Composite artificial cornea and preparation method thereof
A kind of artificial cornea and composite technology, applied in the field of biomedical engineering, can solve the problems of artificial cornea that cannot be healed, the process is complicated, and the joint strength is not enough, and it is beneficial to surgical suture, with good biocompatibility and high tensile strength. high effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
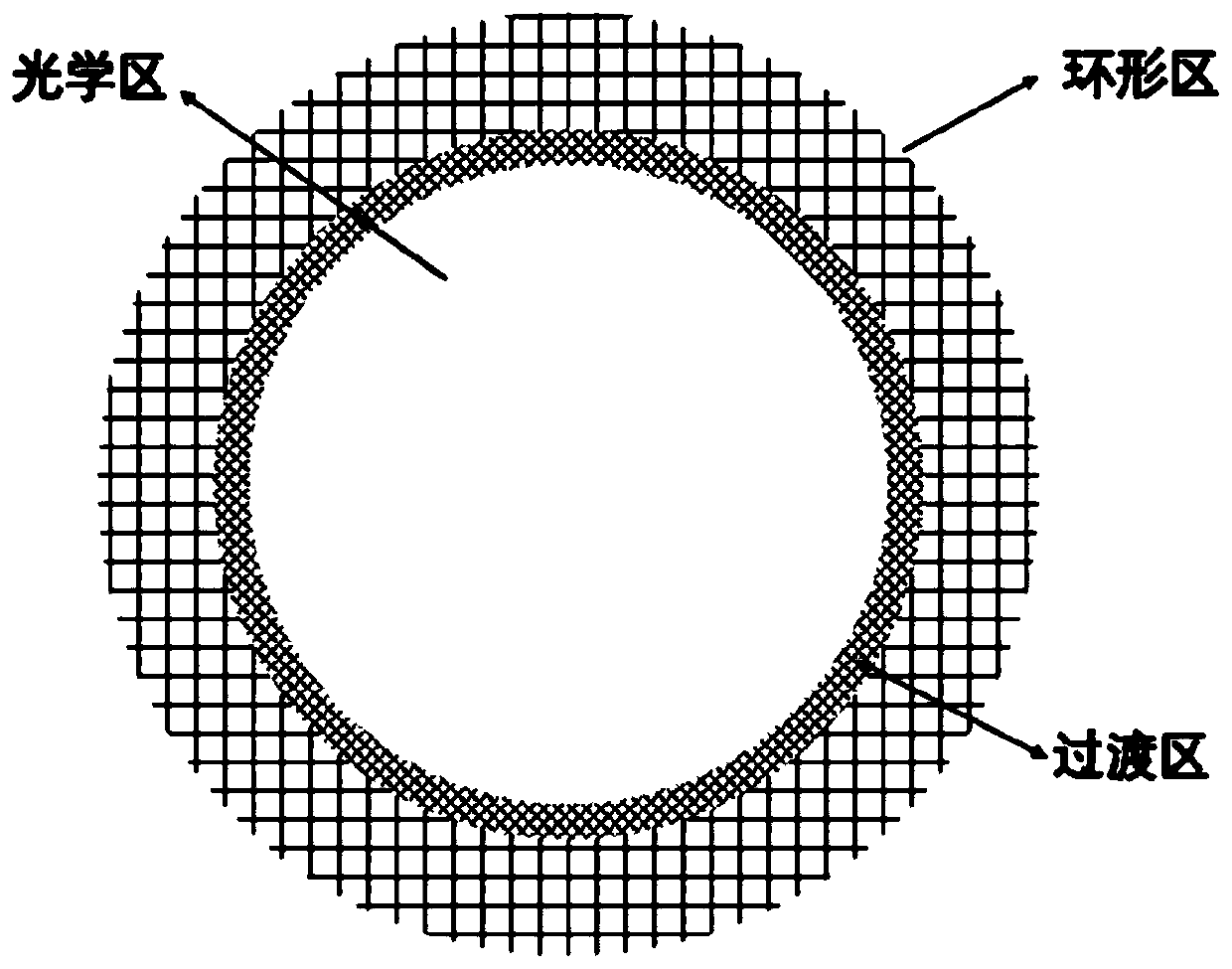
[0044] now refer to figure 1 , which shows a composite artificial cornea constructed and operated according to a specific embodiment, which includes a central optic zone made of a polymer and a corneal annular zone made of acellular matrix, wherein, in the central optic zone and the corneal annular zone The transition zone forms an interpenetrating network. The preparation method of above-mentioned composite artificial cornea comprises the following steps:
[0045] (1) Putting the acellular matrix material into a designated area in a mold of a specific size, placing the upper and lower molds, and applying appropriate pressure to obtain an artificial corneal stent;
[0046] (2) punch a central hole with a diameter of 2-8mm according to the design requirements in the central part of the artificial corneal stent prepared in step (1), and place it in the cornea forming mold, and drop the solution for preparing the central optical zone in the central hole, Closing the mold and ut...
Embodiment 1
[0055] A preparation method of a composite artificial cornea, comprising the following steps:
[0056] (1) Put the freeze-dried acellular matrix material into a designated area in a circular mold with a diameter of 9 mm, place the upper and lower molds, and apply pressure to obtain an artificial corneal stent;
[0057] (2) The central part of the artificial corneal stent prepared in step (1) is drilled a central hole with a diameter of 7mm according to the design requirements, and placed in the cornea forming mold, and the polymethacrylic acid 2-hydroxyethyl ether is added dropwise in the central hole The ester solution, the mold is closed, and the polymerization reaction of 2-hydroxyethyl methacrylate monomer with water and initiator is used to form an interpenetrating network in the transition zone between the central optical zone and the corneal annular zone;
[0058] (3) The artificial cornea obtained in step (2) is fully soaked in deionized water for 96 hours to remove th...
Embodiment 2
[0062] A preparation method of a composite artificial cornea, comprising the following steps:
[0063] (1) Put the freeze-dried acellular matrix material into a designated area in a circular mold with a diameter of 9 mm, place the upper and lower molds, and apply pressure to obtain an artificial corneal stent;
[0064] (2) The central part of the artificial corneal stent prepared in step (1) is drilled a central hole with a diameter of 8 mm according to the design requirements, and placed in a cornea forming mold, and the solution for preparing polymethylacrylate is added dropwise in the central hole, and closed a mold, utilizing the polymerization reaction of methyl (meth)acrylate monomer with water and an initiator, so that the transition zone between the central optical zone and the corneal annular zone forms an interpenetrating network;
[0065] (3) The artificial cornea obtained in step (2) is fully soaked in deionized water for 48 hours to remove the residual monomer in ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| pore size | aaaaa | aaaaa |
| tensile strength | aaaaa | aaaaa |
| tensile strength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap