Fidaxomycin crystal form II and preparation method thereof
A technology of fidaxomicin and crystal form, which is applied in the field of fidaxomicin crystal form II and its preparation, can solve the problems of poor crystal form stability and shorten product shelf life, achieve low cost, avoid environmental pollution and human body damage and good stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0046] Take 1.5 g of fidaxomicin, add 70 ml of a mixed solvent prepared by ethyl acetate:methylcyclohexane with a volume ratio of 1:1, and sonicate at room temperature for 1 minute to obtain a supersaturated solution. Crystallized at room temperature for 7 days. Suction filtration under reduced pressure, and the filter cake was vacuum-dried in an oven at 40°C for 2 hours to obtain Fidaxomycin Form II.
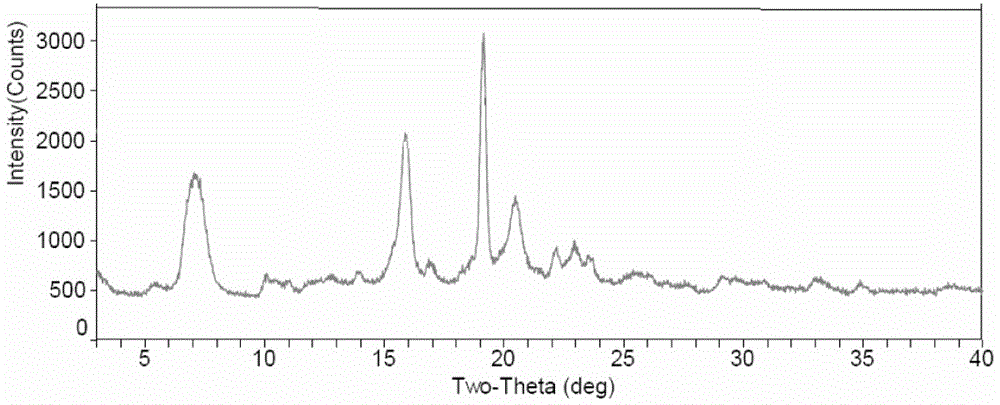
[0047] Measure the XRD value of the made Fidaxomycin crystal form II as shown in Table 1, and the spectrum is as follows figure 1 As shown, the figure shows that there are X-ray powder diffraction peaks in the X-ray powder diffraction pattern at 7.04°, 15.87°, 19.14°, 20.46° and 22.95±0.2°2θ.
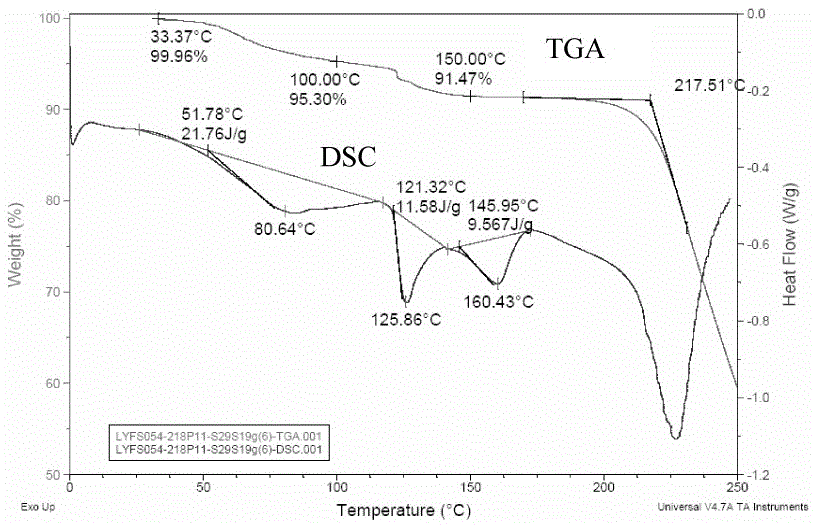
[0048] The TGA and DSC spectra of Fidaxomycin crystal form II are measured as figure 2 shown.
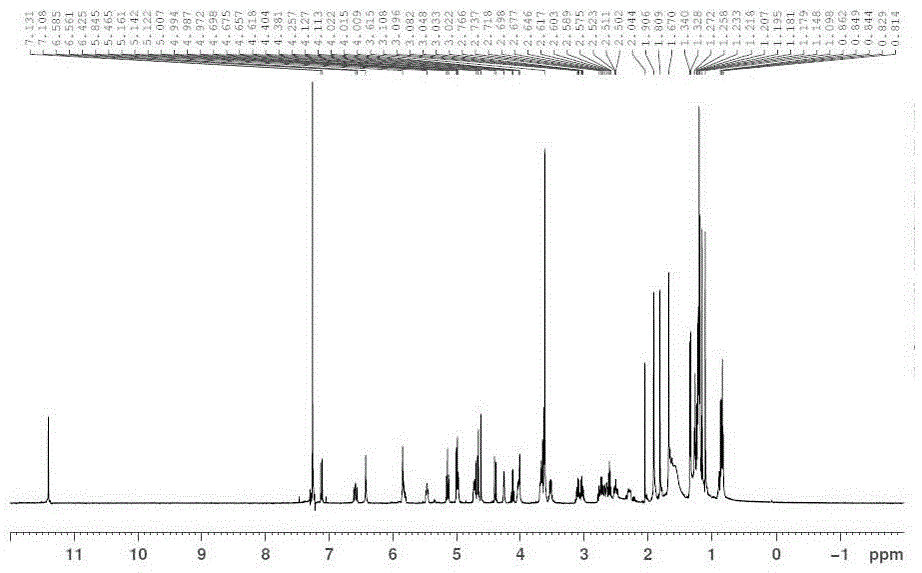
[0049] The 1H-NMR spectrum of Fidaxomycin crystal form II is measured as image 3 shown.
Embodiment 2
[0051] Take 1.5 g of fidaxomicin, add 70 ml of a mixed solvent prepared by heptane:ethyl acetate at a volume ratio of 1:1, and ultrasonicate at room temperature for 3 minutes to obtain a supersaturated solution. Placed at 40°C for 10 days to crystallize. Suction filtration under reduced pressure, and the filter cake was vacuum-dried in an oven at 20°C for 6 hours to obtain Fidaxomycin Form II.
Embodiment 3
[0053] Take 1.5 g of fidaxomicin, add 150 ml of a mixed solvent prepared by ethyl acetate:methylcyclohexane at a ratio of 0.1:1, and sonicate at room temperature for 5 minutes to obtain a supersaturated solution. Crystallize at 10°C for 5 days. Suction filtration under reduced pressure, and the filter cake was vacuum-dried in an oven at 30°C for 4 hours to obtain crystalline form II of Fidaxomycin.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More