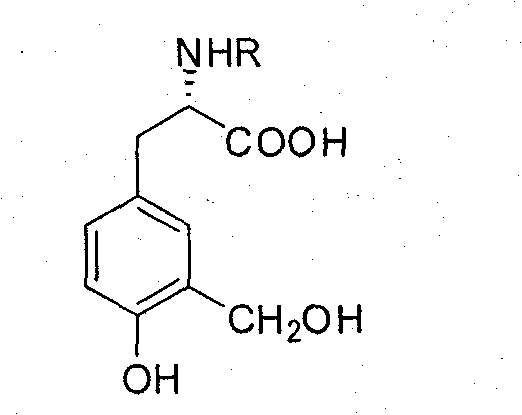
Synthesis of L-3-hydroxyl-4-methoxyl-5-methyl-phenylalaninol/phenylalanine
A compound, C1-C14 technology, applied in the field of synthesis of L-3-hydroxy-4-methoxy-5-methyl-phenylalaninol/acid compounds, can solve the problem of affecting the yield and yield of the synthesis process It is not ideal and needs to be improved to achieve the effect of easy industrialization, cheap reagents and low toxicity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
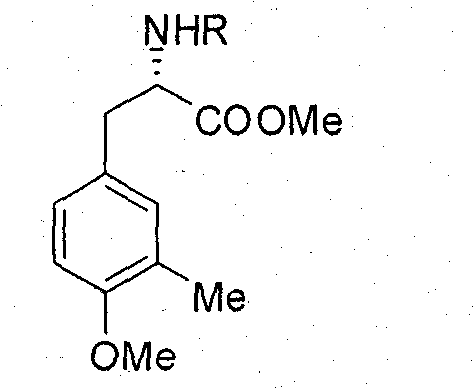
[0033] Example 1: Preparation of compound five (R group is Cbz), namely L-N-Cbz-3-methyl-4-methoxy-phenylalanine methyl ester:
[0034] Add 1.62mmol of LN-Cbz-3-hydroxymethyl-tyrosine (Compound 4 with Cbz as the R group) in a 50mL round bottom flask, under the protection of argon, add 6.5mL of acetone, add 4.85mmol after the product is dissolved Potassium carbonate, 4.85mmol dimethyl sulfate, heated to reflux under magnetic stirring, reflux temperature 60℃, react for 7h under reflux conditions, TLC detects the end of the reaction, after the reaction, acetone is evaporated, appropriate amount of water is added, and ethyl acetate is extracted Three times, anhydrous Na 2 SO 4 After drying and evaporating the solvent, a light yellow liquid crude product was obtained. Under argon protection, add 14mLCH to the crude product 2 Cl 2 , Then add 5.55mmol Et 3 SiH and 22.19mmol CF 3 COOH, stirring at room temperature for 10h, after the reaction, use dilute Na 2 CO 3 Neutralize the solution,...
Embodiment 2
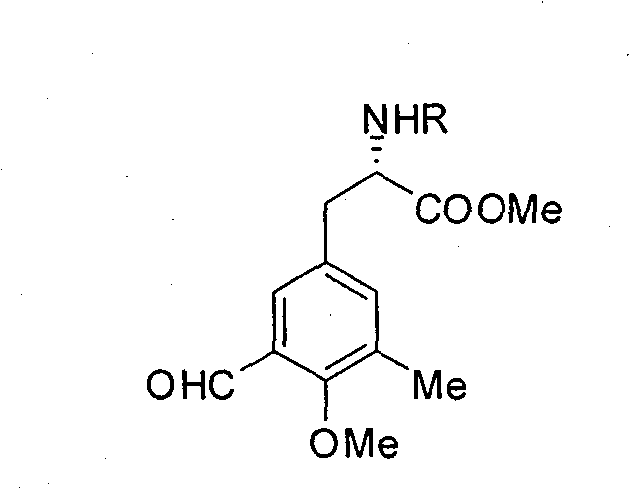
[0035] Example 2: Preparation of compound six (R group is Cbz), that is, L-N-Cbz-3-formyl-4-methoxy-5-methyl-phenylalanine methyl ester:
[0036] Add 1.362mmol of compound five (R group is Cbz) in a 25mL round-bottomed flask, under the protection of argon, add 5mL CH 2 Cl 2 , Then lower the temperature to -10℃, add 3.268mmolTiCl 4 And 1.634mmol Cl 2 CHOCH 3 , React for 3h, after the reaction, pour the reaction solution into a beaker filled with ice water, stir for 1h, extract three times with ethyl acetate, anhydrous Na 2 SO 4 After drying and evaporating the solvent, a yellow-green liquid crude product is obtained. After the crude product is separated by a chromatographic column, compound six (R group is Cbz) is obtained with a yield of 89%; [α] D 26 +63(c 1.1, CHCl 3 ); IR(neat)v max : 3314, 2954, 2866, 1748, 1692, 1541, 1259, 1214, 1058, 1006, 749, 698cm -1 ; 1 H NMR(400MHz, CDCl 3 ): δ (ppm) 10.32 (1H, s), 7.42 (1H, s), 7.25-7.37 (5H, m), 7.19 (1H, s), 5.31 (1H, d, J=8.0), 5.12...
Embodiment 3
[0037] Example 3: Preparation of compound seven (R group is Cbz), namely L-N-Cbz-3-hydroxy-4-methoxy-5-methyl-phenylalaninol:
[0038] In a 25mL round-bottom flask, add 1.1mmol of compound six (R group is Cbz) and 11mL of dichloromethane. After the reactants are dissolved, add 1.65mmol of m-chloroperoxybenzoic acid. After stirring for 13h at room temperature, use dilute Na 2 CO 3 Neutralize the solution, extract three times with dichloromethane, combine the organic phases, wash with saturated NaCl solution, anhydrous Na 2 SO 4 After drying and evaporating the solvent, a yellow liquid crude product was obtained. Without treatment, THF was added to the crude product at a concentration of 0.1 mol / L, and 2 eq of LiBH was added. 4 After reaction for 2h, the solvent was distilled off under reduced pressure, extracted three times with ethyl acetate, anhydrous Na 2 SO 4 After drying and evaporating the solvent, a yellow-green liquid crude product is obtained, which is separated by a chromat...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap