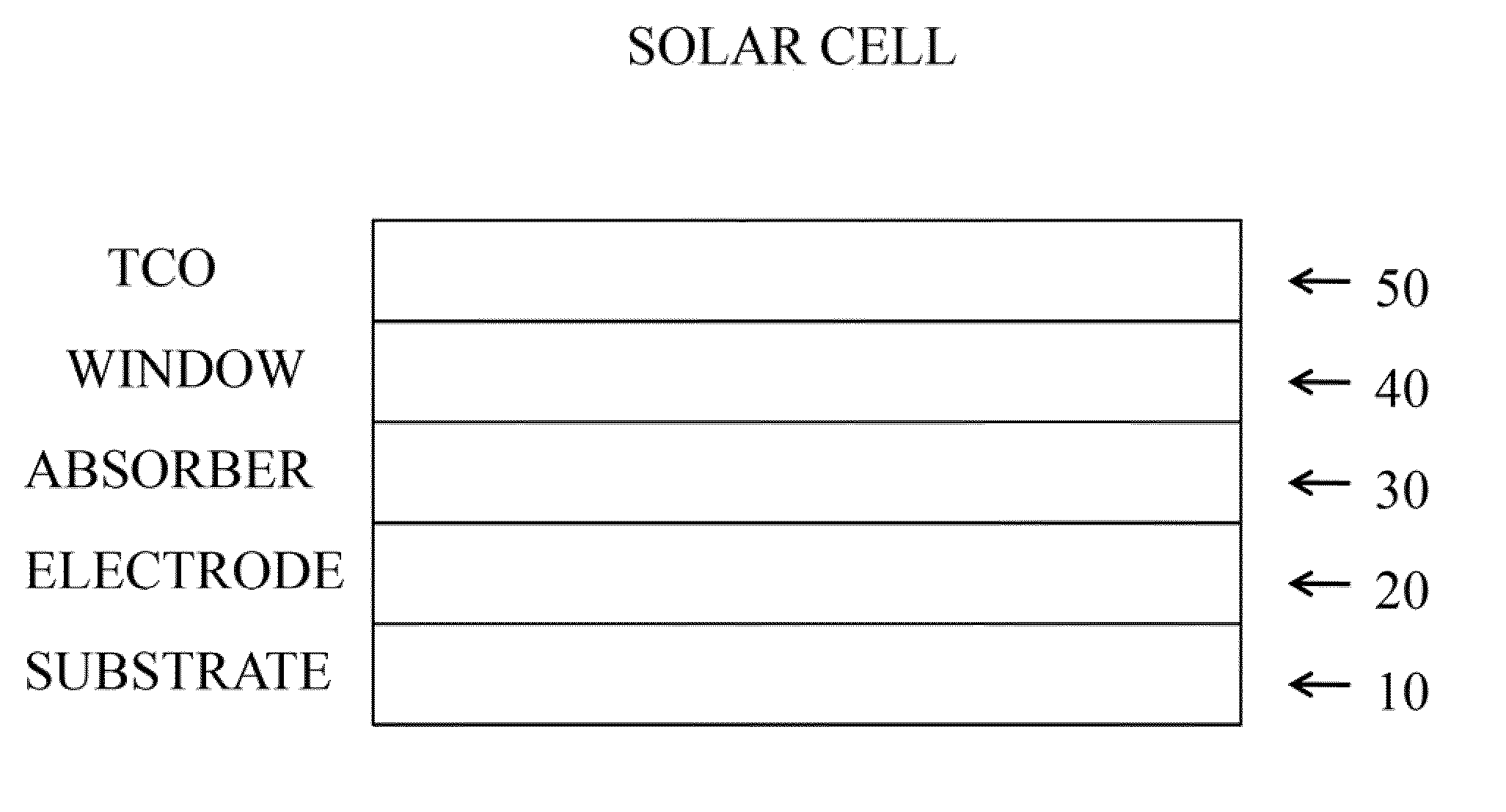
Methods and materials for cis and cigs photovoltaics
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Polymeric Precursor Compounds
[0499]A polymeric precursor represented by the formula {Cu(SesecBu)4In} was synthesized using the following procedure.
[0500]To a stirred solution of In(SesecBu)3 (2.60 g, 5 mmol) in benzene (10 mL) under inert atmosphere was added solid CuSesecBu (1.0 g, 5 mmol). The mixture was stirred at 25° C. for 12 h to produce a pale yellow solution. The solvent was removed from the reaction mixture under reduced pressure leaving a sticky yellow oil. The oil was dissolved in pentane and filtered. Solvent removal from the filtrate under reduced pressure yielded 3.1 g (86%).
[0501]NMR: (1H; C6D6) 0.99 (br, 12H), 1.70 (br d, 12H), 1.81 (m, 4H), 2.02 (br m, 4H), 3.67 (br, 4H).
[0502]In FIG. 8 is shown the TGA for this MPP polymeric precursor. The TGA showed a transition beginning at about 190° C., having a midpoint at about 210° C., and ending at about 230° C. The yield for the transition was 46.6% (w / w), as compared to a theoretical yield for the formula CuInSe2 of 46.5...
example 2
[0503]A polymeric precursor represented by the formula {Cu(SesecBu)4Ga} was synthesized using the following procedure.
[0504]To a stirred solution of Ga(SesecBu)3 (1.20 g, 2.5 mmol) in benzene (10 mL) under inert atmosphere was added solid CuSesecBu (0.51 g, 2.5 mmol). The mixture was stirred at 25° C. for 2 h to produce a pale yellow solution. The solvent was removed from the reaction mixture under reduced pressure leaving a sticky yellow oil. The oil was dissolved in pentane and filtered. Solvent removal from the filtrate under reduced pressure yielded 1.50 g (89%).
[0505]NMR: (1H; CDCl3) 0.98 (t, 12H), 1.58 (br, 12H), 1.74 (br, 4H), 1.96 (br, 4H), 3.44 (br, 4H).
[0506]In FIG. 9 is shown the TGA for this MPP polymeric precursor. The TGA showed a transition beginning at about 100° C. and ending at about 240° C. The yield for the transition was 44% (w / w), as compared to a theoretical yield for the formula CuGaSe2 of 43% (w / w). Thus, the TGA showed that this polymeric precursor can be u...
example 3
[0507]A polymeric precursor represented by the formula {Cu(StBu)4In} was synthesized using the following procedure.
[0508]A 100-mL Schlenk tube was charged with In(StBu)3 (0.55 g, 1.4 mmol) and CuStBu (0.21 g, 1.4 mmol). 10 mL of dry benzene was added. The reaction mixture was heated at 75° C. overnight. A colorless solid formed. The solution was filtered and the solid was washed with benzene at room temperature. The solid was dried under vacuum and collected (0.4 g, yield, 53%).
[0509]Elemental analysis: C, 36.2; H, 6.7; Cu, 13.0; In, 23.9; S, 18.0. NMR: (1H) 1.66 (br s 36H); (13C) 23.15 (s); 26.64 (s); 37.68 (s); 47.44 (s).
[0510]The TGA for this polymeric precursor showed a transition having a midpoint at 218° C., ending at 225° C. The yield for the transition was 46% (w / w), as compared to a theoretical yield for the formula CuInS2 of 45% (w / w). Thus, the TGA showed that this polymeric precursor can be used to prepare CuInS2 layers and materials, and can be used as a component to pr...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Temperature | aaaaa | aaaaa |
| Temperature | aaaaa | aaaaa |
| Temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap