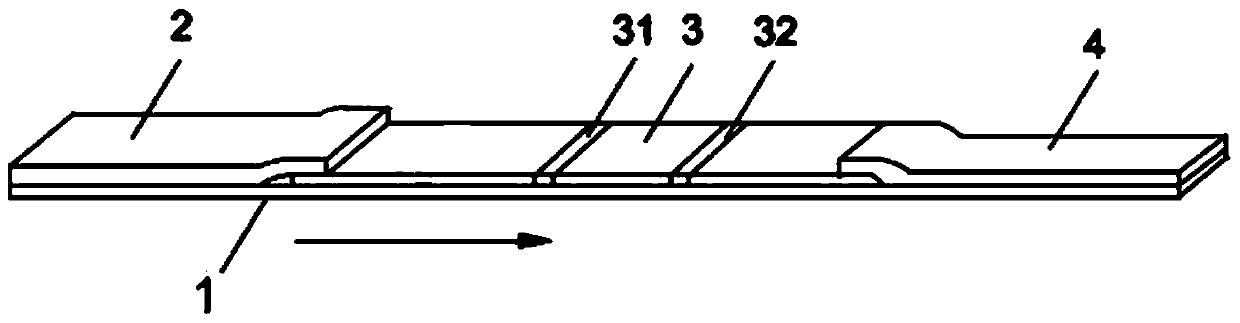
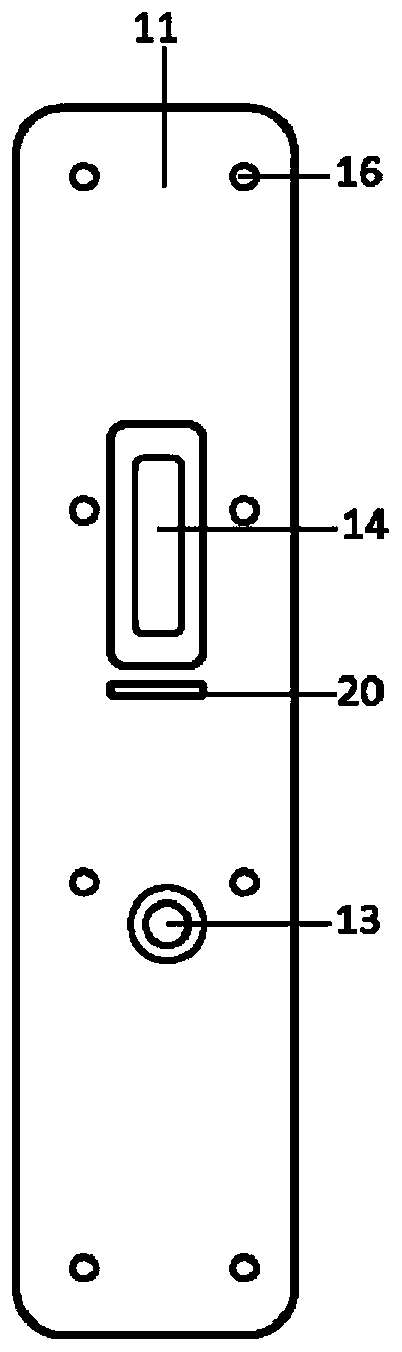
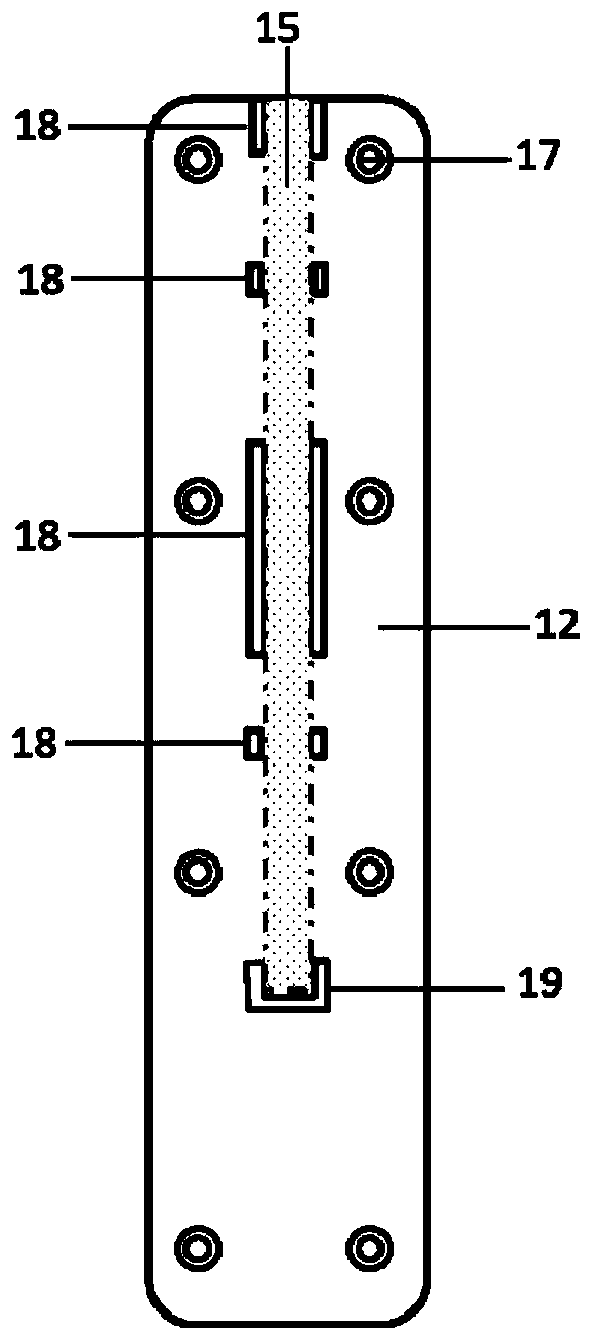
Fluorescent microsphere labeled antibody as well as preparation method and application thereof
A technology for labeling antibodies and fluorescent microspheres, which is applied in the field of immunoassays, can solve the problems of different cost-effectiveness of antibodies or antigens, fixed production or inventory, long transportation cycle, etc., and achieve good sensitivity and specificity, sensitivity and specificity Retention, Sensitivity, and Specificity Consistent Effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0032] 1.1 Preparation of fluorescent microsphere-labeled antibody
[0033] 1.1.1 Microsphere cleaning:
[0034]The original concentration of the fluorescent microspheres was 2.0wt%, and they were centrifuged and washed with 10mM phosphate buffer (pH6.0) at a rotation speed of 14000rpm for 10min. The supernatant was discarded and repeated twice to obtain microsphere precipitates.
[0035] 1.1.2 Activation:
[0036] 1) Preparation of chemical crosslinking agent solution:
[0037] The chemical cross-linking agent is N-hydroxysuccinimide and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide with a weight ratio of 1:1, respectively using 10mM (pH6.0) Phosphate buffer solution prepared into a 500mg / ml solution for subsequent use;
[0038] 2) Activation of fluorescent microspheres:
[0039] Add 10mM phosphate buffer solution (pH6.0) to the washed microsphere precipitate, and ultrasonically mix (40kHz, 6min) to obtain a 100mg / mL fluorescent microsphere solution. The 500mg / ml N Mixe...
Embodiment 2
[0072] 2.1 Preparation of fluorescent microsphere-labeled antibody
[0073] 2.1.1 Microsphere cleaning:
