Application of dodder polysaccharide in preparation of medicine for treating enteritis caused by 5-fluorouracil
A technology of fluorouracil and dodder, applied in the field of medicine, can solve problems such as affecting DNA synthesis and repair, unclear efficacy of intestinal mucositis, disturbing the immune system of the body, etc. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] The preparation of embodiment 1 dodder polysaccharide (PCCL)
[0030] Cuscuta (CCL) was provided by Guangdong Hexiang Pharmaceutical Co., Ltd. Put Cuscuta dry powder (200g) in 1.6L of distilled water and mix evenly, extract at a temperature of about 98-100°C for 4 hours, and repeat twice. Afterwards, the whole extract was filtered and centrifuged at 1000×g (30 min, 4° C.). Concentrate the supernatant to 100ml at room temperature, add 95% ethanol, and precipitate at a ratio of 1:4.3. After 24 hours of precipitation, the centrifuged sample as described above was obtained, and the precipitate was dissolved in 100 ml of distilled water. This precipitation process was repeated three times. Then use sevage reagent (isoamyl alcohol: chloroform = 1:4) and freeze-dry to obtain dodder polysaccharide. The PCCL obtained from the original raw material accounted for about 17.6%. Using the phenol-sulfuric acid method, using glucose as the standard, the PCCL crude sugar content was...
Embodiment 2
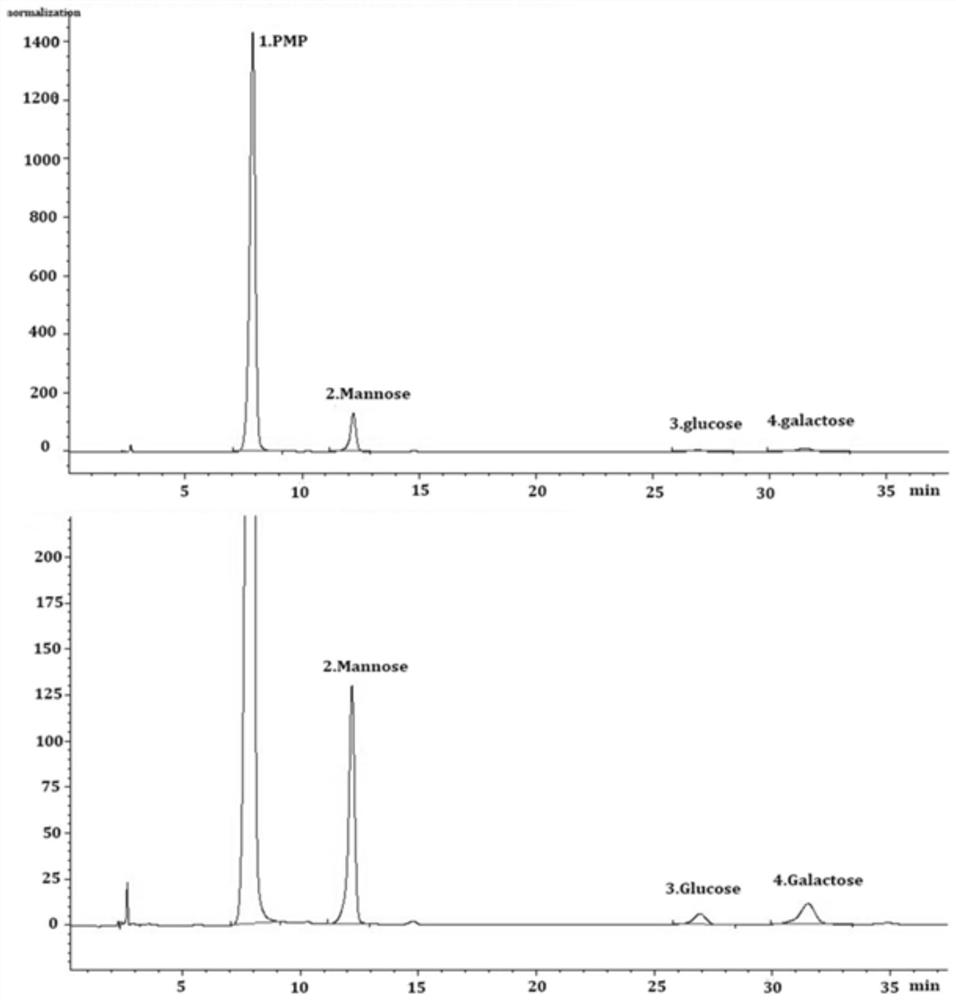
[0031] Composition determination of embodiment 2 dodder polysaccharide
[0032] Before HPLC analysis, 1ml 4mol / L trifluoroacetic acid and 1ml 1g / L PCCL were hydrolyzed at 110°C for 2h and cooled to room temperature. The solution was evaporated to dryness at 70°C. Methanol was added three times to remove residual amounts of trifluoroacetic acid. After drying, dissolve the solution with 1 ml of water. Sodium hydroxide (200 μL; 0.3 mol / L) and PMP methanol solution (200 μL; 0.5 mol / L) were added to 200 μL of the solution. Then place it in a 70°C water tank to cool for 30 minutes, cool to room temperature, and add 200 μL of 0.3 mol / L hydrochloric acid solution, 1.2 mL of water and 2 mL of chloroform. The mixture was vortexed for 60s, centrifuged (1500xg; 5min). The supernatant was filtered through a 0.45 μm filter membrane.
[0033] In this experiment, Luna C18 column was used for HPLC analysis. The mobile phase was 0.1 mol / L potassium dihydrogen phosphate buffer (pH 6.8): ac...
Embodiment 3
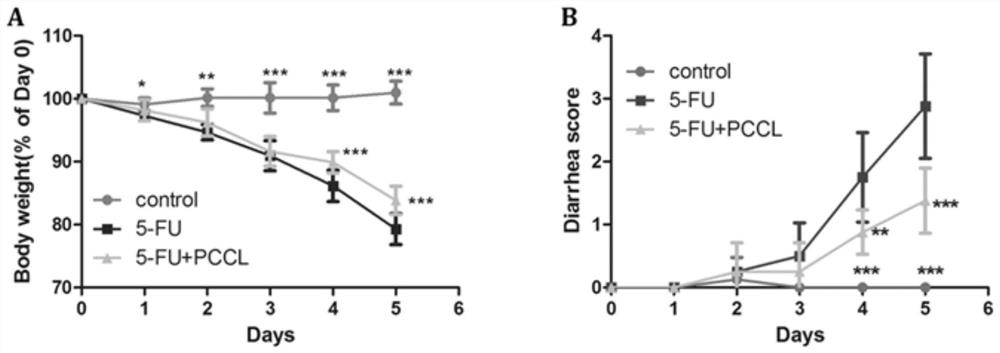
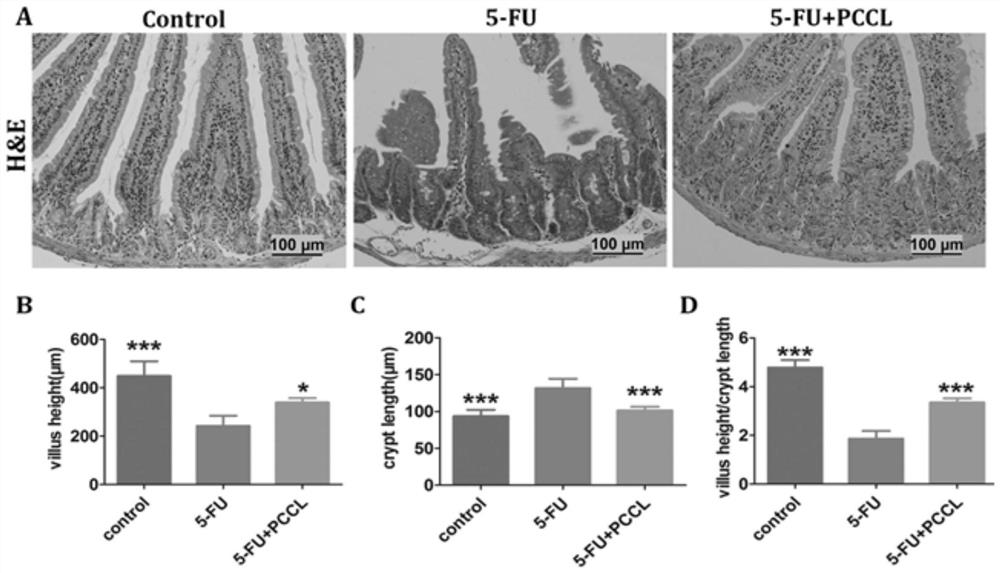
[0035] Example 3 Effect of dodder polysaccharide on weight loss and diarrhea score during 5-FU treatment
[0036] The experimental animals were SPF grade C57BL / 6 mice, 7 weeks old, male, weighing 18-22 g, purchased from Guangdong Medical Experimental Animal Center. All mice were acclimatized for 7 days prior to the experiment. Mice were housed in cages, given food and water, maintained at 22±1° C., and housed under a 12-hour light / dark cycle. All methods were approved by the Guangzhou Institute of Sports Science Animal Ethics Standards. At the same time, the animals were randomly divided into three groups: control group (control, PBS), 5-FU group (5-FU), and 5-FU+PCCL group (5-FU+PCCL). The volume of PBS administered to the control group was equal to the volume of PCCL administered to the 5-FU+PCCL group. Except the control group, the other two groups were intraperitoneally injected with 5-FU (50 mg / kg), once a day for 5 consecutive days. Give PCCL (20mg / kg) orally every d...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap