Pharmaceutical formulations containing lipoic acid derivatives
a technology of lipoic acid and derivatives, which is applied in the direction of anhydride/acid/halide active ingredients, biocide, drug compositions, etc., can solve the problem of no specific guidance regarding the selection of suitable pharmaceutically acceptable carriers
Inactive Publication Date: 2008-10-23
CORNERSTONE PHARMA INC
View PDF31 Cites 30 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
[0010]Further preferred embodiments of this invention include those in which the lipoic acid derivative is present in a therapeutically effective amount. Still further preferred embodiments of this invention include those in which the ion pairing agent is selected from the group consisting of triethanolamine, polyethyleneimine, monoethanolamine, diethanolamine, mefanamic acid, tromethamine and combinations thereof, those in which the ion pairing agent is a polymer-conjugated ion pairing agent, and those in which the ion pairing agent and the at least one lipoic acid derivative is present in a ratio ranging from about 1000:1 to about 1:1000. Further preferred embodiments of the present invention also include those in which the diluent is selected from the group consisting of saline, a sugar solution, an alcohol, dimethylformamide, dimethylsulfoxide, dimethylacetamide and combinations thereof.
[0011]In a se...
Problems solved by technology
However, these patents provide no specific guidance with regard to the selection of suitable pharmaceutically acceptable carriers.
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
example 1
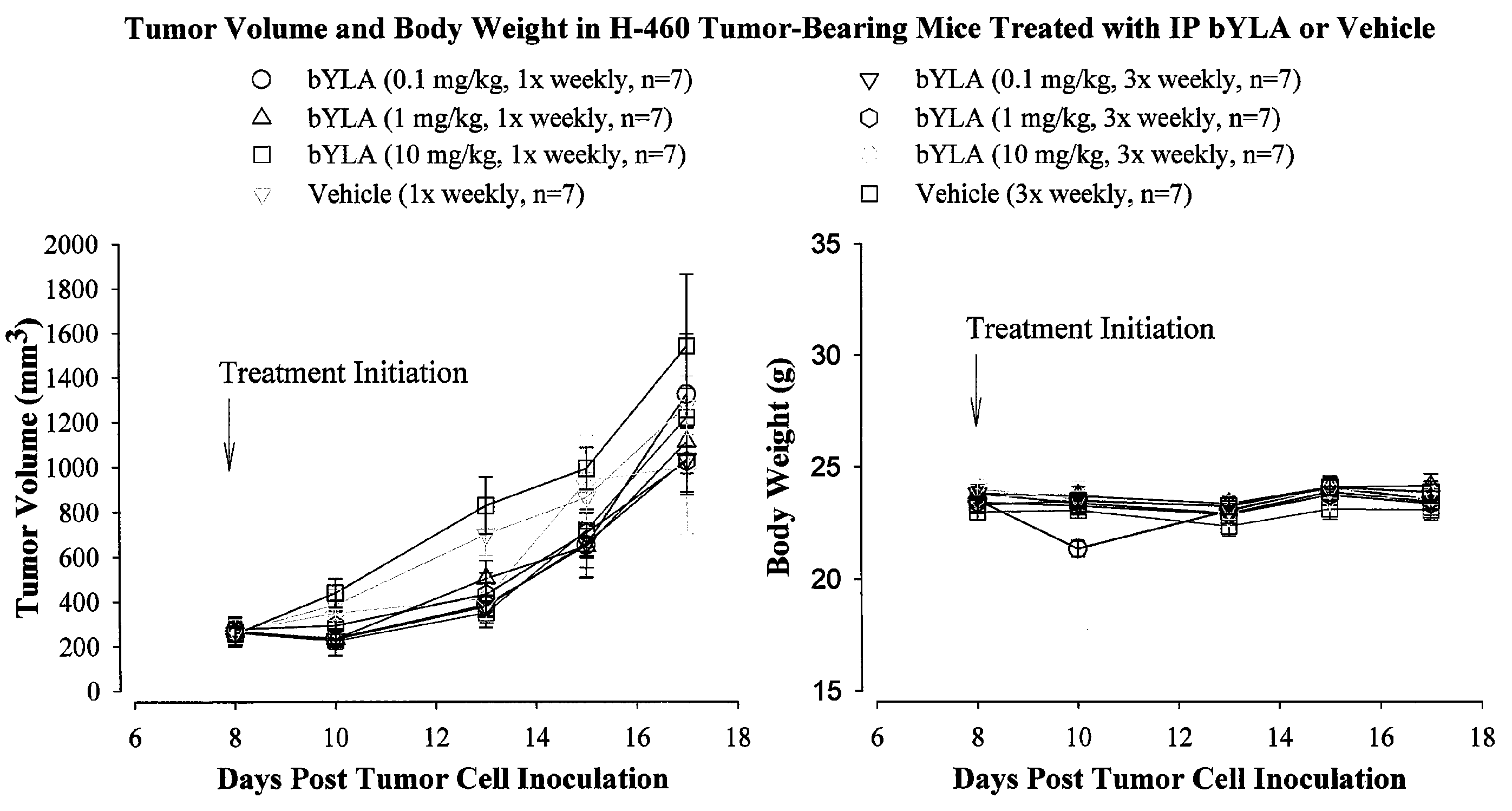
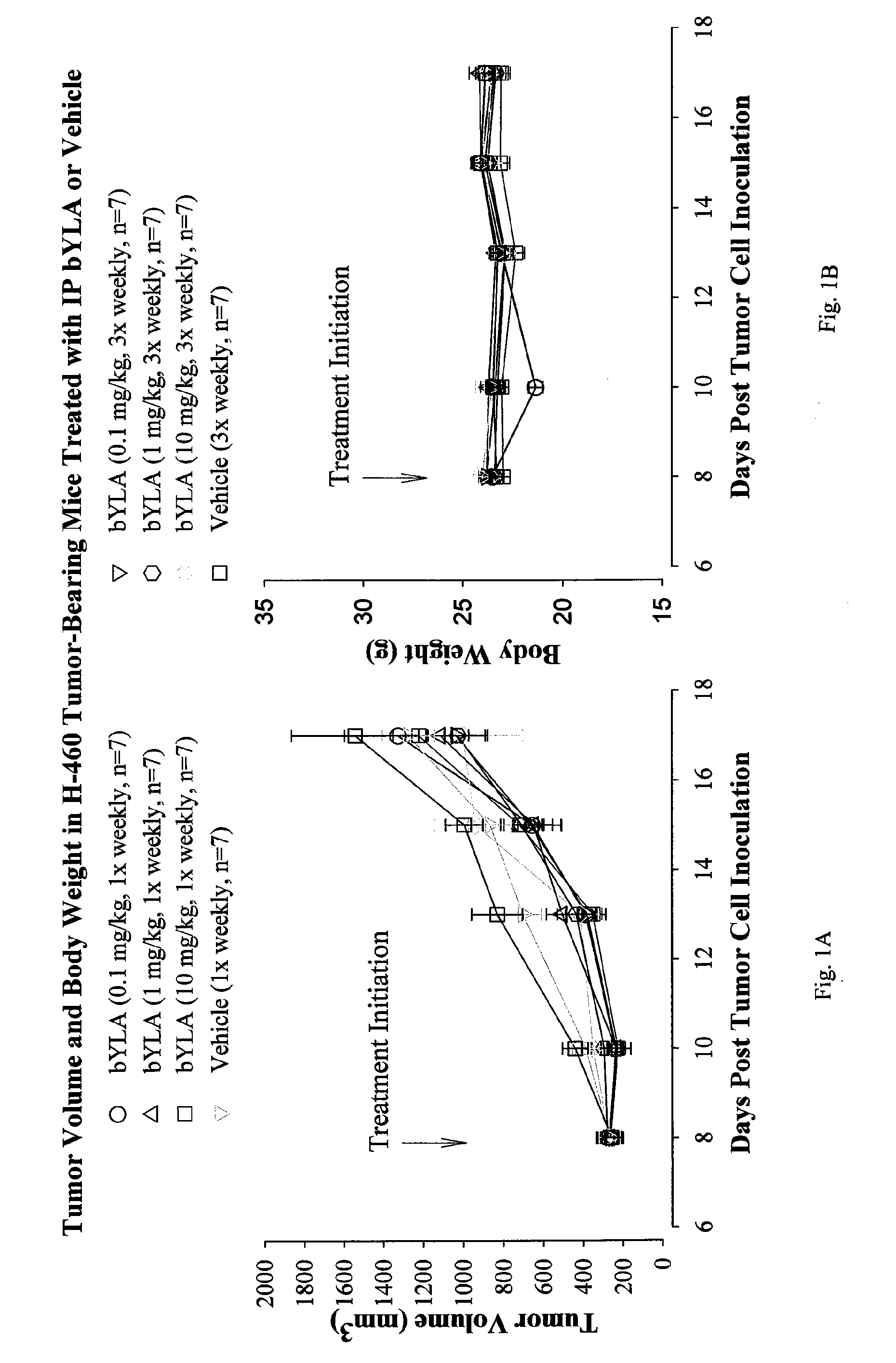
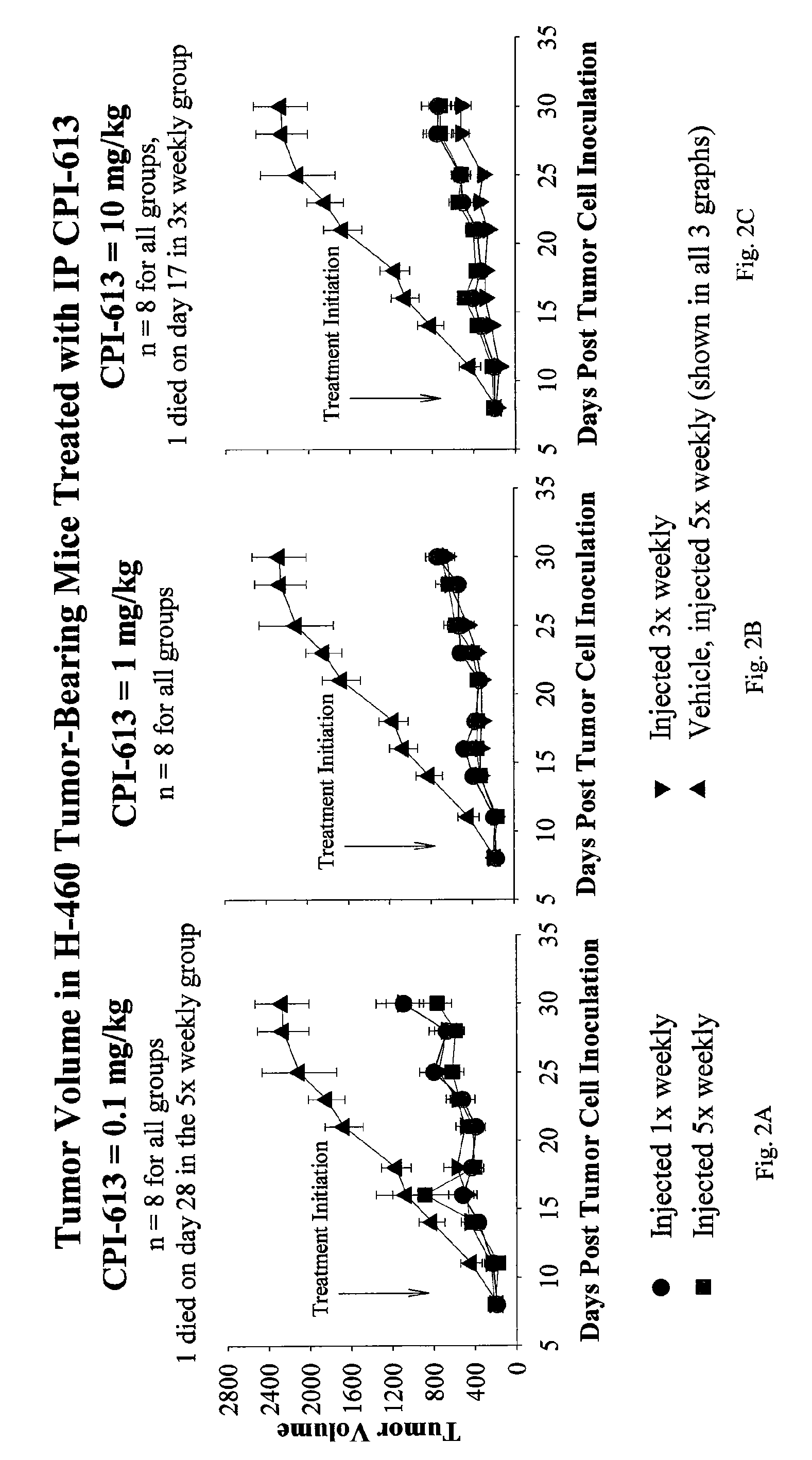
[0055]Bis-benzyl lipoate was provided in a concentrated form at a concentration of 50 mg / mL dissolved in 1M triethanolamine (TEA). The stability of the drug product was assessed by visual observation and by high-performance liquid chromatography (HPLC) assessment, performed at the beginning and the end of the study. The physical appearance did not change and the purity was found to be >99% pure, both at the beginning and the end of the study. The concentrated bis-benzyl lipoate solution was diluted to an appropriate concentration with 5% dextrose (D5W) to formulate 0.1, 1 and 10 mg / kg doses of bis-benzyl lipoate.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Percent by mass | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
Login to View More
Abstract
Pharmaceutical formulations containing lipoic acid derivatives are made.
Description
[0001]This application claims the benefit of U.S. Provisional Patent Application No. 60 / 912,605, filed Apr. 18, 2007.BACKGROUND OF THE INVENTION[0002]1. Field of the Invention[0003]The present invention is directed to pharmaceutical formulations containing lipoic acid derivatives or salts thereof which selectively kill tumor cells by altering cancer cell metabolism and signal transduction pathways linked to the Warburg Effect, as well as to methods of treating a subject with such pharmaceutical formulations.[0004]2. Related Background Art[0005]All mammalian cells require energy to live and grow. Cells obtain this energy by metabolizing food molecules by oxidative metabolism. The vast majority of normal cells utilize a single metabolic pathway to metabolize their food. The first step in this metabolic pathway is the partial degradation of glucose molecules to pyruvate in a process known as glycolysis which yields two ATP units. Glycolysis can occur even under hypoxic conditions. Pyru...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): A61K31/381A61K31/19A61P35/00
CPCA61K31/19A61K31/381A61K47/18A61K47/4803A61K31/20A61K2300/00A61K47/541A61K31/205A61P35/00A61P35/02A61P43/00A61K31/385A61K31/4427A61K47/26
Inventor SHORR, ROBERT G.L.RODRIGUEZ, ROBERT J.BHASIN, RAJINDER
Owner CORNERSTONE PHARMA INC