Methods and compositions for improving cognitive function
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
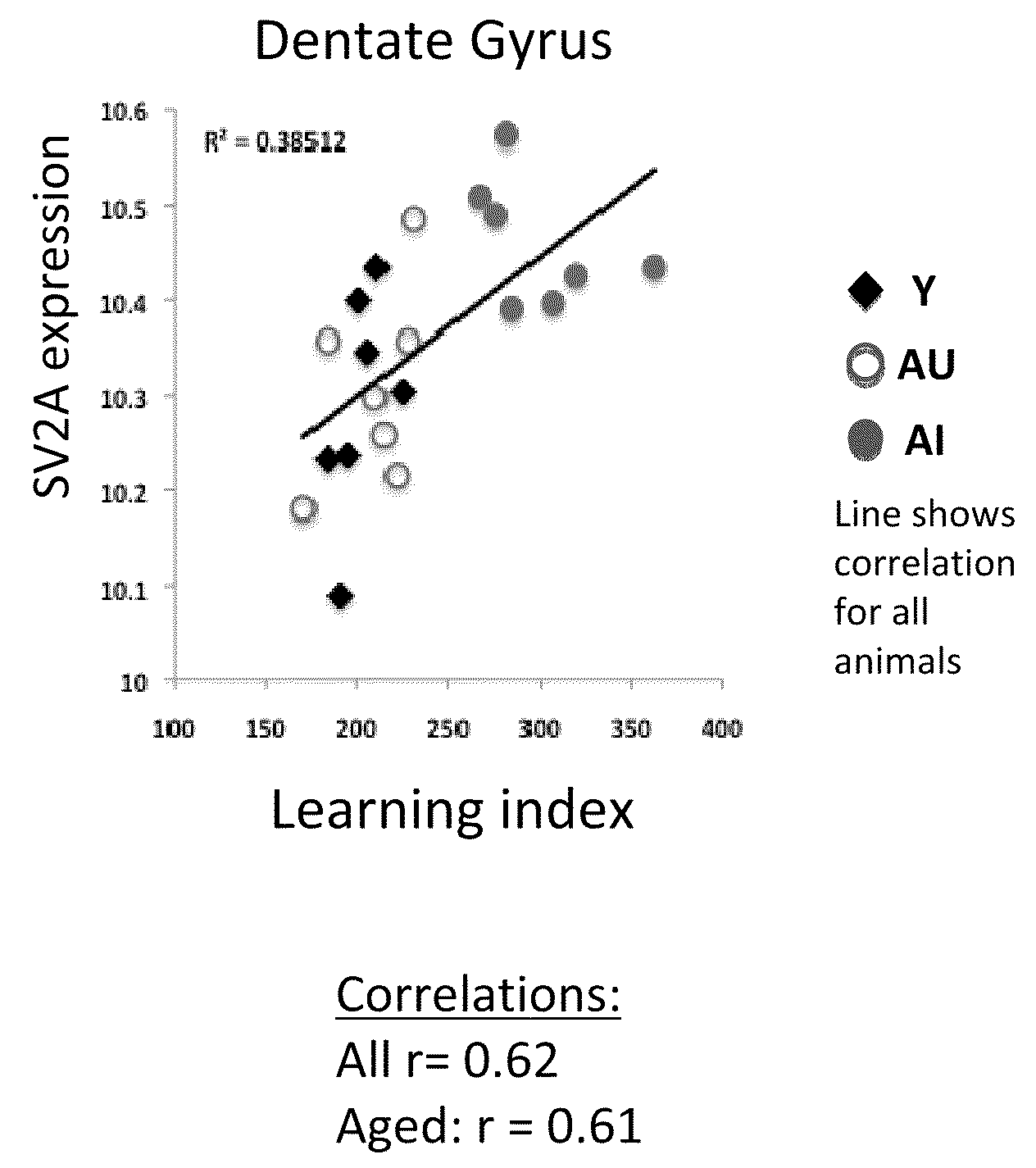
Increased Gene Expression of SV2A in Aged-Impaired Rats
Behavioral Characterization of Young, Aged-Impaired and Aged-Unimpaired Rats in Morris Water Maze (MWM)
[0899]Behavioral tests were performed on young (4 months old) and aged (24 months old) pathogen-free male Long-Evans rats.
