Immunochromatographic reagent strip for fluorescent quantitative detection of PAPP-A (Pregnancy Associated Plasma Protein A) and preparation method of reagent strip
A fluorescent quantitative detection, PAPP-A technology, applied in the field of immunodiagnosis, can solve the problems of many influencing factors, high price, long time consumption, etc., to achieve the effect of ensuring accuracy, simple operation and high sensitivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0032] Preparation of PAPP-A fluorescent immunochromatographic test strips:
[0033] (1) Preparation of fluorescent microsphere-labeled protein
[0034]Take 0.1 mL of 10% fluorescent microspheres, centrifuge at 15,000 rpm for 15 minutes, adjust the concentration of the precipitate to 1% with 50 mM pH 6.5 MES buffer, and ultrasonically disperse; add carbodiimide (EDC) at a final concentration of 2 mg / mL ), mix well, then add N-hydroxysuccinimide (NHS) with a final concentration of 5 mg / mL, mix well; incubate at room temperature for 20 minutes and then centrifuge at 15,000 rpm for 15 minutes, and the precipitate is washed with 50 mM pH6.5 MES buffer dissolve. Ultrasonic disperse the reconstituted fluorescent microspheres, add 0.2 mg PAPP-A monoclonal antibody and 0.1 mg avidin respectively in two tubes, mix well, rotate and mix at room temperature for 2 hours, centrifuge at 15,000 rpm for 15 minutes, and use the precipitate Reconstitute in Tris-HCl (20 mM, pH 8.0) containing 3...
Embodiment 2
[0043] Quantitative detection of pregnancy-associated protein A (PAPP-A) concentration in blood samples by fluorescence immunochromatography
[0044] (1) Standard curve drawing
[0045] Prepare PAPP-A antigen with negative plasma to 1000 ng / mL, 500 ng / mL, 100 ng / mL, 50 ng / mL, 30 ng / mL, 10 ng / mL, 0 ng / mL, use the same batch of reagents, Each concentration point was tested 6 times. Take the fluorescence intensity ratio of the detection line (T band) and the quality control line (C band) as the ordinate, and the concentration of PAPP-A reference substance as the abscissa, establish an equation and fit it into a standard curve, and use the programming software to record the standard curve information Write to the ID chip.
[0046] (2) Detection of samples:
[0047] Take out the test strip from the kit, tear open the aluminum foil bag, lay the test strip flat, and equilibrate for 5 minutes. Take 100 μL of sample and add it to the sample well, and react at room temperature for 15...
Embodiment 3
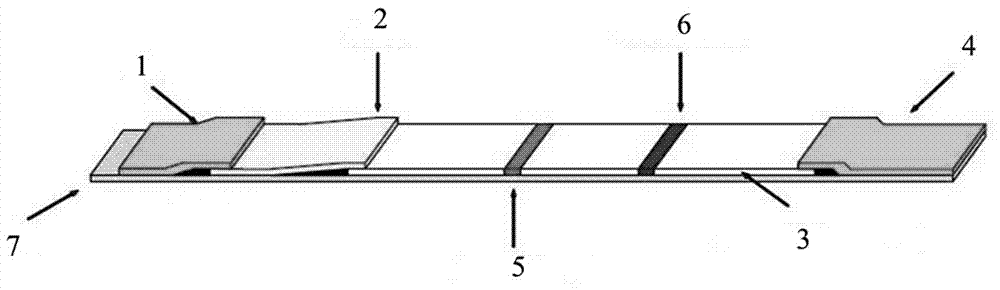
[0050] Please refer to figure 1 , an immunochromatographic test paper for fluorescent quantitative detection of PAPP-A, the immunochromatographic test strip includes a PVC base plate 7, and the PCV base plate 7 is sequentially provided with a sample pad 1, a marking pad 2, and a coating film 3. Absorbent paper 4, the marking pad 2 is connected to the sample pad 1, the coating film 3 is connected to the marking pad 2, the absorbent paper 4 is connected to the coating film 3 are connected; the PAPP-A monoclonal antibody labeled with fluorescent microspheres and avidin labeled with fluorescent microspheres are sprayed on the marker pad 2; detection lines 5 and quality control lines 6 are provided on the coating film 3, The detection line 5 and the quality control line 6 are separated by 4-8 mm, and the detection line 5 is coated with another PAPP-A monoclonal antibody that is at a different epitope from the PAPP-A monoclonal antibody labeled with the fluorescent microspheres , t...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Particle size | aaaaa | aaaaa |
| Excitation wavelength | aaaaa | aaaaa |
| Emission wavelength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap