Cetilistat solid dispersion and medicinal preparation prepared from the solid dispersion
A solid dispersion, the technology of new Lilistat, applied in the direction of drug combination, medical preparations with non-active ingredients, medical preparations containing active ingredients, etc., can solve the problem of affecting drug efficacy, low bioavailability, Problems such as low solubility
Active Publication Date: 2016-07-27
BEIJING WINSUNNY PHARMA CO LTD
View PDF4 Cites 5 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0005] Neolistat has very low solubility in water. EP1897558 discloses that its solubility in water at 37°C does not exceed 10 mg / L. Therefore, when the poorly soluble Neolistat is administered orally, its bioavailability is inevitably low , affecting the efficacy of the drug
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
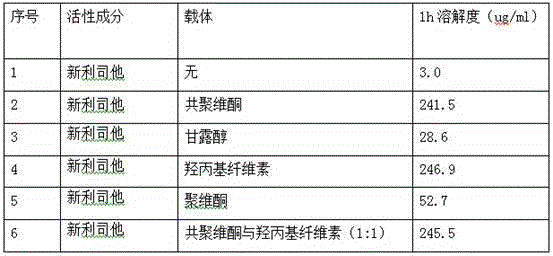
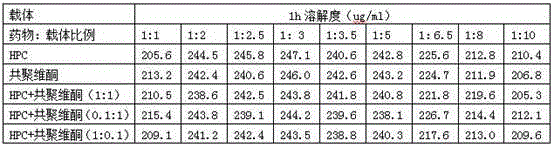
[0059] Mix 120mg of Neolistat and 240mg of hydroxypropyl cellulose evenly, heat in an oil bath, raise the temperature to 120°C, the mixture melts into a viscous shape, and cool rapidly; crush the cooled mixture and pass through a 60-mesh sieve to obtain Solid dispersion.
Embodiment 2
[0061] Dissolve 120 mg neolistat and 120 mg copovidone in 150 ml acetone, and dry in vacuum to obtain a solid dispersion.
Embodiment 3
[0063] Mix 120mg of Neolistat, 300mg of hydroxypropyl cellulose, and 300mg of copovidone evenly, heat in an oil bath, and raise the temperature to 110°C. 60 mesh sieve to obtain a solid dispersion.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention relates to cetilistat solid dispersion, and a medicinal preparation prepared from the solid dispersion. The cetilistat and one or both of the carriers hydroxypropyl cellulose and copovidone are prepared into a solid dispersion to significantly improve the solubility of the cetilistat, and overcome low solubility of cetilistat, so that the prepared medicinal preparation achieves good dissolution, and has increased bioavailability.
Description
technical field [0001] The invention relates to a solid dispersion of medicine for treating obesity and a pharmaceutical preparation thereof, in particular to a new lixistat solid dispersion and a pharmaceutical preparation thereof. Background technique [0002] Obesity is a chronic disease manifested by excess body fat tissue or an excessively high ratio of adipose tissue to other soft tissues. WHO defines obesity in adults: BMI [body mass index: weight / height squared (kg / m 2 )] at 25-29.9 is overweight, 30 is obese, and 40 is severely obese. With the improvement of living standards, the proportion of obese people in the total population of the society is increasing, and the prevalence of obesity is three times that of 20 years ago. The proportion of obese patients with metabolic syndrome such as diabetic lipid metabolism disorder is as high as 7.68%, and the risk of tumors is also increased. Numerous US and European studies have shown that mortality from obesity varies w...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): A61K31/536A61K9/14A61K47/38A61K47/32A61P3/04
Inventor 马贵红陈彦瑶刘丽敏耿玉先产运霞
Owner BEIJING WINSUNNY PHARMA CO LTD