[0900]The MWM apparatus consists of a large, circular pool (diameter 1.83 m; height, 0.58 m) filled with water (27° C.) that is made opaque through the addition of non-toxic pigment or some other substance. In the typical “hidden platform” version of the test, rats are trained to find a camouflaged white escape platform (height, 34.5 cm) that is positioned in the center of one quadrant of the maze about 1.0 cm below the water surface. This platform can be retracted to the bottom of the tank or raised to its normal position from outside the maze during behavioral testing. The location of the platform remains constant from trial to trial. Because there are no local cues that mark the position of the platform, ...
example 2
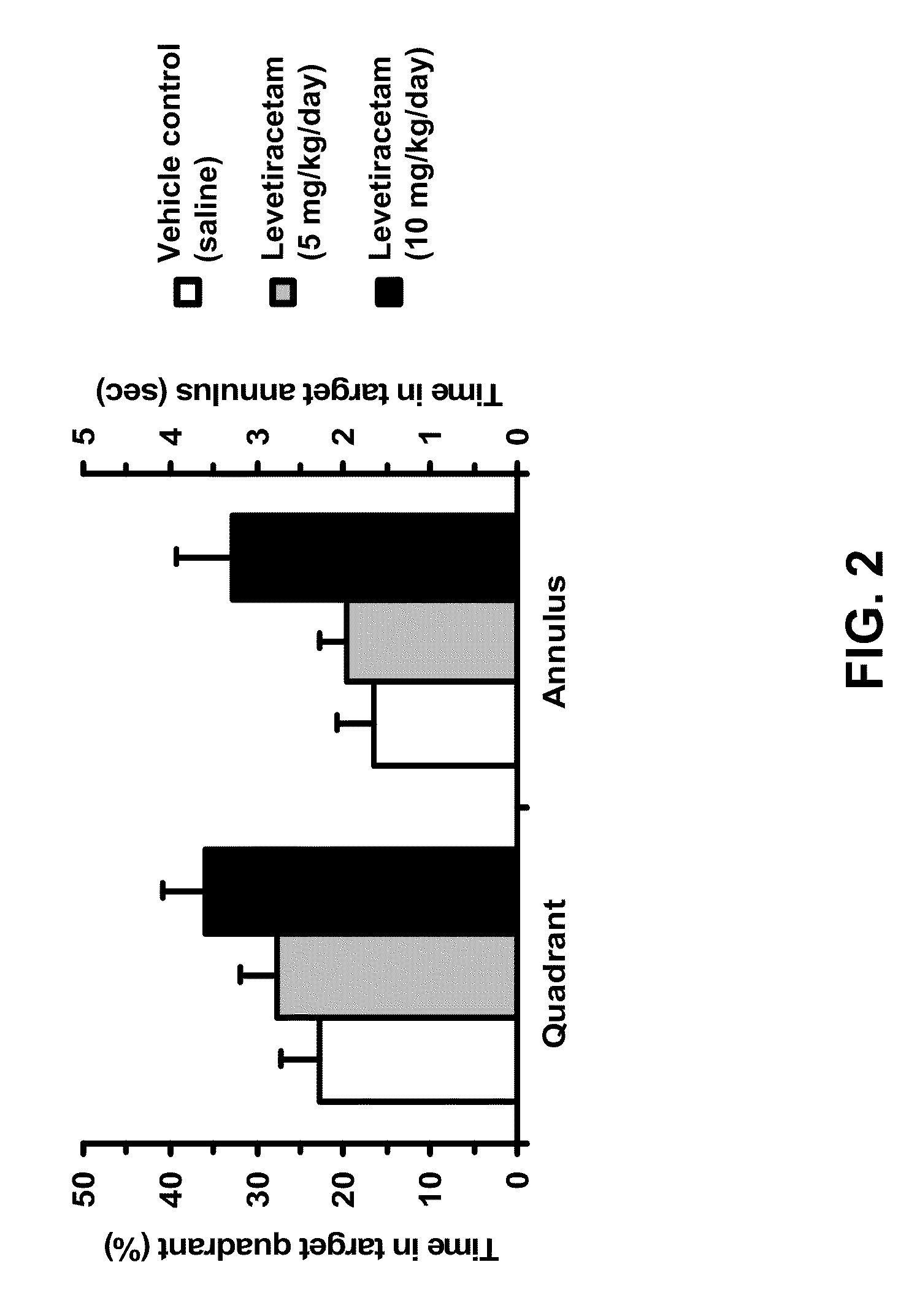
Effect of Levetiracetam in Aged-Impaired Rats
Morris Water Maze Results
[0917]Six Age-Impaired (AI) Long-Evans rats (as characterized above) were tested for their memory of new spatial information in the MWM, under different drug / control treatment conditions (vehicle control and two different dosage levels of levetiracetam). The MWM protocol was substantially the same as the one described in Example 1. Specifically for this study, a retention trial was performed after the training trials, as described below.
[0918]AI rats were given six training trials per training day with a 60-sec inter-trial interval between each training trial for two consecutive days. On each training trial, the rat was released in the maze from one of four equally spaced starting positions around the perimeter of the pool. If the rat did not locate the escape platform within 90 sec on any trial, the experimenter guided the rat to the platform, where it remained for 30 sec. 30 minutes to 1 hour prior to all the tr...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Time | aaaaa | aaaaa |
| Time | aaaaa | aaaaa |
| Mass | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More