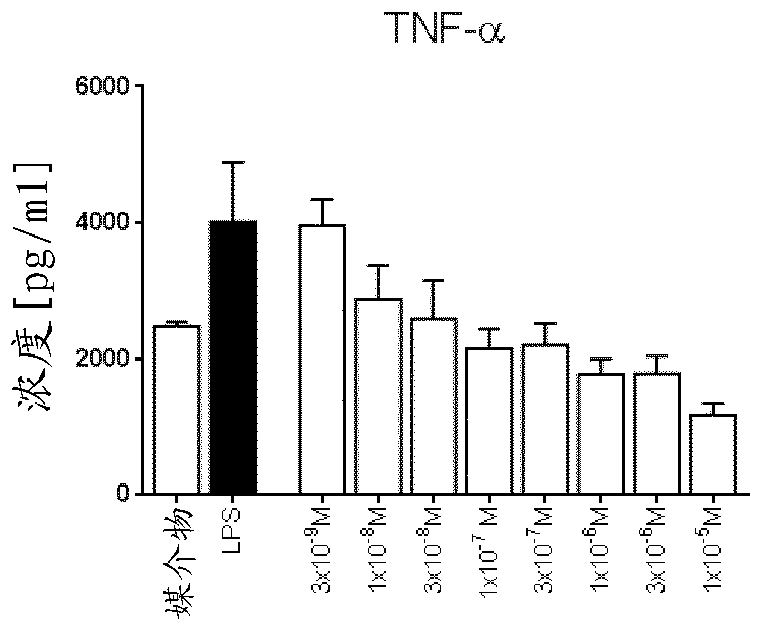
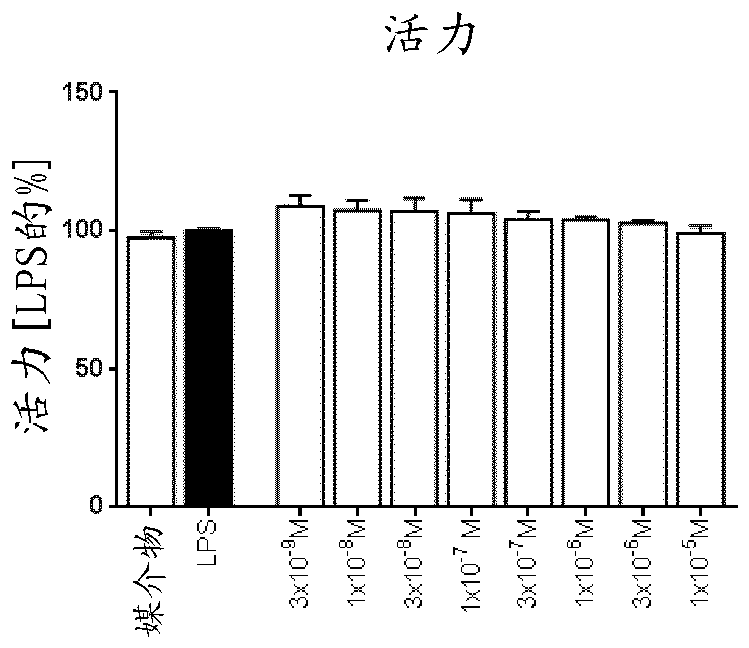
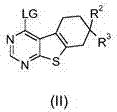
Pyrazolopyridinamines as MKNK1 and MKNK2 inhibitors
A compound, C2-C6- technology, applied in the direction of anti-inflammatory agents, organic chemistry, drug combination, etc., can solve the problems of pyrazolopyridinamine compounds that have not been disclosed to treat or prevent different diseases
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0581] (RS) N,N-Dimethyl-4-(pyrazolo[1,5-a]pyridin-5-ylamino)-5,6,7,8-tetrahydro[1]benzothieno[ 2,3-d]pyrimidine-7-carboxamide
[0582]
Embodiment 1a
[0586] (RS)-4-Chloro-N,N-dimethyl-5,6,7,8-tetrahydro[1]benzothieno[2,3-d]pyrimidine-7-carboxamide
[0587]
[0588] will contain 4.54 g (16.90 mmol) of (RS)-4-chloro-5,6,7,8-tetrahydro[1]benzothieno[2,3-d]pyrimidine-7-carboxylic acid (prepared according to WO2013174744) , 182 mL N,N-dimethylformamide, 8.83 mL N-ethyl-N-isopropylpropan-2-amine, 42.2 mL N-methylmethylamine solution (2M, in tetrahydrofuran) and 40.2 mL 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphorinane 2,4,6-trioxide solution (50%, in N,N- dimethylformamide) was stirred at 23°C for 1 hour. The mixture was poured into water, ice was added, the precipitate was washed with water and ether, and dried to give 3.44 g (65%) of the title compound.
Embodiment 2
[0590] (7S)-4-(pyrazolo[1,5-a]pyridin-5-ylamino)-5,6,7,8-tetrahydro[1]benzothieno[2,3-d]pyrimidine -7-Formic acid
[0591]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com