[0074] The original concentration of the fluorescent microspheres was 2.0 wt%, and they were centrifuged and washed with 10 mM phosphate buffer (pH6.0) at a rotation speed of 14000 rpm for 10 min. The supernatant was discarded and repeated twice to obtain microsphere precipitates.
[0075] 2.1.2 Activation:
[0076] 1) Preparation of chemical crosslinking agent solution:
[0077] The chemical cross-linking agent is N-hydroxysuccinimide and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide with a weight ratio of 1:1, respectively using 10mM (pH6.0) Phosphate buffer solution prepared into a 500mg / ml solution for subsequent use;
[0078] 2) Activation of fluorescent microspheres:
[0079] Add 10mM phosphate buffer solution (pH6.0) to the washed microsphere precipitate, and ultrasonically mix (40kHz, 6min) to obtain a 10mg / mL fluorescent microsphere solution. The 500mg / ml N Mi...
Embodiment 3
[0116] 3.1 Preparation of fluorescent microsphere-labeled antibody
[0117] 3.1.1 Microsphere cleaning:
[0118] The original concentration of the fluorescent microspheres was 2.0 wt%, and they were centrifuged and washed with 10 mM phosphate buffer (pH6.0) at a rotation speed of 14000 rpm for 9 minutes. The supernatant was discarded and repeated twice to obtain microsphere precipitates.
[0119] 3.1.2 Activation:
[0120] 1) Preparation of chemical crosslinking agent solution:
[0121]The chemical cross-linking agent is N-hydroxysuccinimide and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide with a weight ratio of 1:1, respectively using 10mM (pH6.0) Phosphate buffer solution prepared into a 500mg / ml solution for subsequent use;
[0122] 2) Activation of fluorescent microspheres:
[0123] Add 10mM phosphate buffer solution (pH6.0) to the washed microsphere precipitate, and ultrasonically mix (40kHz, 6min) to obtain a 100mg / mL fluorescent microsphere solution. The 500mg / ml ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Granularity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap