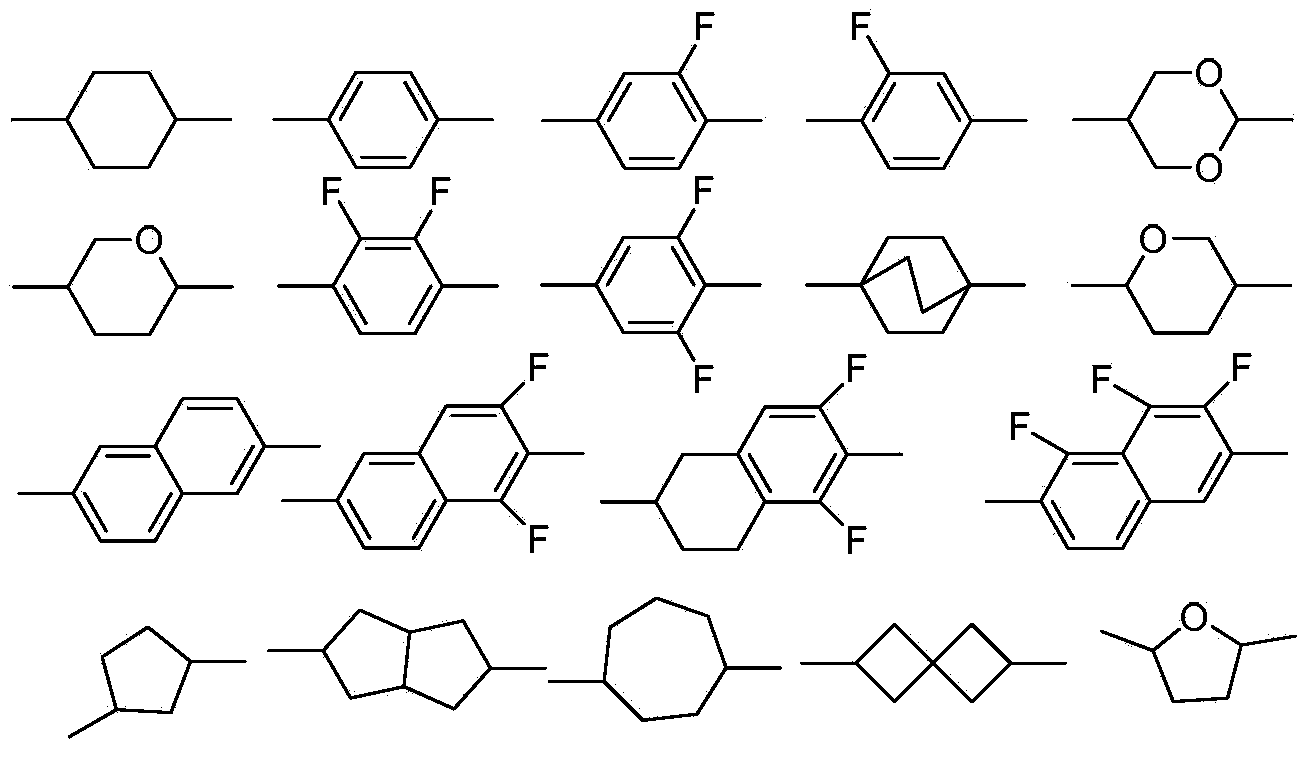
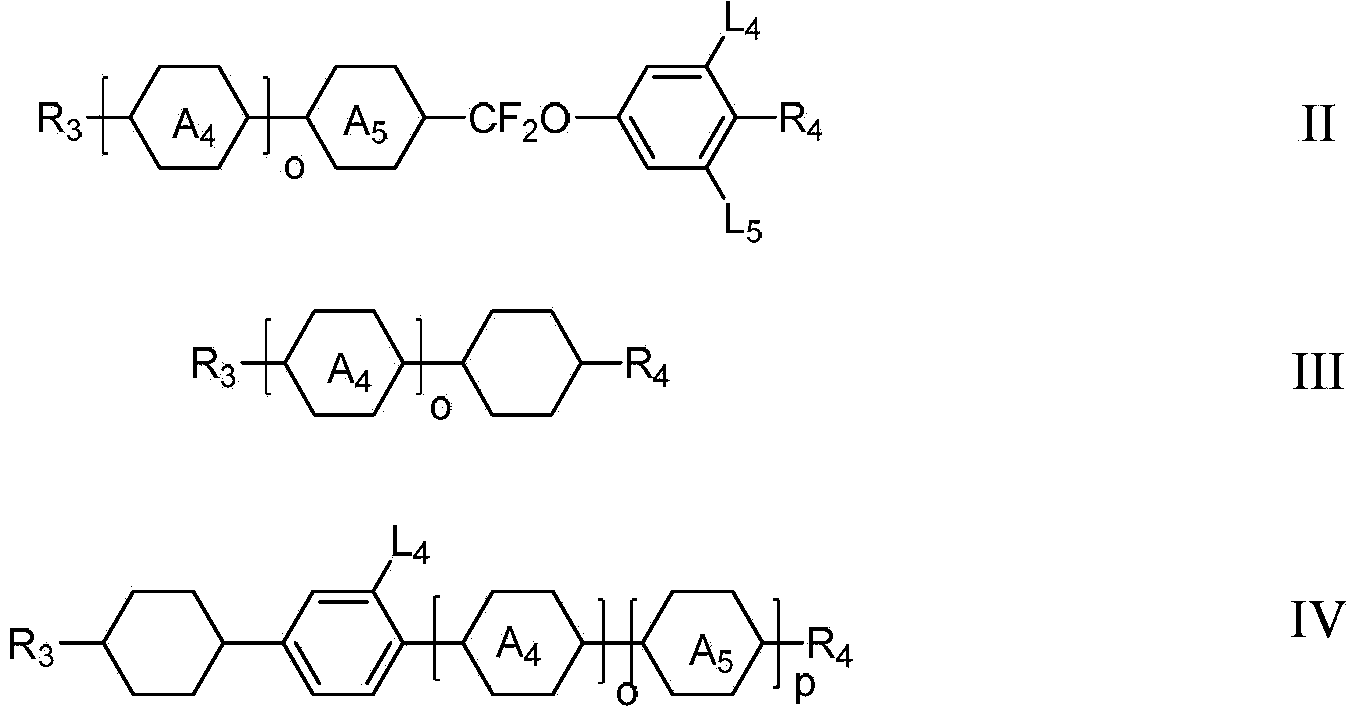
Liquid crystal monomer containing deuterium substitution
A kind of substituent, liquid crystal composition technology, applied in the field of liquid crystal compound synthesis and application
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0132] Embodiment 1, Synthesis
[0133]
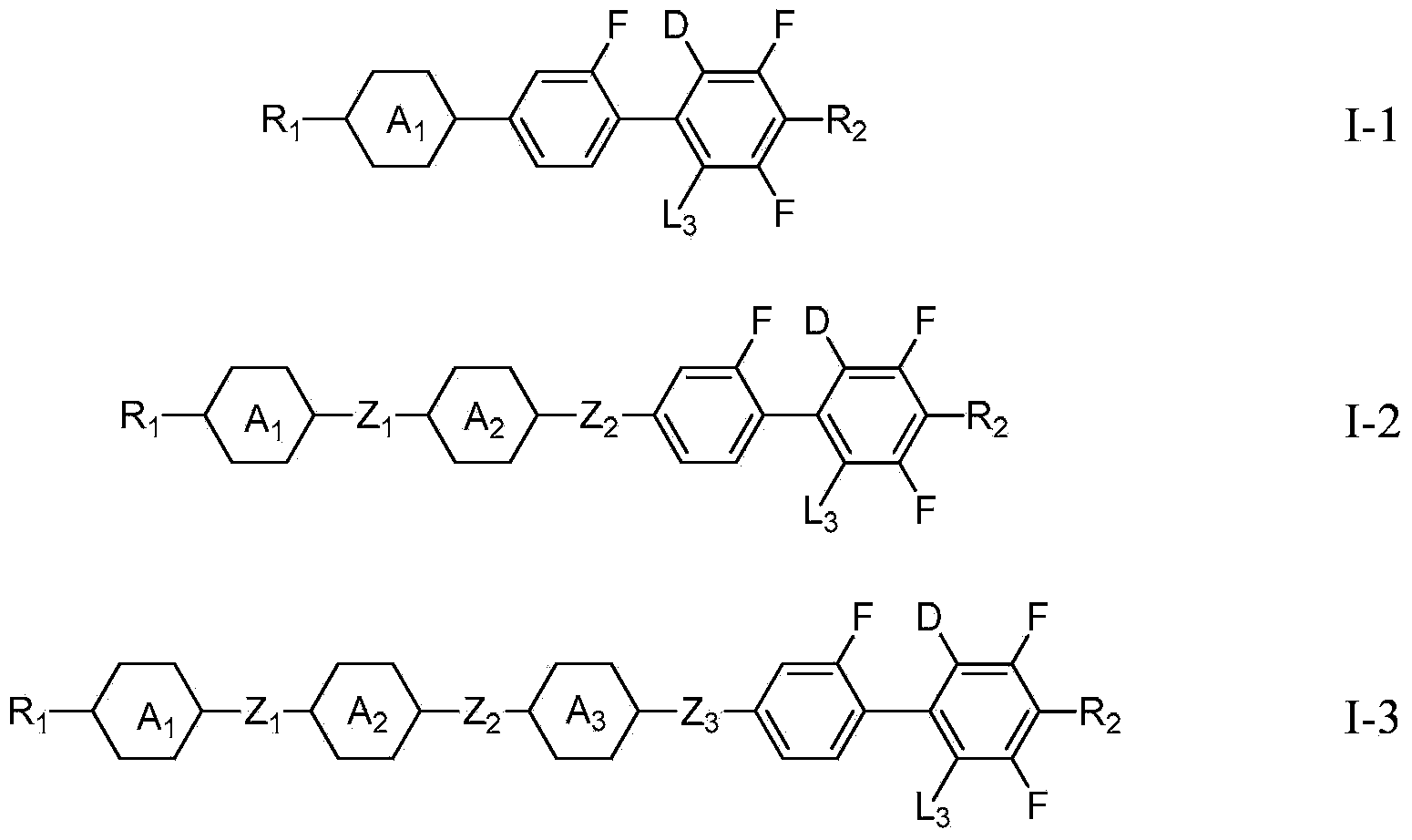
[0134] Synthesis of I-1:
[0135] Add 22.2g (0.11mol) I-1-a (reactant), 18.8g (0.1mol) I-1-b (reactant), 0.1g tetrabutylammonium bromide (phase transfer catalyst) to the reaction flask, 12.7g (0.12mol) sodium carbonate, 50mL toluene (solvent), 15mL absolute ethanol (solvent), 15mL deionized water (solvent), stirring and heating to reflux, adding 0.825g (0.00075mol) tetrakis (triphenylphosphine) palladium, Then reflux for 5h, after cooling down, separate the organic phase in the reaction solution, extract the aqueous phase with 20ml toluene, combine the organic phases, wash with water until neutral, distill the solvent under reduced pressure, dissolve in 100ml petroleum ether, overheated silica gel