A kind of cold compress and preparation method thereof
A technology of cold compress and formula, applied in the field of medical equipment and daily chemical industry, can solve the problems of affecting the effect of magnesium sulfate wet compress, inconvenient operation, easy falling off of gauze, etc., and achieves the promotion of industrial development, good social benefits, energy consumption and low energy consumption. low cost effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
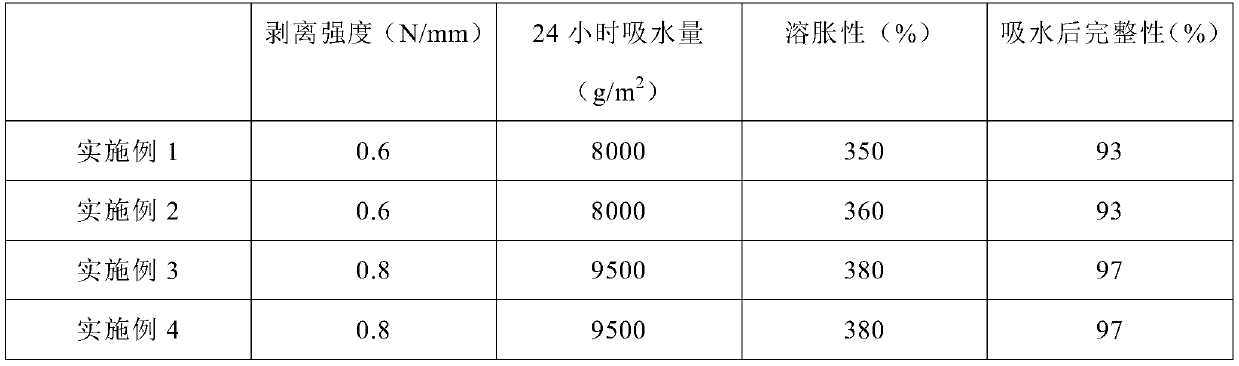
[0033] Such as figure 1 The cold compress shown includes a backing layer 3 (non-woven fabric), and a hydrogel layer 2 and an anti-adhesive layer 1 (polyethylene film) which are successively pasted on the upper surface. The hydrogel layer is made of It is made of hydrogel matrix, which includes the following parts by weight of oil phase, water phase and preservative, oil phase: 22 parts of glycerin, 5.2 parts of polyacrylic acid, 0.15 parts of aluminum dihydroxyaminoacetate, and 1 part of crospovidone ; Aqueous phase: 68.1 parts of purified water, 0.1 parts of disodium edetate, 1.1 parts of anhydrous magnesium sulfate, 2 parts of polyvinylpyrrolidone (K120), 0.2 parts of tartaric acid; 0.15 parts of preservative.
[0034] The specific steps of the above-mentioned preparation method of cold compress are as follows:
[0035] (1) Add the formula amount of polyacrylic acid, dihydroxy amino acetate and crospovidone into glycerin, mix well to obtain the oil phase, dissolve the formula amo...
Embodiment 2
[0041] Such as figure 1 The cold compress shown includes a backing layer 3 (non-woven fabric), and a hydrogel layer 2 and an anti-adhesive layer 1 (polyethylene film) which are successively pasted on the upper surface. The hydrogel layer is made of It is made of hydrogel matrix, which includes the following parts by weight of oil phase, water phase and preservative, oil phase: 22 parts of glycerin, 7 parts of polyacrylic acid, 0.17 parts of aluminum dihydroxyglycolate, and 5 parts of crospovidone ; Aqueous phase: 69 parts of purified water, 0.5 parts of disodium edetate, 20 parts of anhydrous magnesium sulfate, 3 parts of polyvinylpyrrolidone (K90), 0.5 parts of tartaric acid; 0.2 parts of preservatives.
[0042] The specific steps of the above-mentioned preparation method of cold compress are as follows:
[0043] (1) Add the formula amount of polyacrylic acid, dihydroxy amino acetate and crospovidone into glycerin, mix well to obtain the oil phase, dissolve the formula amount of p...
Embodiment 3
[0049] Such as figure 1 The cold compress shown includes a backing layer 3 (non-woven fabric), and a hydrogel layer 2 and an anti-adhesive layer 1 (polyethylene film) which are successively pasted on the upper surface. The hydrogel layer is made of It is made of hydrogel matrix and includes the following parts by weight of oil phase and water phase, oil phase: 18 parts of glycerin, 4 parts of polyacrylic acid, 0.13 parts of aluminum dihydroxyaminoacetate, 1 part of crospovidone, polyethylene 0.5 part of pyridine, 1 part of polymethacrylic acid, 1 part of hyaluronic acid; water phase: 65 parts of purified water, 0.1 part of disodium edetate, 0.5 part of anhydrous magnesium sulfate, polyvinylpyrrolidone (K120) 1 Parts, 0.2 parts of tartaric acid, 0.6 parts of snail mucus extract, 1.5 parts of hydroxypropyl cellulose, 2 parts of polyacrylamide.
[0050] The specific steps of the above-mentioned preparation method of cold compress are as follows:
[0051] (1) Add polyacrylic acid, alu...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap