Composition for preventing or treating cancer containing novel trifluoromethylphenylpyrazole derivative as active ingredient
A trifluoromethyl, phenyl technology, applied in the field of compositions containing novel trifluoromethyl phenylpyrazole derivatives as active ingredients for preventing or treating cancer, can solve the problem of no therapeutic effect and reduce the efficiency of radiotherapy , serious side effects of normal cells, etc., to achieve the effect of reducing radioresistance and excellent apoptosis
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment
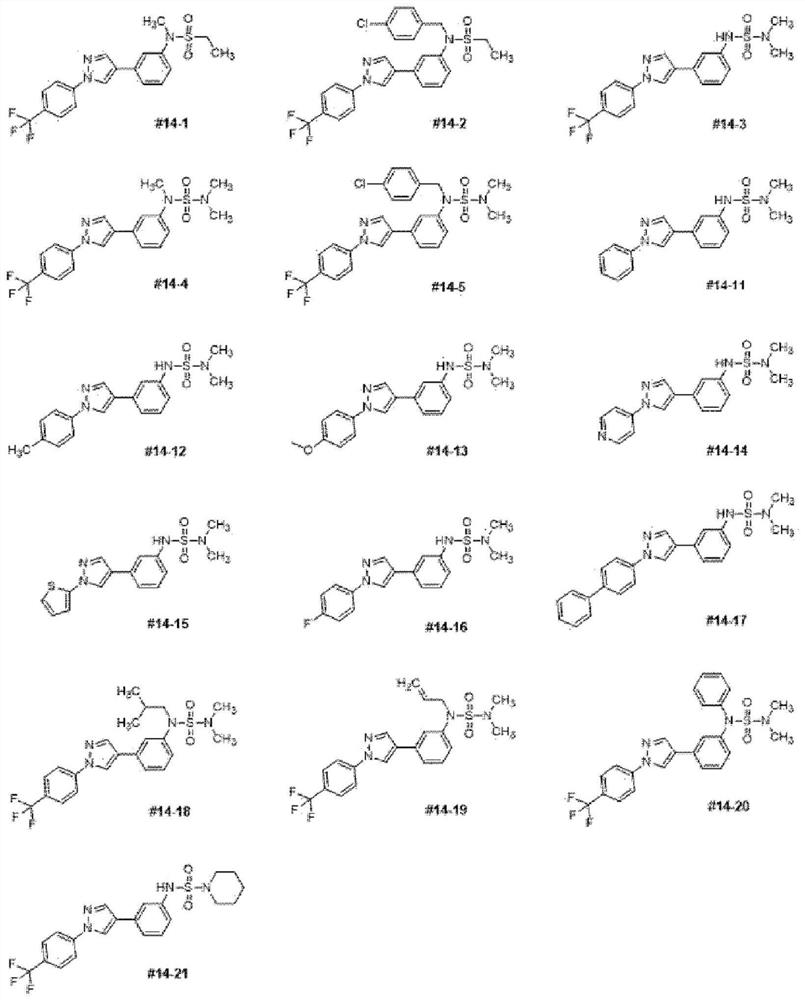
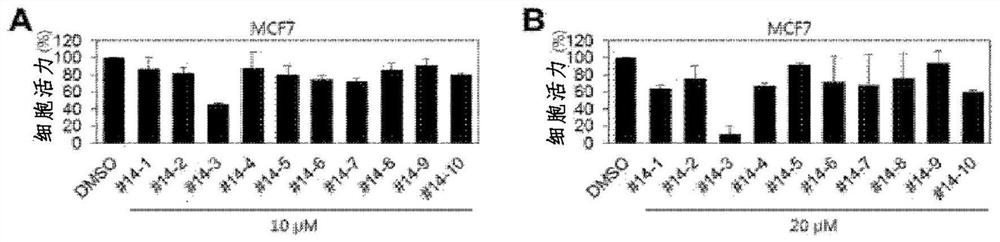
[0110] Hereinafter, the present invention will be described in detail by way of examples. These embodiments are only for illustrating the present invention in more detail, and it will be apparent to those skilled in the art that the scope of the present invention is not limited by these embodiments according to the gist of the present invention.
preparation example
[0111] 3-(1-(4-(trifluoromethyl)phenyl)-1H-pyrazol-4-yl)aniline
[0112]
[0113] Step 1: Combine 4-iodotrifluorotoluene (1.00 g, 3.68 mmol), 4-chloro-1H-pyrazole (415 mg, 4.05 mmol), cesium carbonate (2.40 g, 7.36 mmol), CuI (105 mg, 0.55 mmol) ) was added to anhydrous toluene (35 mL) as a reaction solvent, followed by stirring at 110° C. for 24 hours. After the reaction product was confirmed by TLC, the reaction solution was diluted with EtOAc and washed with purified water. Anhydrous Na for organic layer 2 SO 4 It was dehydrated, concentrated under reduced pressure, and separated by silica gel column chromatography to obtain 1.12 g of the title compound (Compound 1). 1 H-NMR (300MHz, CDCl 3 ): δ8.23(s, 1H), 8.02(s, 1H), 7.89(2H, d, J=8.77Hz), 7.76(2H, d, J=8.77Hz); LC-MS (ESI, m / z)=247.0(M+H + ).
[0114] Step 2: The compound 1 obtained in step 1 (800 mg, 3.25 mmol) was put into a reaction vessel, and 3-aminophenylboronic acid (534 mg, 3.90 mmol), Pd(OAc) 2 (73...
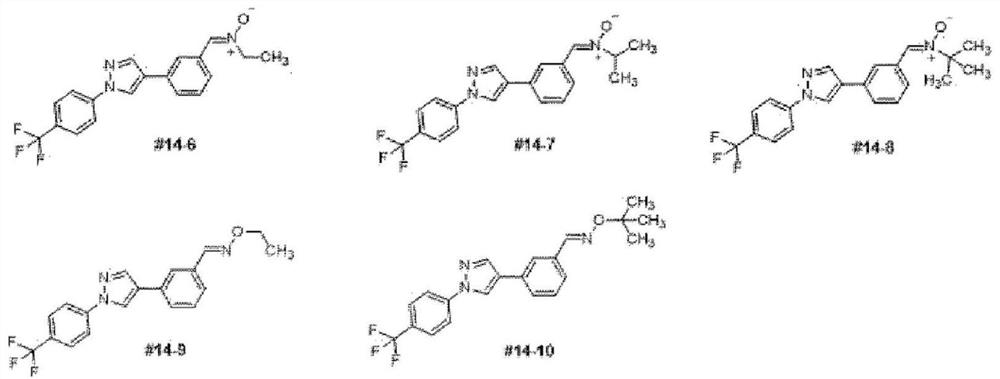
preparation example 2
[0115] 2-(1-(4-(trifluoromethyl)phenyl)-1H-pyrazol-4-yl)benzaldehyde
[0116]
[0117] Compound 1 (800 mg, 3.25 mmol) obtained in Step 1 of Preparation Example 1 was placed in a reaction vessel, and (3-formylphenyl)boronic acid (585 mg, 3.90 mmol), Pd (PPh 3 ) 4 (375mg, 0.33mmol), Na 2 CO 3 (689 mg, 6.54 mmol) was added to dioxane (32 mL) followed by N 2(g) Treatment for 5 minutes. After stirring the reaction at 90°C for 8 hours, the product was confirmed by TLC and the solvent dioxane was removed by concentration under reduced pressure. The reaction concentrate was diluted with EtOAc and washed with purified water. Put the organic layer in anhydrous Na 2 SO 4 It was dehydrated, concentrated under reduced pressure, and separated by silica gel column chromatography to obtain 870 mg of the title compound (Compound 3). 1 H-NMR (300MHz, CDCl 3 ): δ9.85(s, 1H), 8.61(s, 1H), 8.21(s, 1H), 8.08(s, 1H), 7.95(1H, d, J=7.86Hz), 7.81(2H, d, J=8.74Hz), 7.67(2H,d,J=8.77Hz), 7....
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap