A class of pyridylaminophthalide compounds, their preparation method and use
A picolinylaminophthalide and compound technology, which is applied to a class of picolinylaminophthalide compounds, and the fields of their preparation and use, can solve the problem of single action target, poor long-term efficacy in AD patients, and many toxic and side effects, etc. question
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
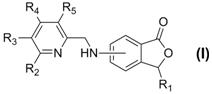
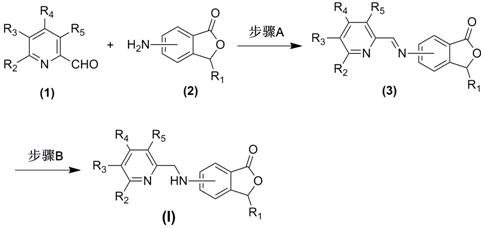
[0036] Example 1 General method for the preparation of pyridylamino-3-substituted phthalides (3)
[0037] Add 2.4 mmol of the corresponding 2-pyridinecarbaldehyde compound (1), 2.0 mmol of the corresponding amino-3-substituted phthalide compound (2) and 20 ml of ethanol into the reaction flask, stir evenly, then raise the temperature and reflux and stir for 2.0~ 12.0 hours (the reaction process is monitored by TLC); after the reaction is completed, cool to room temperature, evaporate the solvent under reduced pressure, and the residue is purified by silica gel column chromatography to obtain the corresponding pyridyl imino-3-substituted phthalide compound ( 3), the yield is 52.6%-83.5%, and its chemical structure has been tested 1 Confirmed by H-NMR and ESI-MS.
Embodiment 2
[0038] Example 2 The General Method for the Preparation of Pyridylaminophthalides (I)
[0039] Add 1.0 mmol of the corresponding pyridylidene amino-3-substituted phthalide compound (3) prepared in Example 1 and 10 ml of tetrahydrofuran into the reaction flask, stir at room temperature, add 3.0 mmol of sodium borohydride, and continue to Stir the reaction for 1.0 to 15.0 hours (the reaction process is monitored by TLC); after the reaction, adjust the pH of the reaction solution to strong acidity with 10% hydrochloric acid aqueous solution, then adjust the pH of the reaction solution to weak alkalinity with saturated aqueous sodium bicarbonate solution, evaporate under reduced pressure Remove tetrahydrofuran, add 20 mL of deionized water to the residual liquid, extract three times with 60 mL of ethyl acetate, combine the organic layers and wash with saturated aqueous sodium chloride solution, dry over anhydrous sodium sulfate and filter, evaporate the solvent under reduced pressu...
Embodiment 3
[0052] Example 3 General method for the preparation of pyridylaminophthalide compound (I) and acid salt formation
[0053]Add 1.0 mmol of the pyridylaminophthalide compound (I) obtained according to the above-mentioned Example 2 and 20 ml of acetone into the reaction flask, stir well, add 3.0 mmol of the corresponding acid, heat and reflux and stir for 20 minutes, after the reaction is completed After cooling to room temperature, the solvent was evaporated under reduced pressure, and the residue was recrystallized by conventional methods to obtain the corresponding salt of pyrylaminophthalide compound (I), whose chemical structure was confirmed by 1 Confirmed by H NMR and ESI-MS.
PUM
| Property | Measurement | Unit |
|---|---|---|
| control rate | aaaaa | aaaaa |
| control rate | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap