexate and enhancer, applied in the directions of non-active ingredients medical preparations, medical preparations containing active ingredients, inorganic non-active ingredients, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
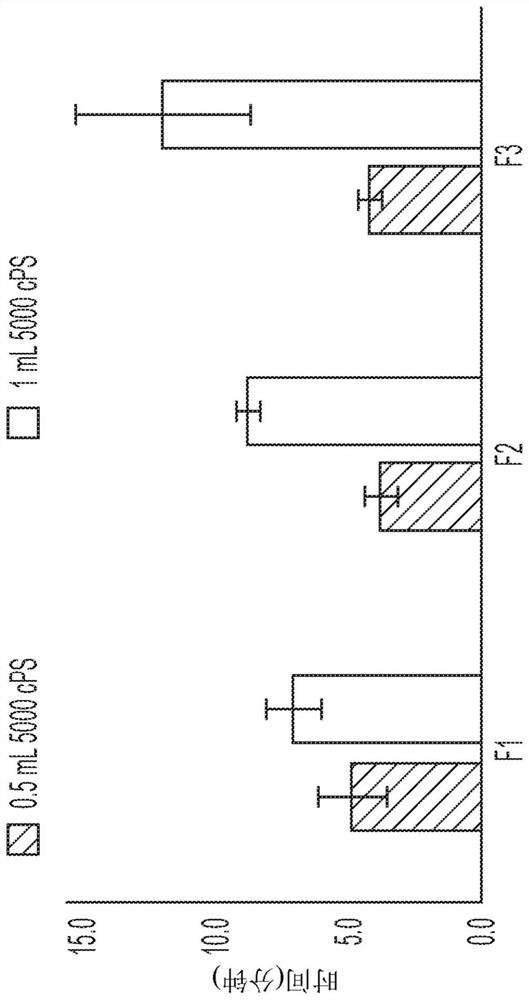
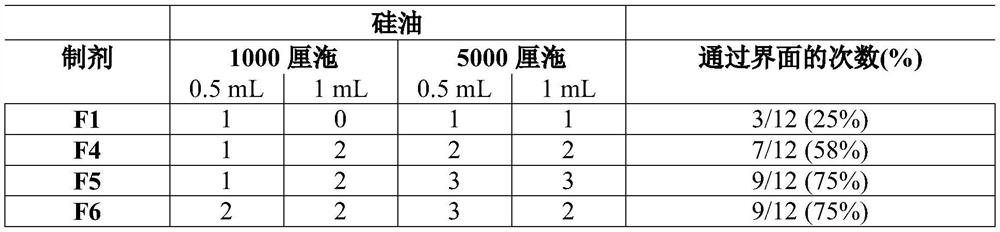
[0192] Example 1: Preparation of Composition and Evaluation by Silicone Oil
[0193] Water is placed in a suitable container, and each ingredient as appropriate for the formulation, eg, surfactants, buffers, bulking agents, methotrexate, etc., is added and dissolved in solution one at a time. The pH was adjusted to 7.4 ± 0.2 by adding 1M sodium hydroxide or HCl. Water was added to make up to the desired final volume.
[0194] Two grades of silicone oil were used: Dimethicone 1,000 cps (Sigma Aldrich, UK) and 5,000 cps (Mistral industrial chemicals, UK) The effect of formulation composition on the travel time of methotrexate through silicone layers of different viscosities and amounts was investigated.
[0195] 1. Test two heights of silicone oil: place 0.5cm or 1cm of oil on top of 2mL of LPBS buffer to confirm the best height to use.
[0196] 2. Place a drop of formulation (-50 mg) on top of the silicone layer.
[0197] Time taken (cm / min) for the API (formulation dropl...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| density | aaaaa | aaaaa |
| density | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More