Protein detecting device
a protein and detection device technology, applied in the direction of liquid/fluent solid measurement, electrochemical variables of materials, instruments, etc., can solve the problem that the fluorescence label cannot be used, and achieve the effect of precise detection/determination
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
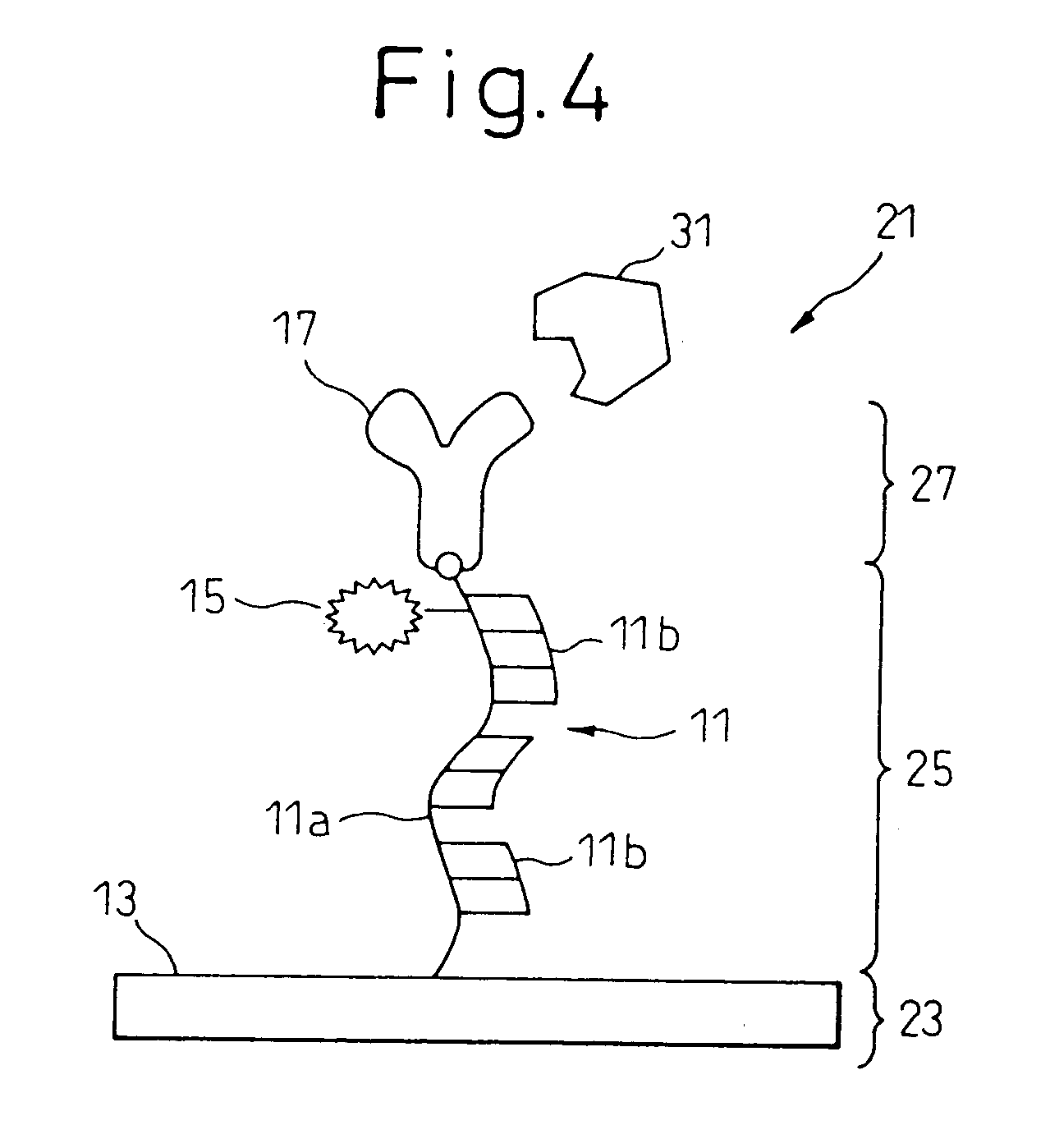
[0082] As schematically shown in FIG. 4, a one-chain polynucleotide 11a, to one end of which a mercapto group was introduced through a spacer, was synthesized, and was hybridized with a complementary one-chain polynucleotide 11b, which was similarly synthesized, to form a polynucleotide double strand structure 11, which was then reacted with a polished gold electrode 13 at room temperature for 24 hours to be bonded to the gold electrode 13.
[0083] Using a solution in which iron porphyrin, as a charge separating group 15 causing charge separation, was dissolved, the charge separating group 15 was absorbed to (intercalated in) the pi-electron stack of the polynucleotide-double-strand structure 11 on the gold electrode 13. Alternatively, a group derived from such a charge separating group may be covalent-bonded to the polynucleotide chain, or part of a nucleotide base may be substituted with a group derived from such a pigment. Subsequently, a Fab fragment 17 of a monoclonal immunoglob...
example 2
[0092] A one-chain polynucleotide, to the 5′ end of which a mercapto group was introduced through a spacer, was synthesized, and was hybridized with a one-chain polynucleotide, which had a complementary sequence and the 5′ end to which a fluorescent pigment (Cy3 (trade mark)) was introduced, to form a polynucleotide double strand structure, which was then reacted with a polished gold electrode positioned on a support at room temperature for 24 hours to be bonded to the gold electrode. It is preferable that the degree of polymerization of the polynucleotide chain is such that it has 12 to 100 monomer-residues. The mercapto group and the fluorescent pigment may be introduced to the ends of one of the one-chain polynucleotides, respectively, or may be introduced to the 3′ ends of both the one-chain polynucleotides.
[0093] The polynucleotide double strand structures were fixed in a circular area of 10 micrometers, around which a spacer area having no polynucleotide double strands fixed ...
example 3
[0101] A detecting unit was fabricated as in Example 1, except that Cy3 or FITC was used as an light emitting group in place of the charge separating group used in Example 1.
[0102] A one-chain polynucleotide, to one end of which a mercapto group was introduced through a spacer, was synthesized and was hybridized with a complementary one-chain polynucleotide 11b, which was similarly synthesized, to form a polynucleotide double strand structure, which was then reacted with a polished gold electrode at room temperature for 24 hours to be bonded to the gold electrode.
[0103] Using a solution in which a light emitting group was dissolved, the light emitting group was intercalated in the pi-electron stack of the polynucleotide double strand on the gold electrode. Alternatively, a nucleotide base may be replaced by a derivative of the light emitting group. Subsequently, a Fab fragment of a monoclonal immunoglobulin IgG was fixed to the end of the polynucleotide chain.
[0104] A detecting u...
PUM
| Property | Measurement | Unit |
|---|---|---|
| thick | aaaaa | aaaaa |
| width | aaaaa | aaaaa |
| width | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More