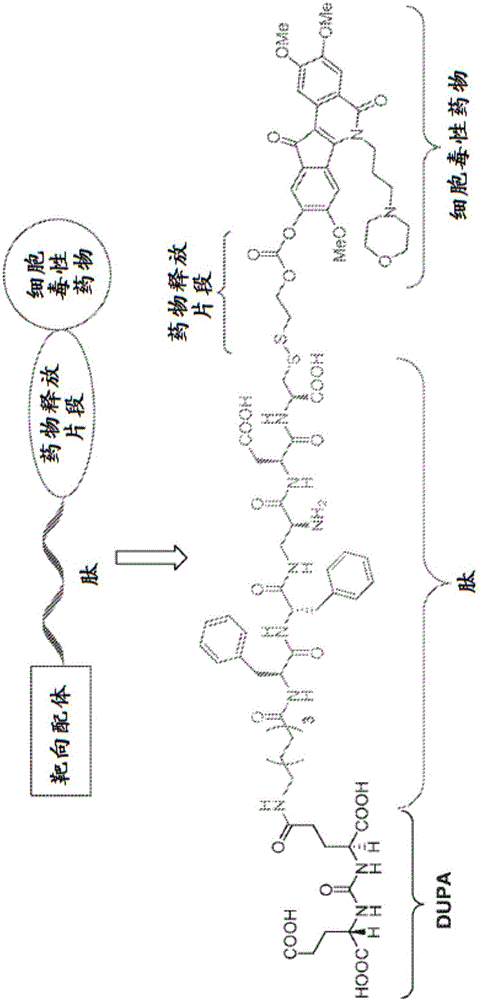
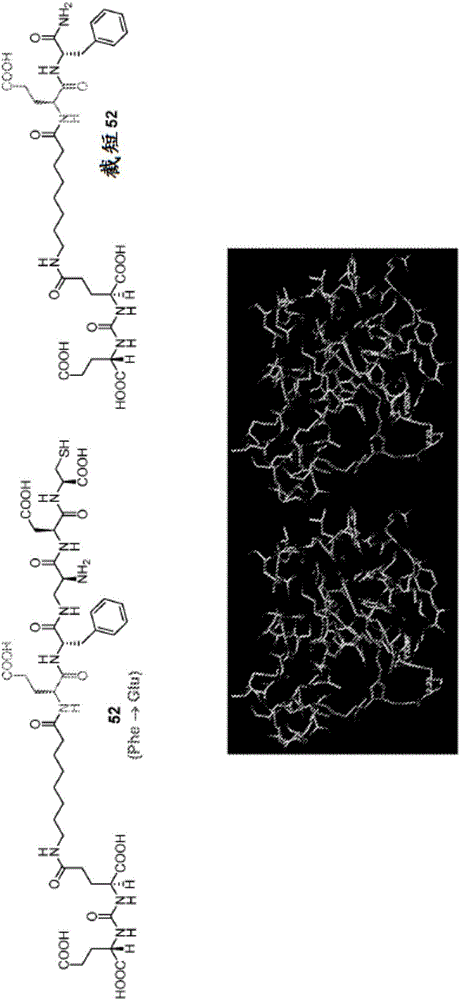
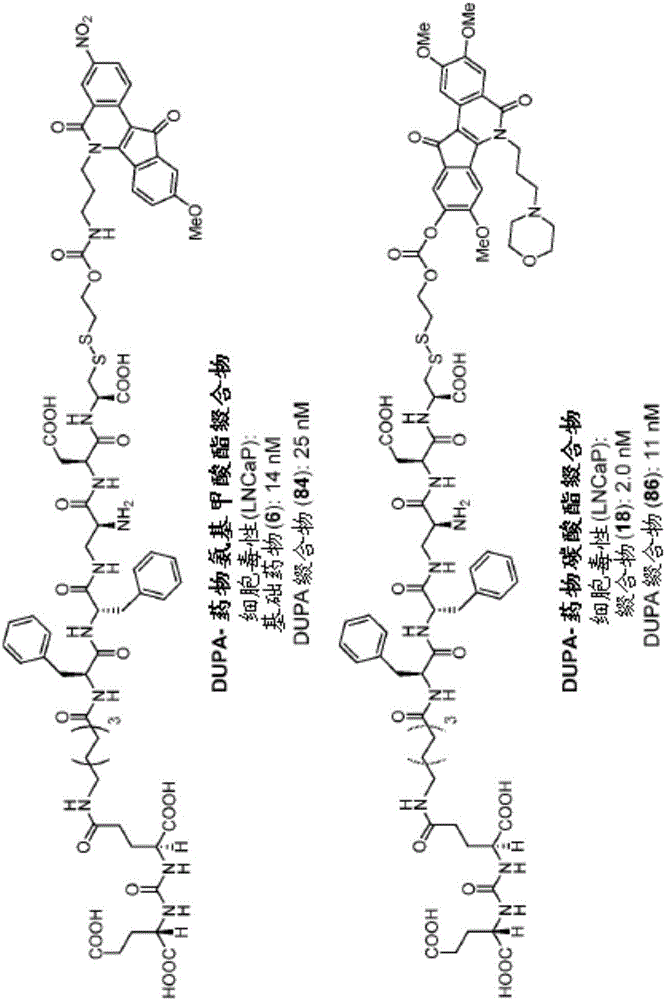
Dupa-indenoisoquinoline conjugates
A technology of indenoisoquinoline and conjugates, which is applied in the field of targeting ligand-cytotoxic drug conjugates, and can solve the problems of use discount, disappointing results, tumor tolerance and the like
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0308] Example 1: N-[4-(benzyloxy)benzylidene]-3-bromo-1-propanamine (54)
[0309] 3-Bromopropylamine hydrobromide (3.56 g, 16.2 mmol) was diluted in CHCl 3 (50mL) and Et3 in N (1.64g, 16.2mmol). The mixture was stirred for 5 minutes, then compound 53 (3.00 g, 14.1 mmol) and Na 2 SO 4 (4.02g, 28.3mmol). The mixture was stirred at room temperature for 16 h, then washed with H 2 O (100 mL x 3) and brine (100 mL) washes. The organic layer was washed with anhydrous Na 2 SO 4 Dried, filtered and concentrated to give product 54 (4.69 g, 100% + residual solvent) as a light yellow syrup. IR (film) 2839, 1645, 1605, 1509, 1246, 1166, 830cm -1 ; 1 H NMR (300MHz, CDCl 3 )δ8.26(s, 1H), 7.68(dd, J=1.8 and 6.9Hz, 2H), 7.45-7.33(m, 5H), 7.02(dd, J=1.9 and 6.9Hz, 2H), 5.11(s , 2H), 3.74(dt, J=0.9 and 6.2Hz, 2H), 3.51(t, J=6.6Hz, 2H), 2.27(m, 2H); ESIMS m / z (relative intensity) 332 / 334(MH + , 100 / 97).
Embodiment 2
[0310] Example 2: cis-4-carboxy-3,4-dihydro-N-(3-bromopropyl)-3-[4-(benzyloxy)phenyl]-7-nitro-1(2H )-isoquinolones (55)
[0311] Schiff base 54 (4.69 g, 14.1 mmol) was diluted in CHCl at 0 °C 3 (50 mL) and added anhydride 58 (2.80 g, 13.5 mmol). The red mixture was stirred at 0 °C for 2 h, then allowed to warm to room temperature and stirring continued for 2 h. Filter the milk-orange mixture and wash with CHCl 3 The residue was washed to give the product 55 (5.18 g, 71%) as an off-white solid: mp 140-141 °C. IR (film) 3076, 1727, 1630, 1525, 1347, 1187, 738cm -1 ; 1 H NMR (300MHz, DMSO-d 6 )δ8.71 (d, J=2.6Hz, 1H), 8.39 (dd, J=2.6 and 6.0Hz, 1H), 7.92 (d, J=8.2Hz, 1H), 7.40-7.30 (m, 5H), 6.92-6.83(m, 4H), 5.19(d, J=6.4Hz, 1H), 5.03(d, J=6.3Hz, 1H), 4.98(s, 2H), 3.90(m, 1H), 3.59(m , 2H), 3.03(m, 1H), 2.16(m, 1H), 2.04(m, 1H); ESIMS m / z (relative intensity) 415 ([MH–COOH–Br] + , 100); MH + HRESIMS calculated value: 539.0818, measured value: 539.0812.
[0312] Scheme 8...
Embodiment 3
[0314] Example 3: 6-(3-bromopropyl)-9-hydroxy-3-nitro-5H-indeno[1,2-c]isoquinoline-5,11(6H)-dione (1)
[0315] Dilute cis acid 56 (0.50 g, 0.93 mmol) in SOCl 2 (50mL) and stirred at room temperature for 16h. The resulting yellow solution was evaporated to dryness. The yellow syrup was diluted in 1,2-dichloroethane (50 mL) at 0 °C and stirred for 15 minutes, followed by the addition of AlCl 3 (0.25 g, 1.85 mmol). The black mixture was heated at reflux for 2 h then evaporated to dryness. The remaining residue was washed with CHCl 3 (100mL) diluted with HCl 6N (100mL), H 2 O (100 mL x 3) and brine (100 mL) washes. The organic layer was washed with anhydrous Na 2 SO 4 dried, filtered and concentrated, adsorbed onto SiO 2 above, and with CHCl 3 Eluted flash column chromatography (SiO 2 ) to give product 1 (57 mg, 15%) as a red solid: mp 281-283 (decomposition) °C. IR (film) 3273, 1659, 1613, 1531, 1345, 1270, 755cm -1 ; 1 H NMR (300MHz, DMSO-d 6 )δ10.82(s, 1H), 8.83(...
PUM
| Property | Measurement | Unit |
|---|---|---|
| strength | aaaaa | aaaaa |
| strength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com