Viral vector targeting cancer stem cells
a cancer stem cell and vector technology, applied in the field of recombinant adenoviral vectors, can solve the problems of unknown activity of cd133 promoters in cancer stem cells, little progress in the study of biological analyses and cancer treatments (in particular, cancer stem cell-specific treatments) that target cancer stem cells, and achieve high infiltration/metastatic ability, efficient development, and the effect of determining the prognosis of a cancer patien
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
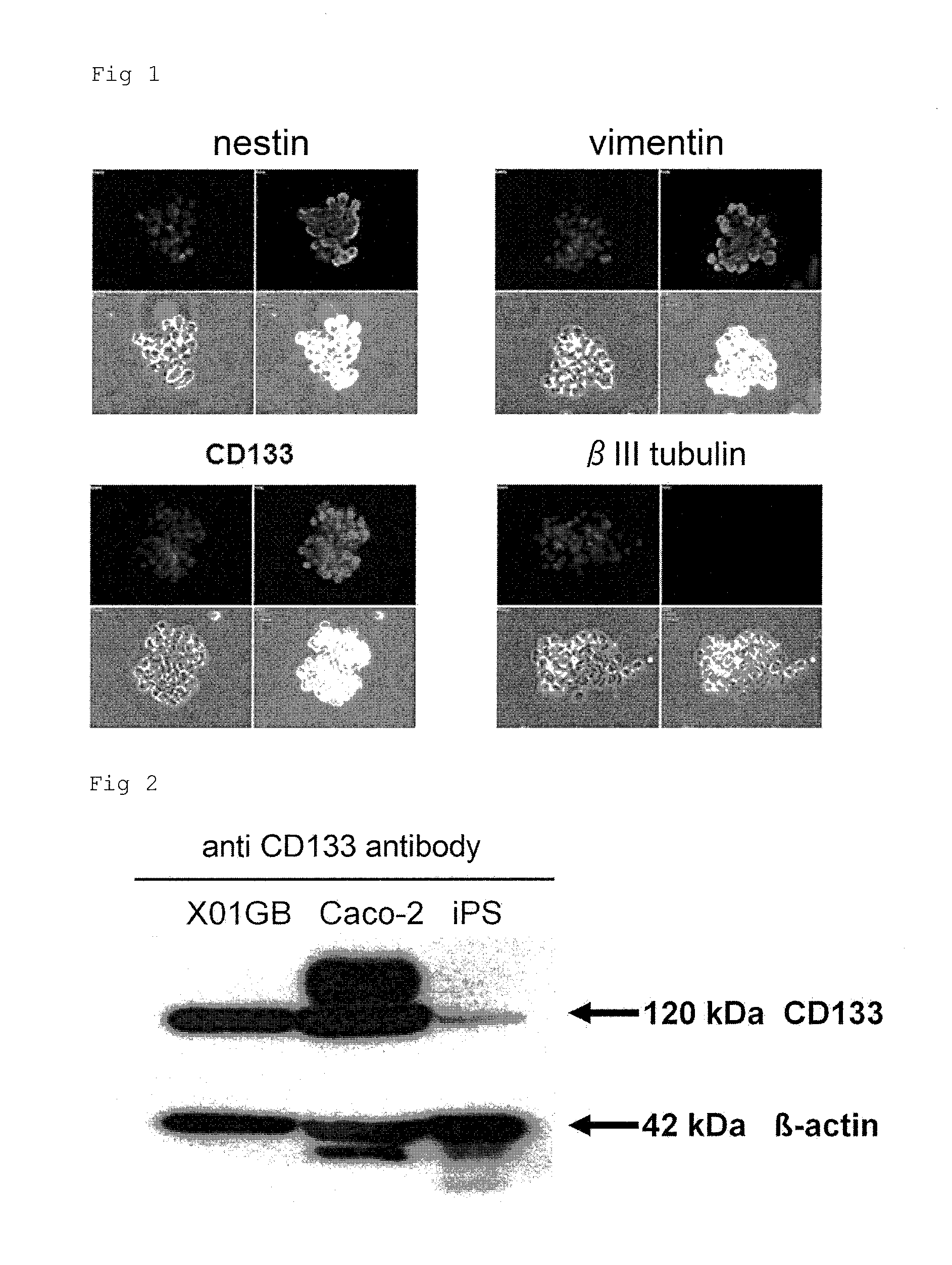
Confirmation of the Cancer Stem Cell Properties of Human Glioblastoma Stem Cells and the Expression of a Cancer Stem Cell Surface Marker CD133 by Performing Immunostaining on the Cells
(1) Cells
[0117]Human glioblastoma stem cells described in Soeda A, et al., J Biol Chem 2008; 283; 10958-66 were used herein. X01 GBS cells are a fraction of cancer stem cells in X01 GB cells, and are a group of cells prepared by “concentration” of a cancer stem cell fraction using a medium under undifferentiation conditions according to a Sphere culture method. Thus, unless otherwise specified, hereinafter, the term “human glioblastoma stem cells (X01 GBS)” means human glioblastoma stem cells (or X01 GB-CSC) described in Soeda A, et al., J Biol Chem 2008; 283; 10958-66. As culture conditions, glioblastoma stem cells (X01 GBS) were cultured at 5% CO2, 37° C. in a Dulbecco's modified Eagle's medium / F-12 (D6421, Sigma) medium containing B-27 (Invitrogen), 10% FBS, to which recombinant human FGF-2 (20 ng / m...
example 2
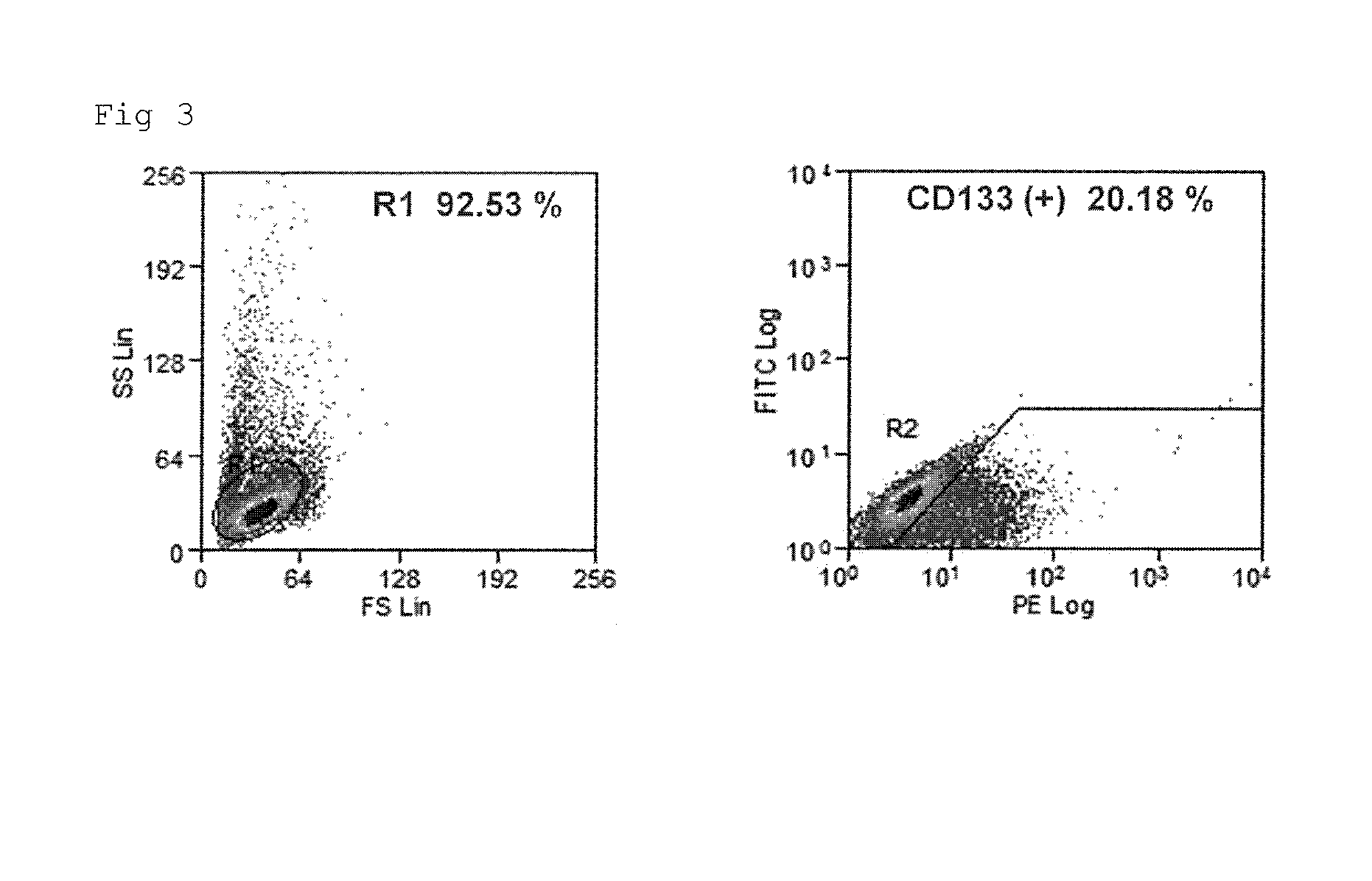
Confirmation by Western Blotting of Expression of Cancer Stem Cell Surface Marker CD133 on Human Glioblastoma Stem Cells (X01 GBS)
(1) Cells
[0130]Human glioblastoma stem cells (X01 GBS), and as positive controls, colon cancer cells (Caco-2) and human iPS cells (201B7, purchased from RIKEN), were used.
(2) Antibodies
[0131]An anti-CD133 rabbit polyclonal antibody (Ab19898, Abcam, UK) was used as a primary antibody (dilution rate 1:1000), and a goat anti-rabbit polyclonal antibody IgG / HRP (Dako, Cytomation) (dilution rate 1:2000) was used as a secondary antibody.
(3) Western Blotting
[0132]A culture solution in a 10-cm dish containing the cultured cells was discarded, and the dish was then washed with PBS. Thereafter, 1 mL of a cell lysis RIPA buffer (0.5% NP40, 0.1% SDS, 0.5% sodium deoxycholate, 150 mM NaCl, and 50 mM Tris (pH7.5)), which contained a 0.5 mM protein protective agent PMSF and Protease inhibitor cocktail (which was added immediately before the reaction), was added, so as to...
