Preparation of recombinant long-acting glucagon peptide analogue
A technology for glucagon and analogues, which is applied in the field of preparation of recombinant long-acting glucagon-like peptide analogues, which can solve the problems of strong biological activity and long half-life, so as to improve curative effect, prolong half-life, and improve protein expression The effect of efficiency and purification product yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0035] Embodiment 1 The cultivation and expression of genetically engineered bacteria
[0036] Shake flask strain culture: 5 Erlenmeyer flasks with a capacity of 1L, each containing 200ml of YPD medium, the medium formula is 10g of yeast extract (1%), 20g of peptone (2%), and 20g of glucose (2%). Dissolve in deionized water and make up to 1L, steam sterilize at 115°C for 20 minutes. Each bottle was inoculated with 100 ul of glycerol strains, and cultured on a shaker at 30° C. at 300 rpm for 24 hours. At this time, the OD600 of the bacterial concentration was between 6 and 20.
[0037] 30L tank fermentation culture: 30L automatic fermentation tank (Biostat c-dcu, Sartourius), specific operation can refer to its instruction manual. 15L of basal salt medium is filled in a 30L tank, and the formula is CaSO 4 2H 2 O0.93g / l, K 2 SO 4 18.2g / l, MgSO 4 7H2O, KOH 4.13g / l, H 3 PO 4 26.7ml / l, 40g / l glycerin were dissolved in deionized water and adjusted to 15L. Connect the pH el...
Embodiment 2
[0038] Example 2 Separation and Purification of Genetically Engineered Bacteria Expression Products
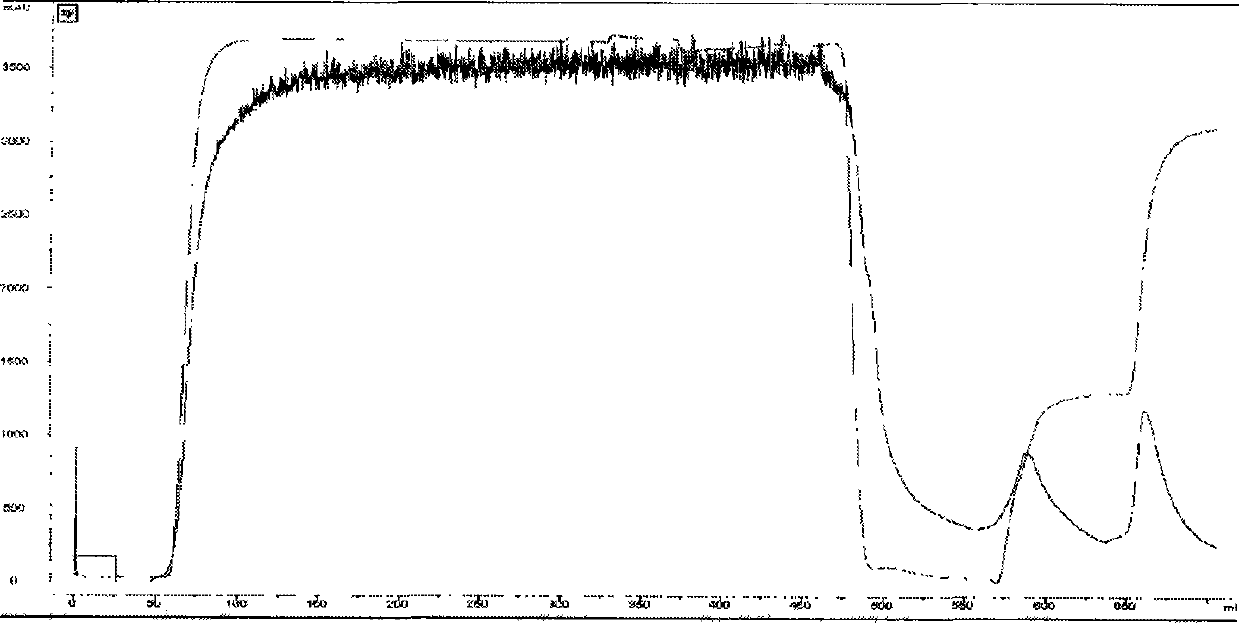
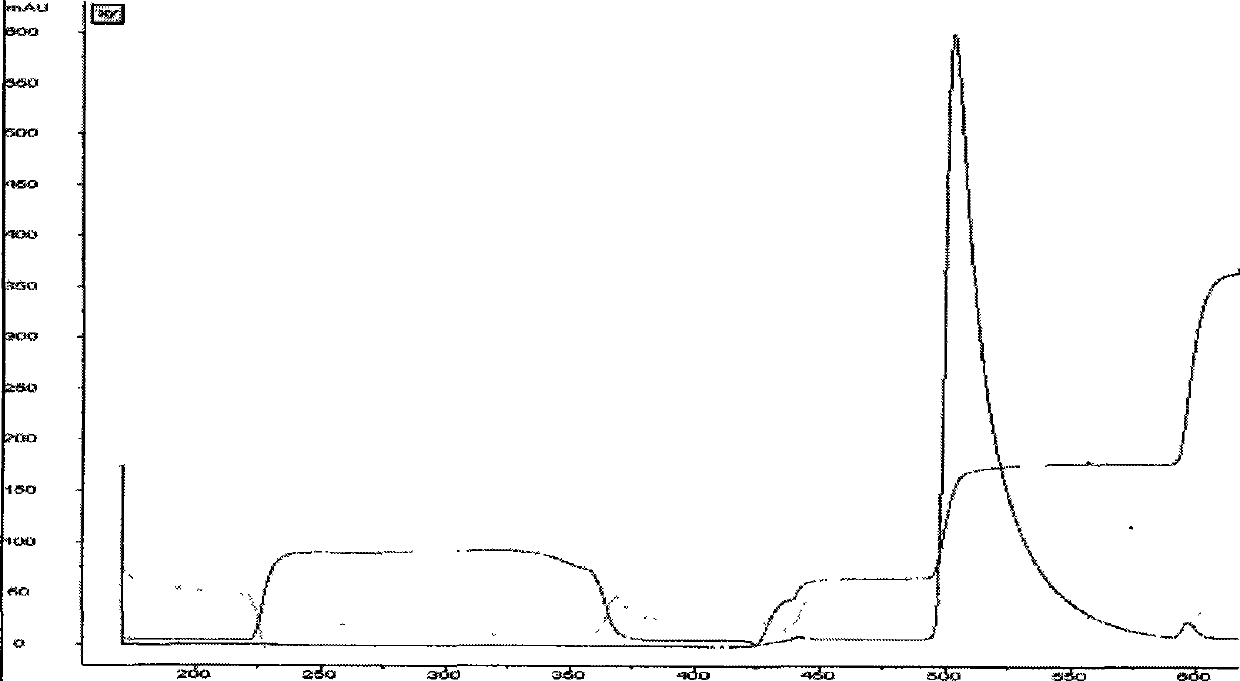
[0039] Blue dye affinity chromatography: adjust the pH of the fermentation supernatant obtained in Example 1 to 7.0 with 6M NaOH, and load it into blue dye affinity chromatography equilibrated with 20 mM phosphate at pH 7.0 and 500 mM NaCl buffer The column has a column bed diameter of 100 mm, a column bed height of 15 cm, and a column bed volume of 1000 ml. The flow rate is 8L / hour, and the sample is loaded after 3 hours. After loading the sample, first wash off unbound substances with 20 mM phosphate buffer at pH 7.0 and 128 mM KSCN buffer, and then wash off the fusion protein bound on the column with 20 mM phosphate buffer at pH 7.0 and 230 mM KSCN buffer. The protein peak was collected for detection by ultraviolet absorption at 280nM, and a total of 3200ml of the eluate was collected.
[0040] Hydrophobic chromatography: Add 630ml of 3M ammonium sulfate solution to the c...
Embodiment 3
[0042] The oral glucose tolerance test of embodiment 3 mice
[0043] Kunming mice aged 8-10 weeks were used, and they were adaptively fed for 1 week after purchase, with free access to food and water. Fasting for 16-18 hours one day before the test, intravenously inject 5 mg / Kg of human serum albumin or the recombinant glucagon-like peptide analog albumin fusion protein injection formulated above according to body weight. One hour after the administration, 1.5 mg / g of body weight glucose was administered by oral infusion, and blood was taken from the tail vein 10 minutes before and 10, 20, 30, 60, and 120 minutes after the administration of glucose, and the glucose level was measured.
[0044] The result is as follows:
[0045] time HSA group (n=6) mM / L E4 / HSA group (n=6) mM / L 10 minutes before sugar 4.2 3.0 10 minutes after giving sugar 9.8 6.3 20 minutes 10.2 6.5 30 minutes 9.6 6.1 60 minutes 6.2 4.3 120 minutes 5.5 3.8...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap