Nitrogen-containing fused ring compounds as well as preparation method and application thereof
A compound and heterocycloalkyl technology, applied in the field of nitrogen-containing fused-ring compounds and their preparation, can solve the problems of low target inhibition activity, easy generation of drug resistance, poor pharmacokinetic properties of compounds, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
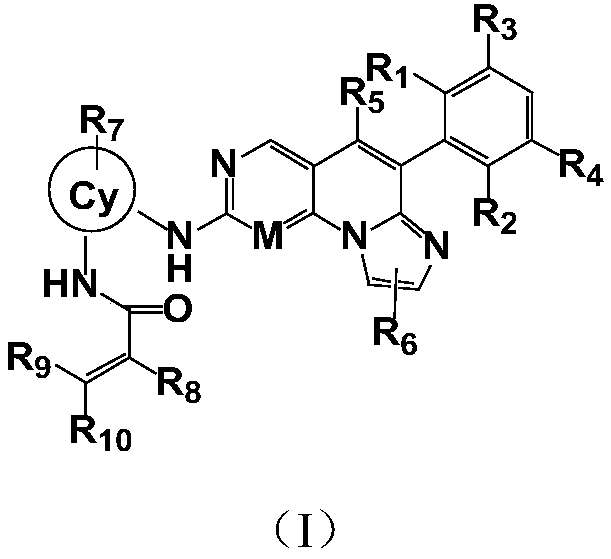
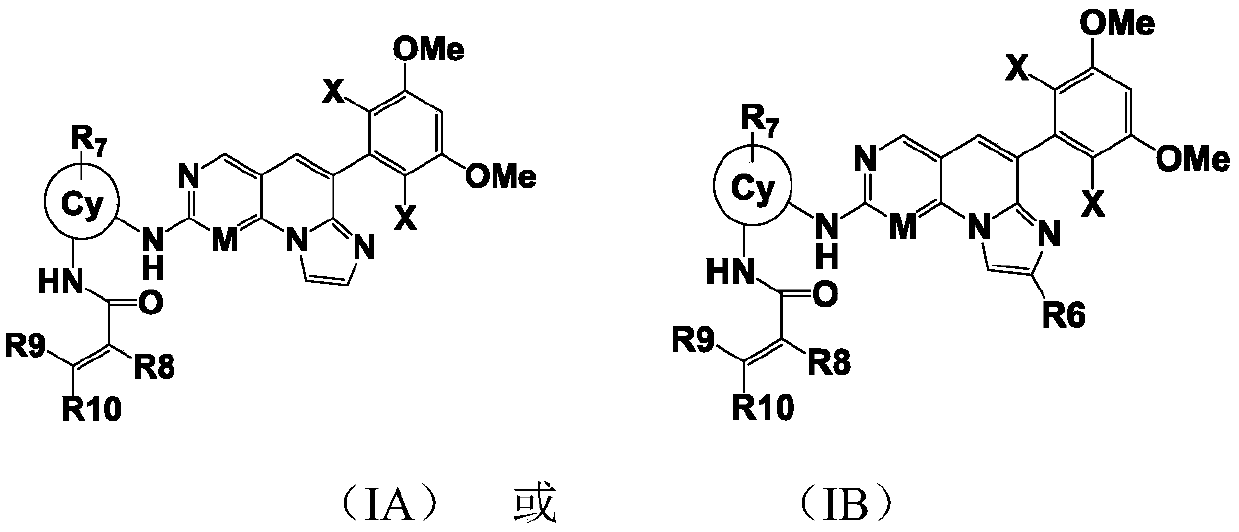
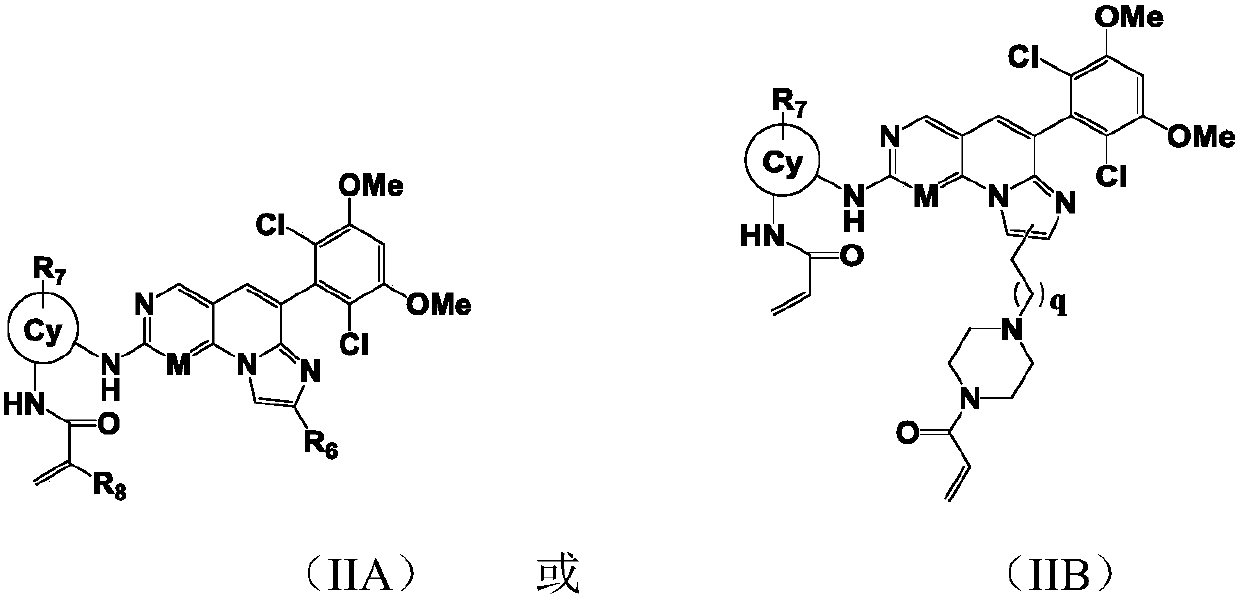
[0236] Example 1: N-(2-((6-(2,6-dichloro-3,5-dimethoxyphenyl)imidazol[1',2':1,6]pyridin[2,3- d] pyrimidin-2-yl) amino) phenyl) -2-fluoroacrylamide
[0237] LC-MS:ESI[M+H] +=553.2 / 555.2; 1H-NMR (400MHz, CD3OD) δ9.83(s, 1H), 9.50(s, 1H), 9.14(s, 1H), 8.16(s, 1H), 7.87(d, J=7.6 Hz,1H),7.69(d,J=6.8Hz,1H),7.52(s,1H),7.50(s,1H),7.21-7.31(m,2H),7.06(s,1H),6.24(dd , J=2.0, 17.2Hz, 1H), 5.72(dd, J=2.0, 10.4Hz, 1H), 3.99(s, 6H).
Embodiment 2
[0238] Example 2: N-(2-((4-(2,6-dichloro-3,5-dimethoxyphenyl)imidazo[1,2-a][1,6]naphthyridine-8- Base) amino) phenyl) -2-fluoroacrylamide
[0239] LC-MS:ESI[M+H] + =552.4 / 554.4; 1H-NMR (400MHz, CD3OD) δ9.88(s, 1H), 8.83(s, 1H), 8.74(s, 1H), 8.33(s, 1H), 7.70(d, J=7.6 Hz,1H),7.63(d,J=7.6Hz,1H),7.50(s,1H),7.46(s,1H),7.15-7.28(m,3H),7.06(s,1H),6.24(dd , J=2.0, 16.8Hz, 1H), 5.72(dd, J=2.0, 10.4Hz, 1H), 4.00(s, 6H).
Embodiment 3
[0240] Example 3: N-(3-((6-(2,6-dichloro-3,5-dimethoxybenzene)imidazol[1',2':1,6]pyridine[2,3-d ]pyrimidin-2-yl)amino)-1-methyl-1H-pyrazol-4-yl)-2-fluoroacrylamide
[0241] LC-MS:ESI[M+H] + =557.2559.2; 1H-NMR (400MHz, CD3OD) δ10.91(s, 1H), 10.07(s, 1H), 9.14(s, 1H), 8.64(s, 1H), 8.47(s, 1H), 7.51-7.60(m,2H),7.07(s,1H),6.37(dd,J=1.6,8.8Hz,1H),5.84(d,J=10.4Hz,1H),3.99(s,6H),3.93 (s,1H).
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com