column, petroleum Rinse with ether, distill petroleum ether under reduced pressure, and recrystallize with 100ml of ethanol to obtain white crystal I-121g, GC: 99.9%, yield: 60%.
[0136] The structural confirmation data of this produc...
Embodiment 2
[0144] Embodiment 2, Synthesis
[0145]
[0146] Synthesis of I-2:
[0147] With reference to the method of the above-mentioned Example 1, only the substituents in the corresponding reactants are replaced according to the definition of the substituents in the product to obtain the compound shown in formula I-2.
[0148] The structural confirmation data of this product are as follows:
[0149] GC-MS: M+35152.3%, 322100%, 1833.6%
[0150] 1 H NMR (300MHz, CDCl 3 )δ7.69(d,J=7.8Hz,1H),7.13(d,J=7.8Hz,1H),6.90(d,J=7.8Hz,1H),7.03–6.90(m,1H),2.72( t,J=7.6Hz,1H),1.66(t,J=7.6Hz,4H),1.52(t,J=7.6Hz,4H),1.43(t,J=7.6Hz,1H),1.31(t, J=7.6Hz,2H),1.25(m,2H),0.99(t,J=7.3Hz,3H).
[0151] It can be seen from the above that the product has a correct structure and is a compound shown in formula I-2.
[0152] The liquid crystal properties of the product are as follows:
[0153] Mp: 62.4°C Cp: 20.3°C
[0154] ?n: 0.1203△ε: 11.56
[0155] It can be seen from the above that this compound ...
Embodiment 3
[0156] Embodiment 3, Synthesis
[0157]
[0158] Synthesis of I-3:
[0159] With reference to the method of the above-mentioned Example 1, only the substituents in the corresponding reactants are replaced according to the definition of the substituents in the product to obtain the compound shown in formula I-3.
[0160] The structural confirmation data of this product are as follows:
[0161]GC-MS: M+45551.6%, 426100%, 1832.7%
[0162] 1 H NMR (300MHz, CDCl 3 )δ7.83(d,J=7.8Hz,1H),7.58(d,J=7.8Hz,1H),7.36(m,4H),7.02(d,J=7.8Hz,1H),7.30–6.93( m,1H),2.72(t,J=7.6Hz,1H),1.67(t,J=7.6Hz,4H),1.54(t,J=7.6Hz,4H),1.43(t,J=7.6Hz, 1H),1.31(t,J=7.6Hz,2H),1.29(m,J=7.6Hz,4H),1.25(m,2H),0.99(t,J=7.3Hz,3H).
[0163] As can be seen from the above, the product has a correct structure and is a compound shown in formula I-3.
[0164] The liquid crystal properties of the product are as follows:
[0165] Mp: 76.8°C Cp: 203.5°C
[0166] Δn: 0.2117 Δε: 19.2
[0167] It can be seen from the...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap