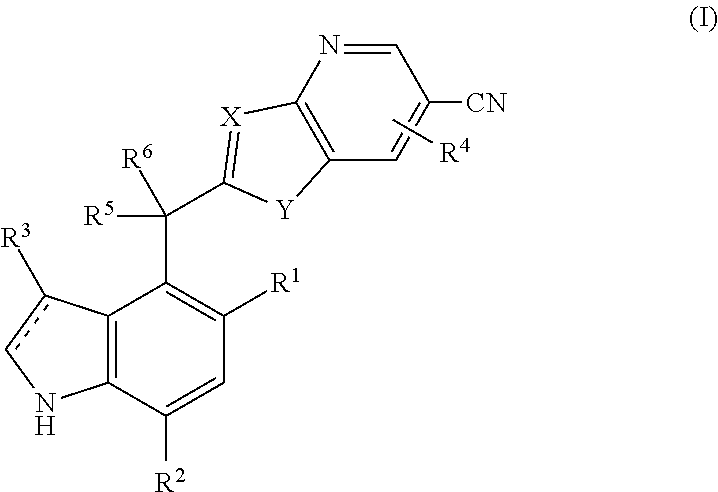
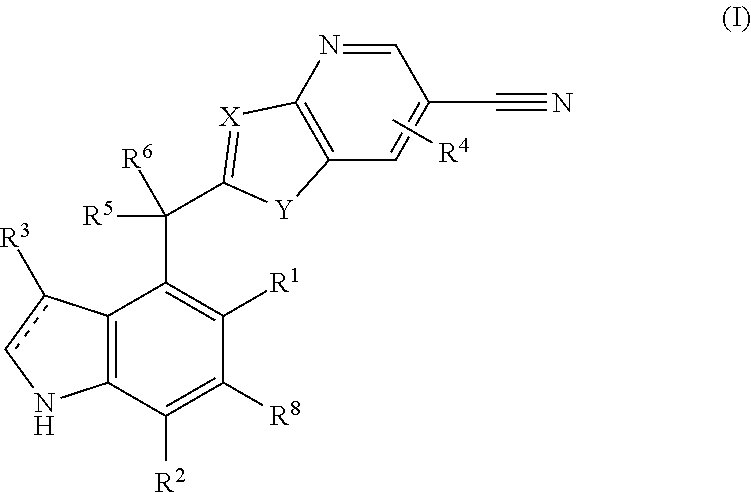
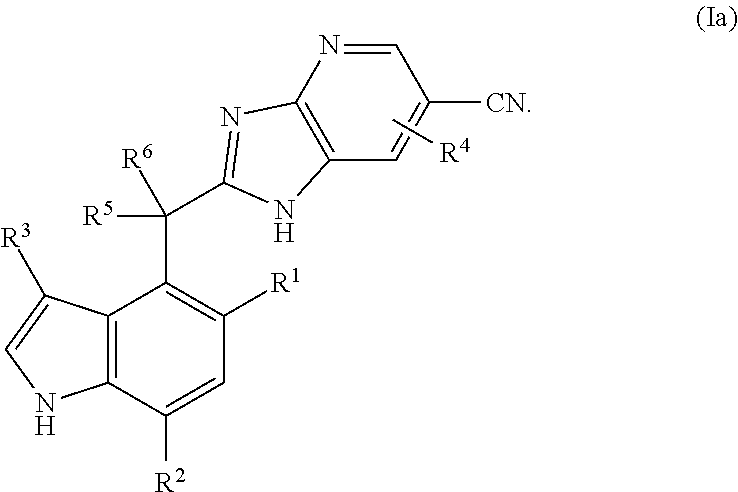
Complement factor B inhibitors and uses there of
a technology of complement factor and inhibitor, which is applied in the field of inhibitors of complement factor b, can solve the problems of deterioration of sight, retinal displacement, hemorrhage and scarring, and increase the risk of age-related macular degeneration
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
biological example 1
Human Complement Factor B ELISA Assay
[0368]CVF-Bb complex prepared from purified cobra venom factor (1 μM), recombinant human complement factor B (expressed in drosophila cells and purified using standard methods) and human complement factor D (expressed in E. Coli, refolded and purified using standard methods). CVF-Bb complex at 3 nM concentration was incubated with test compound at various concentrations for 1 hour at room temperature in PBS pH 7.4 containing 10 mM MgCl2 and 0.05% (w / v) CHAPS. Human complement C3 substrate purified from plasma was added to a final concentration of 1 μM. After 1 hour incubation at room temperature, the enzyme reaction was stopped by addition of a cocktail of concentrated pan-protease inhibitors. The product of the reaction, C3a, was quantified by means of an enzyme-linked-immunosorbent assay. IC50 values were calculated from percentage of inhibition of CVF-Bb activity as a function of test compound concentration.
biological example 2
Human Complement Factor B TR-FRET Assay
biological example 2.1
(+) or (−)-tert-Butyl 3-(3-hydroxyphenyl)piperazine-1-carboxylate
[0369]
[0370]Resolution of the enantiomers of (±)-tert-butyl 3-(3-hydroxyphenyl)piperazine-1-carboxylate (CAS#889956-76-7) was achieved by chiral HPLC using a CHIRALPAK AD column with heptane / EtOAc / MeOH 90 / 5 / 5+0.1 diethylamine to give (+) or (−)-tert-butyl 3-(3-hydroxyphenyl)piperazine-1-carboxylate (tr=9.7 min) and (−) or (−)-tert-butyl 3-(3-hydroxyphenyl)piperazine-1-carboxylate (tr=15.7 min).
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| temperatures | aaaaa | aaaaa |
| enantiomeric excess | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More