example 3
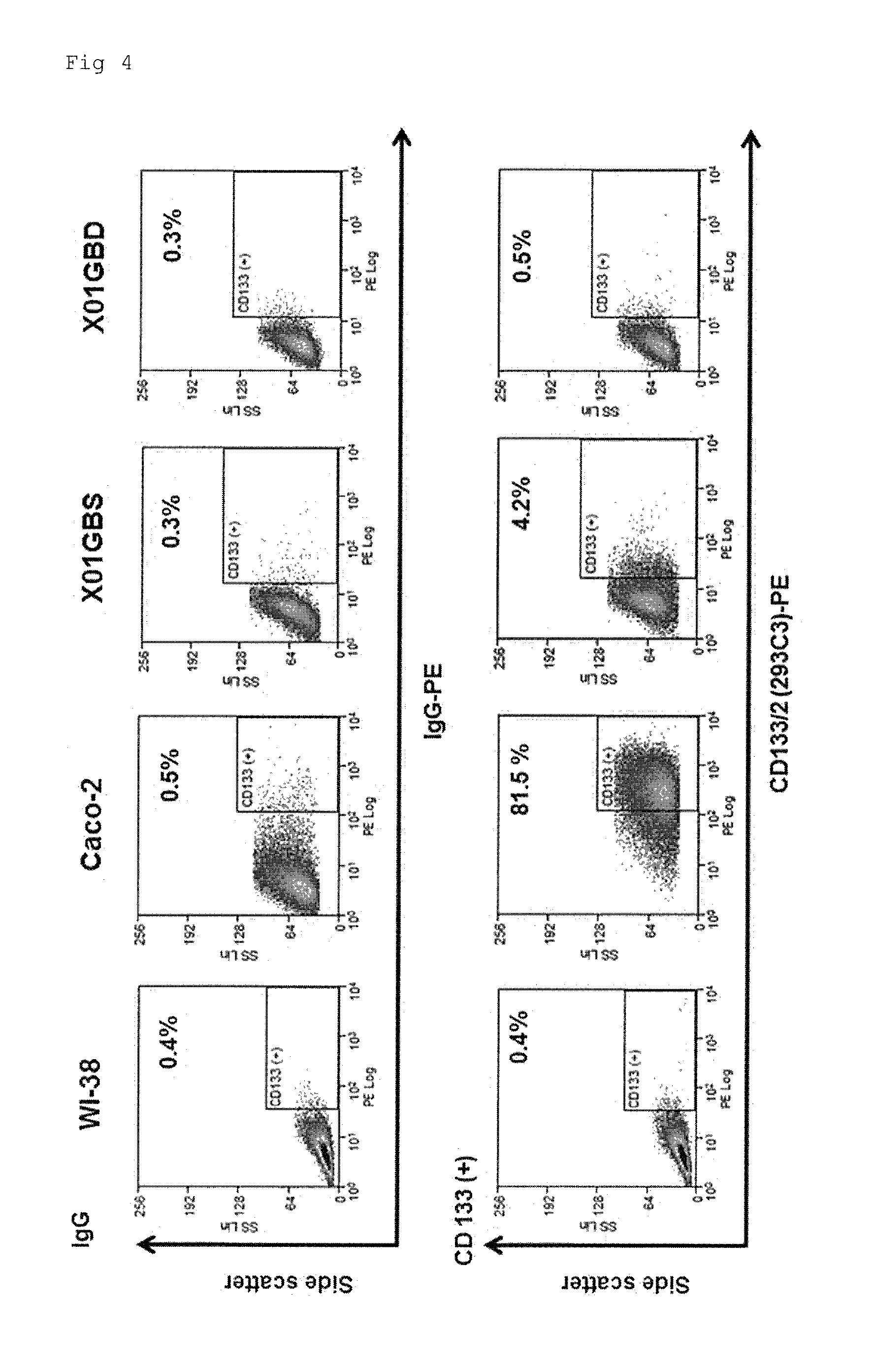
Confirmation by Flow Cytometry (FCM) of the Number of CD133(+) Cells
(1) FCM (1st Time)
[0134]FCM analysis was carried out in accordance with protocols associated with the antibody used in the analysis, produced by the manufacturer Miltenyi Biotec. When human glioblastoma stem cells (X01 GBS) were used, the content of CD133(+) cells was determined.
[0135]In the figure, cells contained in the range enclosed with an ellipse were defined as total cells, and were subjected to the analysis.
(2) FCM (2nd Time)
[0136]FACS was carried out in the same manner as in the 1st FCM. A mouse anti-human CD133 / 2 (293C3)-PE and mouse IgG-PE (both of which were manufactured by Miltenyi Biotec) were used as antibodies. Detection was carried out using BD FACSAria™ II Flow Cytometer.
[0137]From among a plurality of experiments performed, representative results are shown in FIG. 3. In the figure, the horizontal axis (PE Log) indicates CD133(+) cells as logarithmic values, and the longitudinal axis (FITC Log) has...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Composition | aaaaa | aaaaa |
| Antimicrobial properties | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap